铜陵重金属富集的尾矿废弃地酸性废水中细菌及古菌的多样性
来源期刊:中国有色金属学报(英文版)2014年第10期
论文作者:杨 扬 李 杨 孙庆业
文章页码:3332 - 3342
Key words:acid mine drainage; microbial community; clone library; geochemical variables
摘 要:为了加深对安徽省铜陵市不同重金属富集程度的尾矿附近酸性废水中微生物多样性的了解,运用PCR-16S rDNA克隆文库技术对酸性废水中细菌及古菌的群落结构进行研究。在铜陵的5个样点采集了8个水样。系统发育分析结果表明,酸性废水中的细菌主要是Betaproteobacteria, Gammaproteobacteria, Alphaproteobacteria, Deinococcus-Thermus, Nitrospira, Firmicutes, Actinobacteria, Deltaproteobacteria, Bacteroidetes, Chloroflexi,古菌是Thermoplasma, Ferroplasma 以及Thermogymnomonas。根据细菌和古菌的组成对样品进行聚类分析,结果表明,5个样品由于优势菌株Thermomonas和Meiothermus拥有较高的相似度。2个样品由于Acidithiobacillus 和 Leptospirillum占主导也拥有较高的相似度。剩余的样品多样性较高,拥有最高的香浓-微纳指数2.91。典型相关分析CCA的结果表明微生物的多样性组成与理化因子有密切的关系,如水的pH以及Hg2+、Pb2+、Cl-、SO42-、Fe3+的含量。
Abstract: To expand knowledge on microbial communities of various metal-rich levels of mine drainage environments in Anhui province, China, the archaeal and bacterial diversities were examined using a PCR-based cloning approach. Eight acid mine water samples were collected from five areas in Tongling. Phylogenetic analyses revealed that bacteria mainly fell into ten divisions, which were Betaproteobacteria, Gammaproteobacteria, Alphaproteobacteria, Deinococcus-Thermus, Nitrospira, Firmicutes, Actinobacteria, Deltaproteobacteria, Bacteroidetes, Chloroflexi. Archaea fell into three phylogenetic divisions, Thermoplasma, Ferroplasma and Thermogymnomonas. The unweighted pair group method with arithmetic mean (UPGMA) cluster analysis based on the microbial communities’ compositions revealed that five samples shared similarity with the dominance of Meiothermus and Thermomonas. Two samples had the preponderant existence of Acidithiobacillus and Leptospirillum. The remaining sample owned higher microbial communities’ diversity with the Shannon-Weaver H up to 2.91. Canonical correlation analysis (CCA) suggested that microbial community structures had great association with pH and the concentration of Hg2+, Pb2+, Fe3+, Cl-, SO42- in water.
Trans. Nonferrous Met. Soc. China 24(2014) 3332-3342
Yang YANG, Yang LI, Qing-ye SUN
School of Resources and Environmental Engineering, Anhui University, Hefei 230601, China
Received 5 September 2013; accepted 27 February 2014
Abstract: To expand knowledge on microbial communities of various metal-rich levels of mine drainage environments in Anhui province, China, the archaeal and bacterial diversities were examined using a PCR-based cloning approach. Eight acid mine water samples were collected from five areas in Tongling. Phylogenetic analyses revealed that bacteria mainly fell into ten divisions, which were Betaproteobacteria, Gammaproteobacteria, Alphaproteobacteria, Deinococcus-Thermus, Nitrospira, Firmicutes, Actinobacteria, Deltaproteobacteria, Bacteroidetes, Chloroflexi. Archaea fell into three phylogenetic divisions, Thermoplasma, Ferroplasma and Thermogymnomonas. The unweighted pair group method with arithmetic mean (UPGMA) cluster analysis based on the microbial communities’ compositions revealed that five samples shared similarity with the dominance of Meiothermus and Thermomonas. Two samples had the preponderant existence of Acidithiobacillus and Leptospirillum. The remaining sample owned higher microbial communities’ diversity with the Shannon-Weaver H up to 2.91. Canonical correlation analysis (CCA) suggested that microbial community structures had great association with pH and the concentration of Hg2+, Pb2+, Fe3+, Cl-, SO42- in water.
Key words: acid mine drainage; microbial community; clone library; geochemical variables
1 Introduction
Acid mine drainage (AMD) water is a worldwide environmental problem caused by active and abandoned mines [1]. It has caused severe environmental problems, including contamination of rivers, streams and ground water. AMD arises when metal sulfide minerals, particularly pyrite (FeS2), are exposed to oxygen and water during the mining of metals and coals [2]. The overall procedure can be described as follows: FeS2+ 14Fe3++8H2O→15FeS2++2SO42-+16 H+ [3], and the ongoing oxidation of sulfide minerals really depends on the regeneration of ferric iron. At pH values below 4, the rate of chemical oxidation of ferrous iron by O2 is negligible [4]; therefore, the activities of acidophilic iron-oxidizing microorganisms play a pivotal role in converting ferrous to ferric iron in acidic environment [1]. Due to the low pH of AMD, the solubility of transition metals is greater and so AMD often typically contains elevated concentrations of metals, including iron, aluminium, manganese and other toxic transition metals who present depending on the mineralogy of the host rock [3].
Despite the acidic nature and elevated dissolved metal concentrations, the water still has the presence of microorganisms. Previous studies based on the diversity of microorganisms in AMD with extremely low pH (<2) showed that acidophilic bacteria mainly included a group of sulfur and/or iron-oxidizers, classifying into Acidithiobacillus ferrooxidans, Acidithiobacillus thiooxidans, Leptospirillum ferrooxidans and Ferroplasma spp. [4,5]. With the application of unculture-based molecular phylogenetic techniques, the understanding of the diversity of acidophilic microorganisms was accelerated, including Legionella, Thiomanas and Gallionella [6].
The distribution of acidophiles in AMD has close association with temperature, pH and metal concentrations [3]. In general, mesophilic acidophiles can be exclusively detected in AMD, though increased temperature makes the moderate thermophiles dominate in the microbial populations, such as Bacillus spp. and Sulfobacillus acidophilus [7]. Conversely, in many low- temperature AMD waters, Acidithiobacillus ferrivorans, the newly described psychrotolerant species, tend to dominate over A. ferrooxidans and L. ferrooxidans. Another controlling factor of microbial populations is pH. A. ferrooxidans and L. ferrooxidans, as the typical iron-/sulfur-oxidizing microbes, dominate in extreme acidic environment (pH<2); the moderate acidophiles, such as Thiomonas and Halothiobacillus, dominate in moderately acidic AMD (pH 3-6) [1,8]. The low pH of AMD results in the solution of metals. The composition of AMD is also influenced by the concentrations of metals [3], such as Fe, Cu, and As. Besides, the low concentration of arsenic might facilitate a higher community proportion of Acidithiobacillus [9]. The composition of various tailing ponds may influence the dissolved elements in AMD. Previous studies showed that the microbial communities differed with the various tailing ponds. HAO et al [6] detected the dominance of chemoorganotrophic and photosynthetic metabolisms, such as Acidiphilium, based on the research of sulfide mine in Xiang Mountain, Anhui province. XIAO et al [10] showed that samples collected from Yunfu sulfide mine were rich in Acidithiobacillus, while waters from Yinshan lead-zinc mine were dominanted in Leptospirillum; in addition, there was no detection of archaea in the study [10].
Although there have been some reports on the bacterial ecology of AMD, few detailed studies were on the various acidification levels of AMD involving different abandoned tailing ponds. The major objectives of this study were: to investigate the archaeal and bacterial communities of mine drainage from different mineral compositions of five main tailing ponds using a PCR-based cloning approach, and to explore the geochemical factors, resulting in community changes of mine drainage in tailing ponds.
2 Experimental
2.1 Sites description
Tongling, located in the southern bank of the middle and lower reaches of Yangtze River, has the subtropical monsoon climate, with an annual average temperature of 16.2 °C and the summer average temperature of 27.4 °C. On average, the frost-free period is 237-258 d, and rainfall is abundant with the mean annual precipitation of 1390 mm and mean annual humidity of 75%-81% [11,12].
Among these six study areas, there were five tailing ponds, Jinkouling, Tongguanshan, Shuimuchong Yangshanchong and Heishahe, discarded from 1980s to 2010s; the rest one was still under use, named Chaoshan gold mine [13]. Sample A was collected from Jinkouling tailing pond, which had been discarded for 20 years; and the bed rock was composed of pyrrhotite, pyrite, magnetite, hematite, limonite, siderite and a small amount of chalcopyrite. Tongguanshan tailing pond was discarded in 1991, from which samples B and C were collected. Samples D and G were collected from Shuimuchong tailing pond and Chaoshan gold mine, respectively. There were four samples collected from Yangshanchong tailing pond, named E, F, H and I. This tailing pond began natural ecological restoration process after being discontinued in 1991. Tailing wasteland is mainly composed of stone powder and sandy texture, and the main mineral in tailings is calcium iron (aluminum) garnet, quartz, feldspar and pyroxene. Sample J was collected from Heishahe tailing pond, which has been discharged for more than 50 years. Due to the fluctuation of the Yangtze River, many naturally colonized plants grew here; moreover, oilseed rape and some other crops were planted by the local farmers, so tailing substrate was greatly improved [11].
2.2 Samples collection
A total of ten AMD samples were collected from five different tailing ponds in Tongling, Anhui Province, China, on February, 2012. The pH values of samples B (7.83) and J (6.08) went over 6.0, so they did not belong to the acidic waste water. They were excluded from the samples. For the eight remaining samples, approximately 10 L of water was collected from these tailing ponds, and then transported to the laboratory within 24 h. About 500 mL original water of each sample was taken out for geochemical analysis, and the remaining water of each sample was filtered through 0.22 μm hyper filtration membrane with vacuum pump, respectively. These filters were then immediately transferred to a tube and stored at -20 °C until they were used for analysis. The filtered water samples were for chemical analysis.
2.3 Geochemical analysis of water samples
Latitude and longitude of sampling sites were determined using a hand holding GPS. The measurements of SO42-, Cl- and PO4-P were carried by ion chromatography (ICS-1500), NH4-N and NO3-N by UV spectrophotometry.
Elemental analysis was carried out using inductively coupled plasma-atomic emission spectrometry (ICP-AES) (XSP Intrepid II, USA). Each sample was tested in the presence of 20 elements, Al, As, Ba, Be, Ca, Cd, Co, Cu, Fe, Hg, K, Li, Mg, Mn, Na, Ni, Pb, Se, Ti and Zn.
2.4 DNA extraction and purification
Each millipore filter was cut and placed into a 1.5 mL falcon tube, and then nucleic acids extraction was carried out according to the established procedures [13]. The crude DNA was purified by agarose gel electrophoresis (1%) and the Wizard DNA Clean-Up Kit (Promega, Madison, Wis.). DNA was quantified by UV-visible spectrophotometry (Thermo, USA).
2.5 PCR and amplification of 16S rDNA
In the reactions, bacterial 16S rDNA genes were amplified with the primer set 27F (5’-AGAGTTTGATCCTGGCTCAG-3’) and 1492R (5’-CGGCTACCTTGTTACGACTT-3’). A PCR amplifier (Biometra, T-Grandient, Germany) was used to incubate reactions through an initial denaturation at 94 °C for 2 min, followed by 35 cycles at 94 °C for 40 s, 55 °C for 30 s, and 72 °C for 1 min, and completed with an extension period of 10 min at 72°C. Products from the amplification reactions of expected size (about 1500 bp) were pooled and purified before ligation later.
PCR amplification of archaeal 16S rDNA genes was carried out following the PCR reactions described as above with archaea-specific primer set: Arch-21F(5’-TTCYGGTTGATCCYGCC RGA-3’) and Arch-958R (5’-YCCGGCGTTGAMTCCAWTT-3’) to yield 900 bp PCR products.
2.6 Cloning and sequencing
The purified PCR products were ligated to the pEASY-T3 cloning vector using a rapid ligation kit according to the instructions of the manufacturer (Transgen, Beijing, China), and then transformed to competent Escherichia coli cells (Transgen, Beijing, China). The transformed cells were plated onto Luria-Bertani agar plates in the presence of ampicillin. After about 14 h of incubating at 37 °C, about 150 white colonies were randomly selected from each library, followed by the sequencing of single clone (BGI, Beijing, PRC).
2.7 Phylogenetic analysis
The nucleotide sequences were manually checked for chimeras using Ribosomal Database Project II. Identified chimeric sequences were discarded. Operational taxonomic units (OTUs) were determined by neighbor-joining analysis of Mega 4.0, which shows the phylogenetic relationships of bacterial 16S rRNA gene sequences recovered from these AMD samples. The coverage of clone libraries was calculated using the equation: Rcov=1-n/N, where n is the number of clones that occurred only once and N is the total number of analyzed clones in each clone library [14].
2.8 Statistical methods
Canonical correspondence analysis (CCA) was performed to analyze the geochemical variables and community structure with CANOCO (Version 4.5, Biometris-Plant Research International, Netherlands) (Terbraak 1988). To evaluate the community diversity, Shannon-Weaver index (H) was calculated using the formula: H=-∑(pi)(lnpi), where pi is the relative abundance of the clone sequenced in each sample. Unweighted pair-group method with arithmetic means (UPGMA) was performed to reveal the distance relationship between sample by the archaeal and bacterial communities. Statistical product and service solution (SPSS) were conducted to reveal the correlation between geochemical variable and community structure.
2.9 Nucleotide sequence accession number
Sequences have been submitted to GenBank with accession numbers as follows: KC537424-KC537737, KC620584-KC620676, KC620745- KC620899 and KC749063-KC749417.
3 Results
3.1 Physico-chemical characteristics of samples
The samples were all collected from the exposed pit outdoor, and the temperatures of them were all about 10 °C. The pH values of the eight sites were not substantially different from each other in the range of 2-4. The pH values of samples A, D, G, I were under 2.5, and the colors of these samples were dark red, especially for sample D, with the strongest acidity (pH=2.04) and the deepest color (brown). The samples all contained high levels of SO42-, Ca2+, Fe3+ and Mg2+; and the concentration of Ca2+ was even higher than that of Fe3+, maybe resulting from the surrounding tailings(Table 1). Besides, sample D had the highest concentrations of SO42-, nitrate and electrical conductivity (EC). The ionic contents of samples A, G and I were also generally higher than other samples, and the ion concentrations varied widely. As shown in Table 1, sample C had the highest concentration of Mg, while sample E had the highest NH4-N, sample F had the highest PO43--P. Meanwhile, other ion concentrations also fell in a wide range. As to the rest ions, Co, Cd, Ba, Li, Hg levels were ignorable.
3.2 Analysis of 16S rDNA cloning libraries
The PCR products of 16S rDNA gene with the expected size (bacteria 1500bp and archaea 900bp) were successfully amplified from community genome DNA of the samples. After TA cloning, a total of 856 positive colonies (603 for bacteria, 253 for archaea) were obtained from the eight AMD samples.
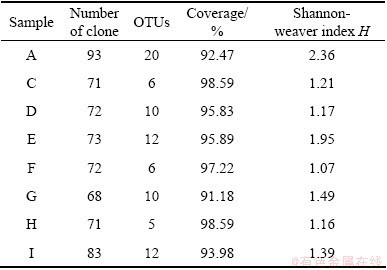
In order to test whether the analyzed clones could represent the bacteria community in each sample, the coverages of all clone libraries were calculated (Table 2). All samples had the coverage over 90%, which suggested that the obtained data could represent the real communities of bacteria. Number of OTUs in each sample is quite different from each other, with a range of 5-20. Sample A had the most OTUs (27), sample E (12) and sample I (12) the second, sample C (6), sample F (6) next, sample H (5) was the lowest. This suggested that there contained less kinds of bacteria in samples C, F and H.
Table 1 Physico-chemical characteristics of collected AMD samples
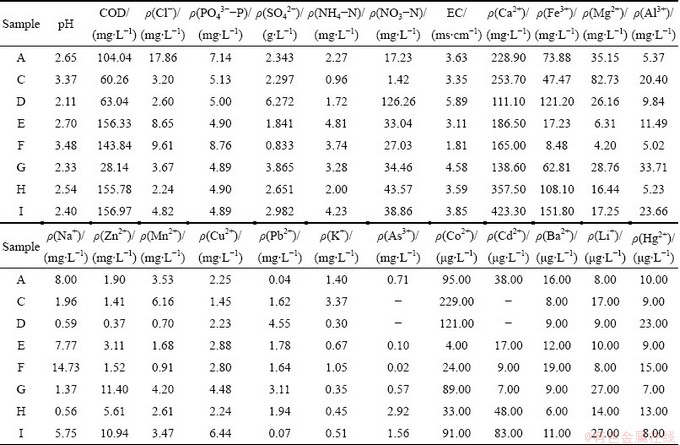
Table 2 Analyses of bacterial 16S rDNA clones libraries constructed for AMD from Tongling
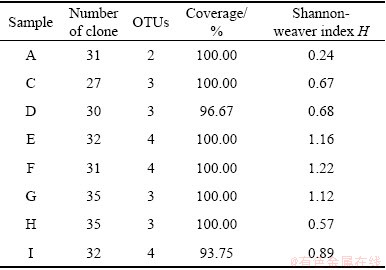
There were at least 27 archaeal cloning sequences obtained for each sample, but only four patterns of OTUs were found, and not all samples contained the four OTUs (Table 3). From the coverage, it could be shown that the number of clones tested in the experiment was sufficient to detect the level of archaeal community diversity within these eight samples.
Table 3 Analyses of archaeal 16S rDNA clones libraries constructed for AMD from Tongling
3.3 Phylogenetic analysis of bacterial sequences
Phylogenetic analysis was established by a bootstrap neighbor-joining method, and the 16S rRNA gene sequences of the bacterial library fell into ten phylogenetic divisions: Betaproteobacteria, Gammaproteobacteria, Alphaproteobacteria, Deinococcus-Thermus, Nitrospira, Firmicutes, Actinobacteria, Deltaproteobacteria, Bacteroidetes, Chloroflexi and some unknown bacterial clusters (Table 4).
The Gammaproteobacteria group was ubiquitous in these eight samples. For samples A, D, I, the group was mainly classified by Acidithiobacillus with the proportion of 17.20% (A), 70.83% (D) and 40.96% (I) of total bacterial clones. Sample A still contained the genus of Halothiobacillaceae and Legionella. For samples C, E, F, G, H, Gammaproteobacteria was almost composed of Thermomonas (45.07%, 31.51%, 40.28%, 35.29% and 43.66%). Betaproteobacteria was detected in samples but sample D, with the dominant in sample A. The group was mainly constituted of Massilia, Herminiimonas (4.30% and 5.38%) in the family of Oxalobacteraceae; Polaromonas (5.38%) in Comamonadaceae; Thiobacillus and Sulfuricella (8.60% and 20.43%) in Hydrogenophilaceae for sample A. Alphaproteobacteria widely distributed in the samples except sample I. For sample D, the group was almost composed of Rhodobacter (5.56%), Phyllobacteriaceae. Samples C, E, F, G and H had the proportions of 7.04%, 4.11%, 9.72%, 14.71%, 8.45%, respectively.
The genus of Meiothermus, in the family of Deinococcus- Thermus, had the largest proportions in samples C, E, F, G and H (39.44%, 27.40%, 47.22%, 36.76% and 39.44%), but it is not detected in other samples. Leptospirillum in the family of Nitrospira appeared in samples A, D, E and I, and gained superiority especially in sample I (42.17%). Firmicutes was detected in samples D, E and I with no significant advantage. Actinobacteria was found in samples A and D, the remaining Deltaproteobacteria was detected in samples E and I; Bacteroidetes, Chloroflexi only appeared in samples A and D, respectively.
The phylogenetic diversity was evaluated for each sample from the clone data by Shannon-Weaver index H. The results showed that sample A (H=2.36) had the preeminent diversity, followed by sample E (H=1.95), while sample F (H=1.07) possessed the lowest H, which was less than half of sample A (Table 3). The remaining five samples approximately shared almost the same diversity, in the range of 1.16-1.49.
Table 4 Relative abundance of bacterial clones affiliated with each major group

3.4 Phylogenetic analysis of archaeal sequences
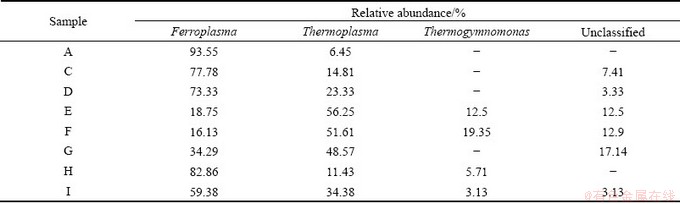
Archaeal lineages reported from AMD environments were restricted to the Thermoplasmatales and Sulfolobales [15]. In our study, the archaeal clones were tested and all belonged to Thermoplasmatales, with no appearance of Sulfolobales. Archaea detected in the ten samples fell into three known phylogenentic divisions, Thermoplasma, Ferroplasma, Thermogymnomonas and a genus unclassified. The difference between each other was the percentage of archaea communities. For samples E, F and G, Thermoplasma (E:56.25%; F:51.61%; G:48.57%) occupied an important advantage over Ferroplasma (E:18.75%; F:16.13%; G:34.29%). For the rest five samples A, C, D, H and I, Ferroplasma (A: 93.55%; C: 77.78%; D: 73.33%; H: 82.86%; I: 59.38%) was several times. Thermoplasma (A: 6.45%; C: 14.81%; D: 23.33%; H: 11.43%; I: 34.38%). Samples E, F, H and I had Thermogymnomonas with the relative abundance of 12.50%, 19.35%, 5.71% and 3.13%, respectively (Table 5).
Table 5 Relative abundance of archaeal clones affiliated with each major group
The diversity index of archaea was generally low for these eight samples, and the highest diversity index reached 1.22 only, which showed that the archaeal communities of acidic waste water were quite limited. Especially for sample A, there were only two OTUs detected, with the diversity index of 0.24, which was the lowest value in all. And the rest samples were almost limited (Table 3).
3.5 Statistical analysis
3.5.1 UPGMA cluster analysis of clone libraries
UPGMA cluster analysis based on the microbial communities’ compositions including bacteria and archaea in the eight samples was used to reveal the microbial communities’ relationship among them. This suggested that samples C, E, F, G and H had more similarity in microbial community structure among the eight samples (Fig. 1). The percentage of similarity reached between 66.98% and 88.24% for the predominance of Thermomonas and Meiothermus. Besides, samples D and I had 62.67% similarity with the rich Acidithiobacillus and Leptospirillum. There was a varied quite different from others for the abundance of microbial communities’ diversity.
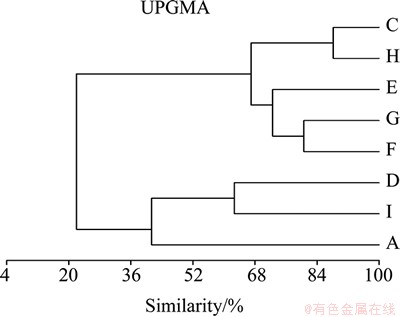
Fig. 1 UPGMA cluster analysis based on microbial communities’ compositions of water samples
3.5.2 Relations between phylogenetic microbial community and geochemical variables
To determine the key geochemical variables shaping microbial community structure, canonical correspondence analysis (CCA) was performed with the phylogenetic microbial communities of OTUs and each geochemical variable; at last, there were six significant correlation geochemical variables (Fig. 2). The result showed that geochemical variables influenced distribution of samples, especially the pH and the contents of Hg2+, Pb2+, Cl-, Fe3+ and SO42-. Fe3+ and pH made more contribution to the distribution of microorganisms. The distribution of samples D and I might have close association with the contents of Cl- and SO42-. The complexity of the microbial communities of samples C, F, G and H might be due to the effects of many geochemical variables.

Fig. 2 CCA based on microbial communities’ compositions and some significant correlation geochemical variables
4 Discussion
AMD is an extremely acidic environment with low pH and high levels of metal concentrations [16]. The investigation on this extreme environment can not only help us to understand the community structures of different acid mines relative to environment pollution, but also provide useful information about bioleaching. In this work, the bacterial community structures of these tailing ponds were identified. Moreover, multivariate analysis was performed to analyze the relationship between the geochemical variables and microbial communities.
The geochemical property of AMDs is heterogeneous, and due to the low pH of AMD, the solubility of transition metals is great, containing elevated concentrations of metals [3]. In these studied samples, the contents of S, Ca, Fe, Mg, Al, Na, Zn, Mn, Cu elements were very high, which was consistent with the previous reports [17]. AMDs have different characters in geochemical backgrounds (i.e., element composition in bedrock, and wall rock), which depend upon the location of the mining site, thus the element concentration of the AMDs varies in different locations all over the world [8]. For these eight samples, the content of S, Ca, Fe was much higher than others, especially for Ca element, which might result from the composition of bedrock. In addition, AMD may also be affected by other minerals, such as silicates, which will lead to an increase in the dissolved extents of Na, K, Mg, Ca, Si, and Al [8]. In this study, the samples were obtained from five main areas with the pH 2.0-3.5 at the temperature around 10 °C, and the samples were collected in the exposed pit outdoor.
According to the UPGMA cluster analysis, the microbial communities’ compositions could be divided into three parts, including the samples rich in iron-/ sulfur-oxidizing microorganisms, such as samples D and I; the samples rich in moderate acidophiles (Meiothermus, Thermomonas), such as samples C, E, F, G and H [18]; and sample A without dominant species but with abundant diversity.
4.1 Communities of putative iron-/sulfur-oxidizing bacterial in related samples
For these eight samples, four samples had the existence of putative iron-/sulfur-oxidizing bacteria, and the percentage of them in each sample is enumerated in Table 6. The results showed that bacteria participated in the process of iron-/sulfur-oxidizing fell into the following seven genera, with a significant advantage in samples D and I, with the proportion up to 86.11% and 84.34% respectively. Members of the genus Acidithiobacillus and Leptospirillum were ubiquitous in the following four samples, and the rest genus was not dominant.
Acidithiobacillus spp. is widely considered to be the microorganism that controls the generation rate of AMD and Acidithiobacillus ferrooxidans has been used as model microbe in bioleaching system [19]. Acidithiobacillus thiooxidans which can oxidize only sulfur and A. ferrooxidans which can oxidize sulfur and reduce inorganic sulfur compounds in addition to ferrous iron[3], were both detected in our samples. The proportion of Acidithiobacillus in each sample was 17.20% for sample A, 70.83% for sample D, 10.96% for sample E and 40.96% for sample I. The largest proportion of clones affiliated with Acidithiobacillus (70.83%) was detected in sample D, which showed that this site might be the most suitable for the growth of the organisms among these samples. Previous researches reported that Acidithiobacillus spp. was very sensitive to arsenic ions, especially arsenic ion (III) [18]. The concentration of element arsenic in sample D was not detected, and the pH was the lowest among all samples. Acidithiobacillus occupied a higher community proportion which might have been facilitated by the low arsenic concentration and low pH. For sample I, the percentage of Acidithiobacillus was the second, and the concentration of element arsenic was the highest among these five samples (1.56 mg/L). Compared with others, such as the arsenic concentration of 2.5-29 mg/L reported by XIAO et al [10], the arsenic concentration might not reach the inhibition extent. From the result of SPSS, the dominance of Acidithiobacillus had significant association with the concentrations of Fe3+, NO3-and SO42- and EC value.
Members of the genus Leptospirillum were also detected in the four samples, but the percentage was very different, with the largest occupation in sample I (42.17%), and the rest was identically lower. It should be attributed to the concentration of iron element and pH values of four samples. Previous studies with respect to Leptospirillum spp. suggest that these organisms could only use reduced iron as energy resource and occupy a greater percentage of the community structure at low pH [20]. The concentration of iron element was 73.9 mg/L for sample A, 121.2 mg/L for sample D, 17.2 mg/L for sample E, 151.8 mg/L for sample I, and the pH values were 2.6, 2.04, 2.67, 2.4, respectively. Sample D had the highest concentration of element iron and a relatively low pH, which may result in the high percentage of Leptospirillum. Previous studies based on metal ion resistance showed that Leptospirillum spp. exhibited lower tolerance to Cu2+ and Co2+ [21,22], which was the highest in sample I, but compared with other studies [9], that was scores of times low. The statistical product and service solutions (SPSS) based on the percentage of Leptospirillum and geochemical variables revealed that the concentrations of Cu2+ and Fe3+ were influence factors.
The rest five genera always occupied in sample A, which had the highest diversity among all samples; however, the percentage of iron-oxidizing or sulfur-oxidizing bacteria was low. It may be due to the low concentration of toxic metal ions.
Table 6 Percentage of putative iron-oxidizing or sulfur-oxidizing bacteria in four relative samples

4.2 Communities of other predominant bacteria in related samples
Except for the samples rich in iron-oxidizing or sulfur-oxidizing bacteria, there were still five samples occupied with two moderate acidophilic genus including Thermomonas and Meiothermus, and the proportion of them in the following samples reached 58.91%-87.50% (Table 7). Previous reports based on the bacterial communities of AMD had rarely detected these two genera [23,19,10].
Table 7 Percentage of other predominant bacteria in five relative samples

The Thermomonas organism has an optimum growth temperature of 37-50 °C and is distantly related to the species of the Xanthomonas [23]. For these five samples, this organism owed great advantage with the proportion of 31.51%-45.07%. However, a large amount of Thermomonas sp. existed at 25°C during the cultivation of aerobic granular sludge [19]. Thermomonas fusca which was aerobic and Gram-negative had been detected in the the nitrifying sludge microbial community and reported in the denitrification process, and also proved to have high nitrification rate at temperature of 10 °C [24]. KIM et al [25] discovered the existence of Thermomonas haemolytica in mesophilic sludge. T. haemolytica was said to grow in the temperature range of 18-50 °C and can produce acetate and propionate from carbohydrate fermentation [26]. For our samples, Thermomonas was not clearly classified into species, but high NH4-N and NO3-N concentrations in sample showed that this organism had close relationship with denitrification and nitrification process.
Meiothermus demonstrated another dominance of samples C, E, F, G and H with the percentage 27.40%-47.22%. This organism has the common characteristics described as non-sporeforming moderate thermophiles with optimum growth near neutral pH and capable of growth by aerobic oxidation of sugars, sugar alcohols, and some polymers [27]. It is moderately thermophilic and is capable of starch depolymerization and reduction of NO3- to NO2- and some Thermus strains are known to reduce metals [28]. For samples C, E, F, G and H, the more or less NH4-N and NO3-N may have close relationship with the Thermomonas and Meiothermus; the rest microbial community was quite few. In this study, temperature was only 4-10 °C, and pH ranged between 2.31 and 3.81, which were quite different from the former studies. It can be speculated that this environment was also suitable for Meiothermus to survive, thereby it is possible to reduce NO3- to NO2- and reduce metals.
4.3 Communities of abundant diversity in related samples
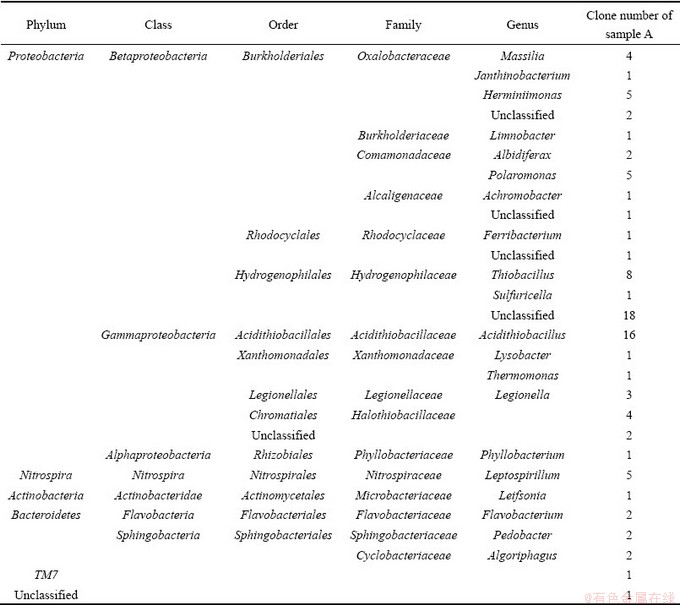
Shannon-Weaver index H was evaluated for the phylogenetic diversity of each sample. The results showed that sample A (H=2.36) had the preeminent diversity. The specific communities can be seen in Table 8.
For sample A, the acidity was almost the same with samples D and I, but the community was extremely plentiful. Except for the composition of putative iron- oxidizing or sulfur-oxidizing bacteria, there still was more other bacteria, such as Herminiimonas, Polaromonas and Halothiobacillaceae. Herminiimonas was ultimately classified into (>98%) Herminiimonas arsenicoxydans, which has been isolated from the activated sludge of industrial plant contaminated with heavy metals [29]. H. arsenicoxydans was said to be able to resist multiple toxic elements, especially arsenic by oxidizing the more toxic As (III) into the less toxic inorganic form As (V) [30]. Polaromonas sp. strain JS666, a  , is shown so far to degrade a wide variety of xenobiotic compounds, including the recalcitrant groundwater contaminant cis-1, 2-dichloroethene (cDCE). Resting cells of this strain can also transform vinyl chloride (VC) and trichloroethene (TCE) [31]. For sample A, Cl- was predominantly rich, with the concentration up to 17.86 mg/L. It can be concluded that the existence of Polaromonas had close relationship with Cl-. The appearance of Halothiobacillaceae, an obligate heterotrophs, had been called moderate acidophiles [18], and was previously considered to grow by sulfur oxidation only, which was latterly isolated from AMD as iron-oxidizing bacteria in moderately acidic (~pH 4) solid media [1].
, is shown so far to degrade a wide variety of xenobiotic compounds, including the recalcitrant groundwater contaminant cis-1, 2-dichloroethene (cDCE). Resting cells of this strain can also transform vinyl chloride (VC) and trichloroethene (TCE) [31]. For sample A, Cl- was predominantly rich, with the concentration up to 17.86 mg/L. It can be concluded that the existence of Polaromonas had close relationship with Cl-. The appearance of Halothiobacillaceae, an obligate heterotrophs, had been called moderate acidophiles [18], and was previously considered to grow by sulfur oxidation only, which was latterly isolated from AMD as iron-oxidizing bacteria in moderately acidic (~pH 4) solid media [1].
Table 8 Bacterial communities of three samples with abundant diversity
4.4 Archaeal communities’ diversity
Archaea were detected in all eight samples; and they were classified into three known genus: Ferroplasma, Thermoplasma, Thermogymnomonas and agenus unclassified, all of which belong to Thermoplasmatales order. According to previous research, all the archaea in this order are heterotrophic except Ferroplasma, which has the ability to gain energy during the oxido-reduction of iron. All the archaea in Thermoplasmatales order tend to be thermophilic, especially for Thermoplasma and Thermogymnomonas, while with the exception of Ferroplasma.
The genus of Ferroplasma is dominant in the most extreme environment of lower pH and higher ionic strength [9], including some mesophilic iron (II)-oxidizing archaea, such as F. thermophilum, Ferroplasma acidiphilium and Ferroplasma acidarmanus [9]. It was reported that F. thermophilum was often flourishing at extremely low pH (lower than 1.4), high concentration of total iron, ferrous iron and other heavy metals [1]. According to ZHANG et al [32], during the bioleaching of chalcopyrite, F. thermophilum L1 was capable of chemomixotrophic growth on ferrous iron and organic matters; moreover, it could relieve toxicity of organic matters to autotrophic bacteria. F. acidiphilumis, described as a strictly chemolithoautotrophic microbe, can oxidize Fe2+ from Fe2SO4 and pyrite FeS2. It is the only described archaeon capable of growth at the relatively low temperatures commonly encountered in AMD. Compared with F. thermophilum, the use of organic compounds as carbon sources has not been reported thus far [33]. The Ferroplasma genus detected in our samples was not classified into species, but it was important for the iron-/sulfur- oxidation.
Archaea in Thermoplasma and Thermogymnomonas were detected in extremely thermoacidophilic environment, such as burning coal refuse pile in USA and volcanic solfataric field in Italy, with the growth temperature of 33-67 °C, pH 0.5-4 [34]. The detection of them in the eight samples showed that the acidic water was not “cold” based on the archaeal communities and the growth temperature for Thermoplasma and Thermogymnomonas was not limited. The existence of heterotrophic archaea in Thermoplasma and Thermogymnomonas may create a more suitable environment for the growth of the iron-oxidizers by removing organic compounds that can be toxic to the autotroph [3]. Samples E and F were abundant in Thermoplasma and Thermogymnomonas, with the percentages of 68.75% and 70.96%, respectively. It may have close relationship with the organic carbon, for the COD of these two samples being relatively high, reaching 156.3 mg/L and 143.8 mg/L accordingly.
5 Conclusions
1) The UPGMA cluster analysis based on the microbial communities’ compositions revealed that these eight samples were divided into three parts, including the samples rich in iron-/sulfur-oxidizing microorganisms; samples rich in moderate acidophiles (Meiothermus, Thermomonas); and sample without dominant species but with abundant diversity.
2) The acidophilic microorganism existing in the mine drainage had close relationship with the surrounding environment, The dominance of Acidithiobacillus spp. had close relationship with the concentration of Fe3+, NO3-, SO42- and EC value, Leptospirillum spp. was significantly associated with the concentrations of Cu2+ and Fe3+.
3) Except for A. ferrooxidans, A. thiooxidans, and L. ferrooxidans dominanting in AMD, there still was some thermophilic microorganism, such as Meiothermus, Thermomonas and Thermoplasmatales at the low pH, while the influence factor causing the abundance of Meiothermus and Thermomonas was not clear. It might be affected synthetically by pH, ion concentration, especially by high NH4-N and NO3-N concentrations.
References
[1] SCHIPPERS A, BREUKER A, BLAZEJAK A, BOSECKER K, KOCK D, WRIGHT T L. The biogeochemistry and microbiology of sulfidic mine waste and bioleaching dumps and heaps, and novel Fe (II)-oxidizing bacteria [J]. Hydrometallurgy, 2010, 104(3-4): 342-350.
[2] JOHNSON D B. Chemical and microbiological characteristics of mineral spoils and drainage waters at abandoned coal and metal mines [J]. Water, Air Soil Pollution Focus, 2003, 3: 47-66.
[3] HALLBERG K B. New perspectives in acid mine drainage microbiology [J]. Hydrometallurgy, 2010, 104: 448-453.
[4] PESIC B, OLIVER D J, WICHLACZ P. An electrochemical method of measuring the oxidation rate of ferrous to ferric iron with oxygen in the presence of Thiobacillus ferrooxidans [J]. Biotechnology and Bioengineering, 1989, 33(4): 428-439.
[5] GUO Yao-guang, HUANG Peng, ZHANG Wu-gang, YUAN Xue-wu, FAN Feng-xia, WANG Huan-li, LIU Jian-she, WANG Zhao-hui. Leaching of heavy metals from Dexing copper mine tailings pond [J]. Transactions of Nonferrous Metals Society of China, 2013, 23(10): 3068-3075.
[6] HAO Chun-bo, WANG Li-hua, GAO Ya-nan, ZHANG Li-na, DONG Hai-liang. Microbial diversity in acid mine drainage of Xiang mountain sulfide mine, Anhui Province, China [J]. Extremophiles, 2010, 14: 465-474.
[7] YUAN Xue-wu, XIE Xue-hui, FAN Feng-xia, ZHU Weng-xiang, LIU Na, LIU Jian-she. Effects of mutation on a new strain Leptospirillum ferriphilum YXW and bioleaching of gold ore [J]. Transactions of Nonferrous Metals Society of China, 2013, 23(9): 2751-2758.
[8] PIETRO M, CRISTINA C, PAOLA C, FRANCESCO F, GABRIELLA L. Mineralogical and chemical evolution of ochreous precipitates from the Libiola Fe-Cu-sulfide mine (Eastern Liguria, Italy) [J]. Applied Geochemistry, 2012, 27(3): 577-589.
[9] HE Zhi-guo, XIAO Sheng-mu, XIE Xue-hui, HU Yue-hua. Microbial diversity in acid mineral bioleaching systems of Dongxiang copper mine and Yinshan lead-zinc mine [J]. Extremophiles, 2008, 12: 225-234.
[10] XIAO Sheng-mu, XIE Xue-hui, LIU Jian-she. Microbial communities in acid water environments of two mines, China [J]. Environmental Pollution, 2009, 157: 1045-1050.
[11] SUN Qing-ye, AN Shu-qing, YANG Lin-zhang, WANG Zhong-sheng. Chemical properties of the upper tailings beneath biotic crusts [J]. Ecological Engineering, 2004, 23(1): 47-53.
[12] XU De-cong, ZHAN Jing, CHEN Zheng, GAO Yi, XIE Xian-zheng, SUN Qing-ye, DOU Chang-ming. Effects of Vetiveria zizanioides L. growth on chemical and biological properties of copper mine tailing wastelands [J]. Acta Ecologica Sinica, 2012, 32 (18): 5683-5691.
[13] ZHAN Jing, SUN Qin-ye. Diversity of free-living nitrogen-fixing microorganisms in wastelands of copper mine tailings during the process of natural ecological restoration [J]. Journal of Environmental Sciences, 2011, 23: 476-487.
[14] HUMAYOUN S B, BANO N, HOLLIBAUGH J T. Depth distribution of microbial diversity in Mona lake, a mermictic soda lake in California [J]. Applied and Environmental Microbiology, 2003, 69: 1030-1042.
[15] QIU Guan-zhou, WAN Min-xi, QIAN Lin, HUANG Zhi-ying, LIU Kai, LIU Xue-duan, SHI Wu-yang, YANG Yu. Archaeal diversity in acid mine drainage from Dabaoshan mine, China [J]. Journal of Basic Microbiology, 2008, 48: 401-409.
[16] ZHOU Zhi-jun, YIN Hua-qun, LIU Yi, XIE Ming, QIU Guan-zhou, LIU Xue-duan. Diversity of microbial community at acid mine drainages from Dachang metals-rich mine, China [J]. Transactions of Nonferrous Metals Society of China, 2010, 20(6): 1097-1103.
[17] XIE Jian-ping, JIANG Hong-chen, LIU Xin-xing, LIU Xue-rui, ZHOU Ji-zhong, QIU Guan-zhou. 16s rDNA based microbial diversity analysis of eleven acid mine drainages obtained from three Chinese copper mines [J]. Journal of Central South University of Technology, 2011, 18: 1930-1939.
[18] MURAVYOV M I, BULAEV A G. Two-step oxidation of a refractory gold-bearing sulfidic concentrate and the effect of organic nutrients on its biooxidation [J]. Minerals Engineering, 2013, 45: 108-114.
[19] SONG Zhi-wei, REN Nan-qi, ZHANG Kun, TONG Long-yan. Influence of temperature on the characteristics of aerobic granulation in sequencing batch airlift reactors [J]. Journal of Environmental Sciences, 2009, 21: 273-278.
[20] CHENG Yi, GUO Zhao-hui, LIU Xue-duan, YIN Hua-qun, QIU Guan-zhou, LIU Hong-wei. The bioleaching feasibility for Pb/Zn smelting slag and community characteristics of indigenous moderate- thermophilic bacteria [J]. Bioresource Technology, 2009, 100: 2737-2740.
[21] HE Zhi-guo, YIN Zhen, WANG Xin, ZHONG Hui, SUN Wei. Microbial community changes during the process of pyrite bioleaching [J]. Hydrometallurgy, 2012, 125-126: 81-89.
[22] LIN Hai, ZHU Yi-jun, DONG Ying-bo, CHENG Huang, HUO Han-xin. Breeding and adsorption properties investigation of a strain resistant to acid and copper ions [J]. Environmental Chemistry, 2013, 32(4): 599-604.
[23] MARTA P A, FRED A R, FERNANDA M N, MILTON S C. Thermomonas hydrothermalis sp. nov.: A new slightly thermophilic γ-Proteobacterium isolated from a hot spring in central portugal [J]. Systematic and Applied Microbiology, 2003, 26: 70-75.
[24] THOMAS F D, MATIAS B V, ANTHONY D S, ARIEL A S, APREL Q E. Characterization of a microbial community capable of nitrification at cold temperature [J]. Bioresource Technology, 2010, 101: 491-500.
[25] KIM W, HWANG K, SHIN S G, LEE S, HWANG S. Effect of high temperature on bacterial community dynamics in anaerobic acidogenesis using mesophilic sludge inoculum [J]. Bioresource Technology, 2010, 101: S17-S22.
[26] WOONG K, KWANGHYUN H, SEUNG G S, SEUNGYON G, SEOKHWAN H. Effect of high temperature on bacterial community dynamics in anaerobic acidogenesis using mesophilic sludge inoculum [J]. Bioresource Technology, 2010, 101(1): S17-S22.
[27] HEDLUND B P, MCDONALD A I, LAM J, DODSWORTH J A, BROWN J R, HUNGATE B A. Potential role of Thermus thermophilus and T. oshimai in high rates of nitrous oxide (N2O) production in 80 °C hot springs in the U.S. Great Basin [J]. Geobiology, 2011, 9: 471-480.
[28] ERROL D C, LIZELLE A P, ESTAVAN H. Reduction of U(VI) by the deep subsurface bacterium, Thermus scotoductus SA-01, and the involvement of the ABC transporter protein [J]. Chemosphere, 2012, 86(6): 572-577.
[29] MULLER D,SIMEONOVA D D,RIEGEL P,MANGENOT S,KOECHLER S,LIEVREMONT D,BERTIN P N,LETT M C. Herminiimonas arsenicoxydans sp. nov., a metalloresistant bacterium [J]. International Journal of Systematic and Evolutionary Microbiology, 2006, 56: 1765-1769.
[30] WEISS S, CARAPITO C, CLEISS J, KOECHLER S, TURLIN E, COPPEE J Y, HEYMANN M, KUGLER V, STAUFFERT M, CRUVEILLER S, MEDIGUE C, VAN-DORSSELAER A, BERTIN P N, ARSENE-PLOETZE F. Enhanced structural and functional genome elucidation of the arsenite-oxidizing strain Herminiimonas arsenicoxydans by proteomics data [J]. Biochimie, 2009, 91: 192-203.
[31] ADEMOLA O O, ADHIKA B, BALAKRISHNA P. Quantitative assessment of the toxic effects of heavy metals on 1, 2-dichloroethane biodegradation in co-contaminated soil under aerobic condition [J]. Chemosphere, 2011, 85(5): 839-847.
[32] ZHANG Li-juan, JI Hou-guo, WANG Yu-guang, WAN Li-li, ZHOU Hong-bo. Synergies between archaea Ferroplasma thermophilum and two moderately thermophilic bacteria under stress of yeast extract or Cu2+ [J]. The Chinese Journal of Nonferrous Metals, 2013, 23(1): 265-273. (in Chinese)
[33] MOHSEN F, TSUYOSHI H. Hydrophilicity of Ferroplasma acidiphilum and its effect on the depression of pyrite [J]. Minerals Engineering, 2012, 36-38: 242-247.
[34] PLUMB J J, HADDAD C M, GIBSON J A, FRANZMANN P D. Acidianus sulfidivorans sp. nov., an extremely acidophilic, thermophilic archaeon isolated from a solfatara on Lihir Island, Papua New Guinea, and emendation of the genus description [J]. International Journal of Systematic and Evolutionary Microbiology, 2007, 57: 1418-1423.
杨 扬,李 杨,孙庆业
安徽大学 资源与环境工程学院,合肥 230601
摘 要:为了加深对安徽省铜陵市不同重金属富集程度的尾矿附近酸性废水中微生物多样性的了解,运用PCR-16S rDNA克隆文库技术对酸性废水中细菌及古菌的群落结构进行研究。在铜陵的5个样点采集了8个水样。系统发育分析结果表明,酸性废水中的细菌主要是Betaproteobacteria, Gammaproteobacteria, Alphaproteobacteria, Deinococcus-Thermus, Nitrospira, Firmicutes, Actinobacteria, Deltaproteobacteria, Bacteroidetes, Chloroflexi,古菌是Thermoplasma, Ferroplasma 以及Thermogymnomonas。根据细菌和古菌的组成对样品进行聚类分析,结果表明,5个样品由于优势菌株Thermomonas和Meiothermus拥有较高的相似度。2个样品由于Acidithiobacillus 和 Leptospirillum占主导也拥有较高的相似度。剩余的样品多样性较高,拥有最高的香浓-微纳指数2.91。典型相关分析CCA的结果表明微生物的多样性组成与理化因子有密切的关系,如水的pH以及Hg2+、Pb2+、Cl-、SO42-、Fe3+的含量。
关键词:酸性矿业废水;微生物结构组成;克隆文库;理化因子
(Edited by Xinag-qun LI)
Foundation item: Project (41171418) supported by the National Natural Science Foundation of China
Corresponding author: Qing-ye SUN; Tel: +86-13866755085; Fax: +86-551-63861882; E-mail: sunqingye@ahu.edu.cn
DOI: 10.1016/S1003-6326(14)63474-9

