
Fabrication and characterization of honeycomb-like superstructures consisting of ZnS nanosheets
LI Jun-ping(李军平), XU Yao(徐 耀), ZHAO Ning(赵 宁), WEI Wei(魏 伟),
WU Dong(吴 东), SUN Yu-han(孙予罕)
State Key Laboratory of Coal Conversion, Institute of Coal Chemistry,
Chinese Academy of Sciences, Taiyuan 030001, China
Received 10 April 2006; accepted 25 April 2006
Abstract: Honeycomb-like superstructures consisting of ZnS nanosheets with uniform thickness of about 20nm and lateral dimension of several micrometers were built up from in-situ generated ZnS nanoparticles in the presence of ethylene diamine tetraacetic acid and butylamine. The result of HRTEM reveals that the size of the primary ZnS nanoparticles constituting the superstructures is about 2-4 nm. Upon their organization into complex superstructures, the quantum-confined effect of the ZnS nanosheets keeps well. The possible growth mechanism for ZnS superstructures was also proposed.
Key words: ZnS nanosheets; colloidal assemblies; quantum-confined effect; UV-absorption spectroscopy
1 Introduction
In the past decade, the synthesis of nanoparticles with controlled size, structure and morphology has stimulated intensive interest, which is the first step towards realization of functional nanosystems. Recently, much effort has been exerted in exploring the strategies to organize these nanoscale building blocks into ordered assemblies or superstructures and investigating their collective properties [1-8]. To date, some ordered superstructures or complex functional architectures have been constructed through covalent or non-covalent interconnect strategies. YU et al [9] reported the assem-
bly of BaCrO4 and BaSO4 nanofibers toward hierarchical superstructures using sodium polyacrylate as structure directing agent in mineralization process. SHI et al [10] reported the fabrication of BaCrO4 superstructures from inorganic nanobelts in catanionic reverse micelles. Recently, KIMURA et al [11] have also demonstrated the organization of Au nanoparticles into a 3D network structure by a site-exchange reaction. However, there are few reports concerning the assembly of ZnS building blocks into hierarchical nanostructures or superstruct-ures.
As typical semiconductor materials of the II-VI group, ZnS nanocrystals have been widely investigated. Various forms of ZnS nanostructures have been synthesized, including nanoparticles [12, 13], nanowires [14,15], and nanotubes [16]. Recently, YU et al[17] reported the synthesis of ZnS(NH2CH2CH2NH2)0.5 hybrid nanosheets using the solvothermal routes. Hollow ZnS nanospheres have also been synthesized in aqueous solution of a triblock copolymer [18]. Here, we report the synthesis of honeycomb-like superstructures under mild conditions using EDTA as stabilizer and BA as the structure directing agents.
2 Experimental
In a typical synthesis, 1.095 g Zn(CH3COO)2?2H2O and 1.60 g EDTA were mixed in 50 mL distilled water, together with 5 mL of BA to form a clear liquid. Following, 50 mL of 0.1 mol?L-1 Na2S aqueous solution was added. The color of the system changed gradually form clear to blue, and finally a white precipitate was obtained. After aged for 24 h at room temperature, the precipitate was filtered and washed thoroughly with hot water and ethanol. To further remove organic residues, the resulting products were dispersed in 100 mL ethanol, followed by solvothermal treatment at 140 ℃ for 24 h. The final products were again filtered, washed with ethanol and hexane, and dried in air at 110 ℃. The obtained products were characterized by scanning electron microscopy (SEM), transmission electron microscopy (TEM) and X-ray diffraction (XRD). The XRD patterns were recorded using Rigaku Dmax-2000 diffractometer with Cu Kα radiation. SEM was performed on Philips XL30S-FEG apparatus. TEM was recorded on a Hitachi Model H-9000NAR instrument with accelerating voltage of 200 kV. UV-vis diffusion reflection spectrum was collected at room temperature on Shimadzu UV-2501PC using BaSO4 powder as a standard.
3 Results and discussion
Fig.1 shows the typical SEM images of as-synthesized products. It reveals that the product exhibits sheet-like morphology and a large number of sheets are arranged in bundles of honeycomb-like superstructures. A higher magnification SEM image indicates that the superstructures exclusively consist of sheets with a thickness of about 20nm and lateral dimensions of about 1.0 μm (Fig.1(b)). Furthermore, each sheet is built of thin layers in stacks.

Fig. 1 SEM images of sample: (a) Low magnification; (b) High magnification
The structure and morphology of the products were further characterized by TEM. The TEM studies suggest that the products are composed of sheets with irregular shape, and no other morphology is observed. A top-view of a sheet is shown in Fig.2(a), the sheets are not perfect planes with lateral dimensions of about 1.0 μm. A high magnification image of the sheets (Fig.2(b)) reveals that the sheets are stacked by a few of very thin layers. HRTEM observation (Fig.2(c)) shows that each layer consists of nanosized ZnS grains with diameter of about 2-4 nm, indicating the layer is built up from ZnS nanoparticles. Energy-dispersive X-ray spectroscopy (EDX) of the samples shows that atomic composition ratio of Zn/S is always about 1∶1, which is in good agreement with that of the stoichiometric composition of ZnS.

Fig.2 TEM images of sheet: (a) Low magnification; (b) High magnification; (c) High resolution
A typical XRD pattern of the samples is shown in Fig.3. All the peaks can be indexed to the hexagonal ZnS (JCPDS: 36-1450), corresponding to the (100), (002), (101), (102), (110), (103) and (112) crystal planes of wurtzite ZnS, respectively. No extra peaks or any impurities are detected, indicating the high purity of the products.
To examine the optical properties of ZnS superstructures, the room temperature UV-vis diffusion reflection spectrum of the solid powders are shown in
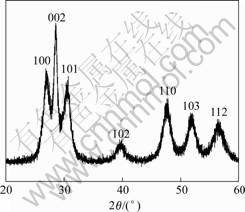
Fig.3 XRD pattern of sample
Fig.4. Compared with bulk ZnS (340 nm), the absorption spectrum of the ZnS superstructures (290 nm) shows a great blue shift, indicating that the ZnS superstructures are quantum-confined. It is reported that the optical absorption of the ZnS nanocrystals with a size of 3.5 nm exhibits an absorption peak at 288 nm. Presented here, the variation of band gap versus the size of ZnS nanocrystals indicated that an absorption peak at 290 nm should correspond to a band gap of 4.27 eV, would correspond to a crystal size of about 3.6 nm [20], which is in good agreement with the size of the ZnS nanoparticles constituting the superstructures (Fig.2(c)). Therefore, the quantum-confined effect of the ZnS superstructures is attributed to the collective behavior of the primary ZnS nanoparticles with size of about 3.6 nm and essentially does not change upon their organization into superstructures.
It is found that EDTA and BA play important roles in the formation of the superstructures. In the absence of EDTA or BA, ZnS nanoparticles are formed but no sheet-like nanosturctures are observed. The possible growth mechanism is proposed as Fig.5. At first, as stabilizing agents, EDTA binds to the surface of freshly generated ZnS, preventing their growth to the bulk phase. At the meantime, BA molecules may form a lamellar liquid crystal template to provide ideal microenvironments for the oriented growth of in-situ generated ZnS nanoparticles into hybrid ZnS thin layers. It is evident from many studies that superstructures with complex form and hierarchy may result from mesoscale transformation of hybrid inorganic-organic building blocks through strong binding interactions between adsorbed organic molecules and the inorganic surfaces [1]. Similarly, the generated hybrid layers interact each other and stack into nanosheets, and then overgrow into honeycomb-like superstructures, as shown in Fig.5. Further work is underway to understand the structural evolvement process, but it is believed that ZnS superstr-
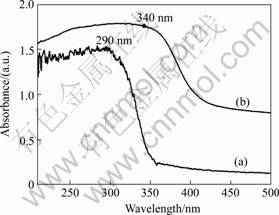
Fig.4 UV-vis absorption spectrum: (a) Obtained ZnS superstr-uctures; (b) Bulk ZnS powder
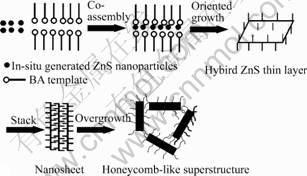
Fig.5 Schematic illustration of growth process of ZnS superstructures
uctures result from the cooperative-role of EDTA and BA.
4 Conclusions
Honeycomb-like ZnS superstructures have been produced via a feasible synthesis route using BA as structure-directing agent and EDTA as stabilizer. The obtained superstructure consists of ZnS nanosheets with uniform thickness of about 20 nm and lateral dimension of several micrometers. Such a superstructure shows strong quantum-confined effect, and thus has potential application in optical and electronic devices or biotechnology.
References
[1] C?LFEN H, MANN S. Higher-order organization by mesoscale self-Assembly and transformation of hybrid nanostructures [J]. Angew Chem Int Ed, 2003, 42(21): 2350-2365.
[2] HOPWOOD J D, MANN S. Synthesis of barium sulfate nanoparticles and nanofilaments in reverse micelles and ,icroemulsions [J]. Chem Mater, 1997, 9(8): 1819-1828.
[3] QI L, MA J, CHENG H, ZHAO Z. Reverse micelle based formation of BaCO3 nanowires [J]. J Phys Chem B, 1997, 101(18): 3460-3463.
[4] POLLEUX J, PINNA N, ANTONIETTI M, NIEDERBERGER M. Ligand-directed assembly of preformed titania nanocrystals into highly anisotropic nanostructures [J]. Adv Mater, 2004, 16(5): 436-439.
[5] TANG Z, KOTOV N, GIERSIG M. Spontaneous organization of single CdTe nanoparticles into luminescent nanowires [J]. Science, 2002, 297(5579): 237-240.
[6] ZHENG J, ZHANG Y, LI Z, WEI W, WU D, SUN Y. Hydrothermally stable mesoporous aluminosilicates with tubular morphology [J]. Chem Phys Lett, 2003, 376(1-2): 136-140.
[7] LI M, MANN S. Emergence of morphological complexity in BaSO4 fibers synthesized in AOT microemulsions [J]. Langmuir, 2000, 16(17): 7088-7094.
[8] DUJARDIN E, HSIN L. B, WANG C. R. C, MANN S. DNA-driven self-assembly of gold nanorods [J]. Chem Commun, 2001(14): 1264-1265.
[9] YU S H, ANTONIETTI M, C?LFEN H, HARTMANN J. Growth and self-assembly of BaCrO4 and BaSO4 nanofibers toward Hierarchical and repetitive superstructures by polymer-controlled mineralization reactions [J]. Nano Lett, 2003, 3(3): 379-382.
[10] SHI H, QI L, MA J, CHENG H, ZHU B. Synthesis of hierarchical superstructures consisting of BaCrO4 nanobelts in catanionic reverse Micelles [J]. Adv Mater, 2003, 15(19): 1647-1651.
[11] KIMURA M, KOBAYASHI S, KURODA T, HANABUSA K, SHIRAI H. Assembly of gold nanoparticles into fibrous aggregates using Thiol-Terminated Gelators [J]. Adv Mater, 2004, 16(4): 335-338.
[12] WANG W Z, GERMANENKO I, EI-SHALL M S. Room-temperature synthesis and characterization of nanocrystalline CdS, ZnS, and CdxZn1-xS [J]. Chem Mater, 2002, 14(7): 3028-3033.
[13] DONAHUE E J, ROXBURGH A, YURCHENKO M. Sol-gel preparation of zinc sulfide using organic dithiols [J]. Mater Res Bull, 1998, 33(2): 323-329.
[14] JIANG X C, XIE Y, LU J, ZHU L Y, HE W, QIAN Y T. Simultaneous in situ formation of ZnS nanowires in a liquid crystal template by γ-irradiation [J]. Chem Mater, 2001, 13(4): 1213-1218.
[15] WAGN Y, ZHANG L, LIANG C, WANG G, PENG X. Catalytic growth and photoluminescence properties of semiconductor single-crystal ZnS nanowires [J]. Chem Phys Lett, 2003, 357(3-4): 314-318.
[16] WANG X, GAO P, LI J, SUMMERS C J, WANG Z L. Rectangular porous ZnO-ZnS nanocables and ZnS nanotubes [J]. Adv Mater, 2002, 14(23): 1732-1735.
[17] YU S H, YOSHIMURA M. Shape and phase control of ZnS nanocrystals: template fabrication of wurtzite ZnS single-crystal nanosheets and ZnO flake-like dendrites from a lamellar molecular precursor ZnSNH2CH2CH2NH2)0.5 [J]. Adv Mater, 2002, 14(4): 296-300.
[18] MA Y, QI L, MA J, CHENG H. Facile synthesis of hollow zns nanospheres in block copolymer solutions [J]. Langmuir, 2003, 19(9): 4040-4042.
[19] JIANG Y, MEGN X M, LIU J, XIE Z Y, LEE C S, LEE S T. Hydrogen-assisted thermal evaporation synthesis of ZnS nanoribbons on a large scale [J]. Adv Mater, 2003, 15(4): 323-327.
[20] NANDA J, SAPRA S, SARMA D D, CHANDRASEKHRAN N, HODES G. Size-selected zinc sulfide nanocrystallites: synthesis, structure, and optical studies [J]. Chem Mater, 2000, 12(4): 1018-1024.
(Edited by HE Xue-feng)
[U1] Foundation item: Project (20133040) supported by the National Natural Science Foundation of China
Corresponding author: XU Yao; Tel: +86-351-4049859; Fax: +86-351-4041153; E-mail: xuyao@sxicc.ac.cn