Leak-off behavior and water shut-off performance of a polymer/chromium (Cr3+) gel in fractured media
��Դ�ڿ������ϴ�ѧѧ��(Ӣ�İ�)2017���6��
�������ߣ���Ӣ� ��� �ܴ��� ������ κ���� ����
����ҳ�룺1418 - 1429
Key words��polymer; gelant; gel; leak-off; water shut-off; fracture
Abstract: During gel treatments for fractures, the leak-off behavior of gelant has a great effect on the water shut-off performance of gel. Experiments were carried out using a polymer/chromium (Cr3+) gel system to explore the leak-off behavior and the water shut-off performance of gel in fractured media. Results of the gelant leak-off study show that the gelant leak-off from fracture into matrix contributes to the formation of the gelant leak-off layer during the gelant injection. Moreover, because of the gradual formation of the gelant leak-off layer along fracture, the initial leak-off ratio of gelant is relatively high, but it declines and finally levels off with the increase of the injection volume. The polymer concentration of gelant has a great effect on the chromium output in fluids produced from fractures. With the increase of the polymer concentration, the chromium concentration first decreases and then increases, and the leak-off depth of gelant into matrix is gradually reduced. Results of the water shut-off study present that the decrease of the chromium concentration inside the fracture greatly reduces the water shut-off performance after the gel formation. Therefore, because of the relatively high degree of chromium leak-off, enough injection volume of gelant is essential to ensure the sufficient chromium concentration inside the fracture and to further achieve a favorable water shut-off performance. On the premise of gel strength assurance inside the fracture, the water shut-off performance of gel gradually declines with the extension of the distance from the fracture inlet, and different leak-off degrees of gelant along the fracture are responsible for this phenomenon. Therefore, a proper degree of gelant leak-off contributes to enhancing the water shut-off performance of gel for fractures.
Cite this article as: LI Jun-jian, XIONG Chun-ming, BAI Ying-rui, JIANG Ru-yi, WEI Fa-lin, ZHANG Miao. Leak-off behavior and water shut-off performance of a polymer/chromium (Cr3+) gel in fractured media [J]. Journal of Central South University, 2017, 24(6): 1418-1429. DOI: 10.1007/s11771-017-3546-1.
J. Cent. South Univ. (2017) 24: 1418-1429
DOI: 10.1007/s11771-017-3546-1
LI Jun-jian(���)1, XIONG Chun-ming(�ܴ���)2, BAI Ying-rui(��Ӣ�)2,
JIANG Ru-yi(������)3, WEI Fa-lin(���)2, ZHANG Miao(����)1
1. College of Petroleum Engineering, China University of Petroleum (Beijing), Beijing 102249, China;
2. Research Institute of Petroleum Exploration & Development, PetroChina, Beijing 100083, China;
3. Department of Science and Technology Management, China National Petroleum Corporation, Beijing 100007, China
 Central South University Press and Springer-Verlag Berlin Heidelberg 2017
Central South University Press and Springer-Verlag Berlin Heidelberg 2017
Abstract: During gel treatments for fractures, the leak-off behavior of gelant has a great effect on the water shut-off performance of gel. Experiments were carried out using a polymer/chromium (Cr3+) gel system to explore the leak-off behavior and the water shut-off performance of gel in fractured media. Results of the gelant leak-off study show that the gelant leak-off from fracture into matrix contributes to the formation of the gelant leak-off layer during the gelant injection. Moreover, because of the gradual formation of the gelant leak-off layer along fracture, the initial leak-off ratio of gelant is relatively high, but it declines and finally levels off with the increase of the injection volume. The polymer concentration of gelant has a great effect on the chromium output in fluids produced from fractures. With the increase of the polymer concentration, the chromium concentration first decreases and then increases, and the leak-off depth of gelant into matrix is gradually reduced. Results of the water shut-off study present that the decrease of the chromium concentration inside the fracture greatly reduces the water shut-off performance after the gel formation. Therefore, because of the relatively high degree of chromium leak-off, enough injection volume of gelant is essential to ensure the sufficient chromium concentration inside the fracture and to further achieve a favorable water shut-off performance. On the premise of gel strength assurance inside the fracture, the water shut-off performance of gel gradually declines with the extension of the distance from the fracture inlet, and different leak-off degrees of gelant along the fracture are responsible for this phenomenon. Therefore, a proper degree of gelant leak-off contributes to enhancing the water shut-off performance of gel for fractures.
Key words: polymer; gelant; gel; leak-off; water shut-off; fracture
1 Introduction
Fractured reservoirs which contain hydraulic fractured reservoirs and natural fractured reservoirs occupy an important place in oilfields. Sometimes the initial oil production of fractured reservoirs is very high because of the high communication between fractures and the wellbore [1]. However, during the oil production, the injected water and formation brine easily break through into oil wells along fractures and make the watered out well become common [2, 3]. The excessive water production not only burdens the subsequent water disposal but also shortens the development life of an oilfield [4, 5]. Therefore, much work should be done to control the useless water production in fractured reservoirs.
Water shut-off agents are commonly used in oilfields for water control in fractured reservoirs, especially in hydraulic fractured reservoirs. Polymer gel system is the principal water shut-off agent; it has drawn lots of attentions and shown favorable water shut-off performance in porous reservoirs [6]. Besides the polymer gel system, many kinds of novel water shut-off agents have been developed. GOUDARZI et al [7] and BAI et al [8] synthesized a kind of preformed particle gel (PPG), respectively, and proved its favorable water shut- off performance in fractured and porous media. Al-GHAZA et al [9] developed a chemical packer which was made from fiber and epoxy resin and efficiently controlled water production in gas wells. New kinds of water shut-off agents contributed to the improvement of the water shut-off efficiency in recent years, but the polymer gel is still a kind of promising agent for water shut-off in fractured reservoirs if the interaction of the gel and reservoir, or the effect of reservoir parameters on the water shut-off performance is clearly understood [10].
Nowadays, during the application of polymer gels in fractured reservoirs, field experiences of polymer gels in porous reservoirs are referenced for the gel design, but the effectiveness is usually unsatisfied because difference exists between porous and fractured media. It is well known that the construction of fractures, especially the hydraulic fractures is very complex [10]. It often has a high fracture height, a variational fracture aperture and a complex fracture length; therefore, the flow behavior of fluids concluding polymer gelant in fracture is different with that in porous media. ZHAO et al [11] explored the influence of fracture aperture on the water shut-off performance of polymer (HPAM) gels using fractured cores, data presented that the critical injection pressure was inversely related to the fracture aperture. SERIGHT et al [12-14] systematically studied the effects of fracture length, fracture aperture and fracture height on the water shut-off performance of chromium (Cr3+)/ polymer (PHPA) gels, a series of equations for the gel design was concluded which is very useful for the gel treatment design for fractures.
In reality, during the injection of the gelant, despite the huge difference between the permeability of the fracture and that of its adjoining matrix, the gelant will leak-off into the matrix and results in the gelant loss. Polymer and chromium are two essential chemical agents of the gelant, but due to different molecular masses between two agents, polymer and chromium have different leak-off and diffusion behaviors during the gelant injection [15]. The initially injected gelant is easily diluted by the formation brine and polymer molecules can adsorb on the fracture face or diffuse into the matrix [16]; thus the gelant viscosity is correspondingly reduced. During the gel treatment for fracture, the reduction of the gelant viscosity reflects the loss of the polymer, and it can greatly affect the gel formation. Therefore, it is important to keep the gelant viscosity stationary during the gelant injection. The common method for the viscosity holding is to improve the polymer concentration or inject the low concentration polymer preflush [17]. However, the viscous advance of the gelant in fracture still remains unclear. Different from polymer molecules which have high molecular mass and long molecular chain, the molecular mass of chromium ions is very small, and thus chromium ions are very easy to lose itself during the gelant propagation and the shut-in period in fracture. Moreover, the chromium concentration is also diluted by the formation brine, and its decrease in concentration can extend the gelation time, reduce the gel strength, and finally make the gel treatment for fracture ineffective [18].
In the present work, experiments were conducted using a polymer/chromium (Cr3+) gel system and prepared fractured cores. The main objective of this study is to determine the leak-off behavior and water shut-off performance of the gel in fractured media. Other sub-objectives are: 1) to explore the effect of polymer on the gelant leak-off and chromium output; 2) to analyze the effect of gelant leak-off on the water shut-off performance after the gel formation; 3) to determine the water shut-off performance of gel along the fracture.
2 Materials and methods
2.1 Materials
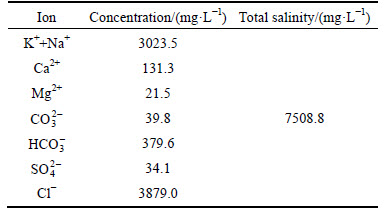
The commercial water-soluble partially hydrolyzed polyacrylamide (HPAM, Hengju Chemical Group Corp., China) was used as the polymer with an average molecular mass of 12000 kDa, a degree of hydrolysis of about 25% and a purity of over 98% (mass fraction). Chromium (Cr3+) acetate (Shandong Shida Oilfield Technical Services Co., Ltd, China) was used as the cross-linker with the effective concentration of 38.4 g/L. The formation brine is made in laboratory, and its compositional analysis result is shown in Table 1. Sodium chloride, calcium chloride, calcium chloride, magnesium chloride, sodium bicarbonate and sodium sulfate (Sinopharm Chemical Reagent Co., Ltd., China) are analytical reagents and used to prepare the formation brine.
Table 1 Compositional analysis of formation brine
The gelant was prepared using fixed concentrations of polymer and chromium (Cr3+). The polymer concentration in gelant is in the range of 0 to 5000 mg/L and the chromium (Cr3+) concentration is kept constant at 357.5 mg/L. The gelant is aged for 2 h before experiments.
2.2 Fractured core preparation
The cubic fractured cores and cylindrical fractured cores were used, as shown in Figs. 1(a) and (b), respectively. The cubic fractured cores were used to test the leak-off behavior and profile propagation during the gelant injection along fractures, and the cylindrical fractured cores were used to visually observe the degree of gelant leak-off into matrix and the gelant film formation on the fracture face. The size of each cubic fractured core is 40 cm in length, 4.5 cm in height and 4.5 cm in aperture; the fracture size of each cubic fractured core is 40 cm in length, 3.5 cm in height and 0.2 cm in aperture, and the water permeability of each core matrix is approximate to 50 mD. The size of cylindrical fractured cores is d 2.5 cm��10 cm, and the fracture size across the core is 10 cm in length, 1.5 cm in height and 0.2 cm in aperture.

Fig. 1 Pictures of fractured cores:
The preparation steps of cubic fractured cores are briefly described as follows: 1) core homogeneous cubic cores with a length of 40 cm, a height of 4.5 cm and an aperture of 4.5 cm from candidate outcrop rocks, the faces of each core were processed to keep them parallel; 2) place cubic cores in an oven at 60 ��C for 24 h to make them dried; 3) put cores into a cubic core holder to vacuumize them using a vacuum pump and then inject the formation brine at an injection rate of 0.5 mL/min to measure its water permeability; 4) completely dry cores again; 5) dissect each homogeneous cubic core into halves along with its longitudinal direction; 6) place a rectangular rigid plastic slice with a thickness of 0.2 cm, a height of 3.0 mm and a length of 40 cm into halves of cores, and clamp cores to prevent them moving; 7) cement halves of cores using an adhesive at two edges which were unclosed due to the small aperture of the plastic slice compared with the diameter of the core; 8) remove the plastic slice from cores to create a fracture;9) equidistantly fix ports 1, 2 and 3 along the fracture and fixing ports 4, 5 and 6 along the matrix (as shown in Fig. 1), and then coat each core using the epoxy resin to build a resin cemented layer around it. Then cubic cores with a synthetic fracture were prepared. Similarly, during the preparation of cylindrical fractured cores, homogeneous cylindrical cores with a length of 10 cm and a diameter of 2.5 cm were cored from outcrop rocks in step 1), and they were prepared base on steps 1)-8) but without step 9) because cylindrical fractured cores did not need to be fixed ports and coated.
2.3 Chromium measurement
Chromium concentration in gelant and produced fluids at the fracture outlet and fracture/matrix ports was determined using the GBC CINTRA3030 ultraviolet- visible spectrophotometer with a wavelength of 358 nm. Before experiments, the standard solution was prepared to calibrate the spectrophotometer.
A series of solution samples with different concentrations of chromium were prepared to determine a series of absorbance values using the spectrophotometer, and then the typical curve of chromium concentration vs absorbance value was achieved. To make the typical curve match well with the straight line, the concentration range of chromium in solution samples was 0-100 mg/L.
During the core flow experiments, four fracture volumes of the gelant were injected in cubic fractured cores. At regular intervals, the fluid produced from the outlet was collected, and then it was mixed with the 5% hypochlorite in a mass ratio of 1: 2 for 12 h. The application of hypochlorite was to destroy the polymer in the produced fluids, free chromium ions bonded to polymer molecules reduced the fluid viscosity. After that, the chromium concentration in fluids was diluted using deionized water to the order of 5-50 mg/L to take advantage of the linear range in the typical curve. Absorbance values of the processed fluids were measured, and then chromium concentrations of produced fluids were obtained according to the typical curve and the dilution multiple. The normalized chromium concentration was calculated and it equaled the ratio of the chromium concentration of the produced fluid to the initial chromium concentration of the fixed gelant (357.5 mg/L).
2.4 Gelant leak-off analysis
The leak-off behavior of the polymer/chromium gelant was studied using both cubic and cylindrical fractured cores. When the gelant was injected into cubic fractured cores, because of the pressure differential between fracture and matrix along with the concentration differential between the gelant in fracture and the brine in matrix, the gelant leaked-off into matrix and flowed out at matrix ports (ports 4, 5 and 6). It could also flow out at fracture ports (ports 1, 2 and 3) if fracture ports were vented to the atmosphere. The leak-off volume of the produced fluid at each port was collected and the leak-off ratio was defined as the ratio of the cumulative volume of the fluid produced from the matrix ports to the totally injected fracture volume of the gelant. When the gelant was injected into the cylindrical fractured cores, the gelant was dyed using the methyleneblue. After the gelant injection, cores were taken out of the core holder and dissected into pieces along its length and diameter directions. Because of the gelant leak-off from fracture into matrix, the methyleneblue also seeped into matrix and dyed sands near the fracture. After that, microscopic images were obtained using an Anyty microscope (3R-MSUSB401/601), to visually observe the leak-off degree of the gelant into matrix and the gelant film on the fracture face. Meanwhile, the average leak-off depth of the gelant in each core was measured using the Anyty processing software. The experimental temperature was 55 ��C which is the same as the real reservoir temperature of Chang 6# oil layer in Changqing oilfield.
2.5 Water shut-off performance test
The water shut-off performance of gel was tested using the subsequent brine injection, and the brief procedure is as follows: 1) prepared gelant was injected into cylindrical and cubic fractured cores; 2) cores were sealed and placed in an oven at 60 ��C. To ensure the gel formation, the placement time of each core was the final gelation time (30 h) of the gelant; 3) the subsequent brine injection was carried out along the opposite direction of the gelant injection; meanwhile, the real-time injection pressure and the injection volume were automatically collected until the injection pressure levels off. It needs to explain that the injection rate of gelant was 0.5 mL/min when it was injected into cubic fractured cores, but it was 0.2 mL/min when the gelant was injected into cylindrical fractured cores. The experimental temperature was 55 ��C. Moreover, the matrix ports (ports 4, 5 and 6) were connected with atmosphere during the gelant injection but they were sealed during the process of the subsequent brine injection.
When the water shut-off performance of gel in cubic fractured cores was evaluated after the gel formation, the fracture ports (ports 1, 2, and 3) were successively opened, i.e., port 3 was first opened at the beginning of the brine injection; when the brine was continually produced from port 3 and the injection pressure was stable, port 2 was opened and port 3 was sealed; next, port 1 was opened and port 2 was sealed when the brine was continually produced from port 2 and the injection pressure was stable; finally, the fracture inlet (gelant inlet) was opened and port 1 was sealed. The fracture can be divided into four sections by three ports, and the purpose of the opening and sealing procedure is to determine the water shut-off performance of the gel in each section along the fracture.
3 Results and discussion
3.1 Leak-off behavior of gelant in fractured media
3.1.1 Leak-off mechanism of gelant in fractured media
There are three flow ways when the gelant is injected into fractured media [19]. The principal flow through fracture (fracture flow), the leak-off flow from fracture into adjacent matrix (leak-off flow) and the parallel flow in the matrix next to the fracture (matrix flow), as shown in Fig. 2. It means that a certain volume of gelant will leak off from fracture into matrix, and the concentration and pressure differences between gelant in fracture and brine in adjacent matrix are mainly responsible for this phenomenon [20]. The flow rate of the gelant which outputs from the matrix outlets can be considered to be the flow rate of the leak off flow. The fracture flow constitutes the main gelant flow in the fractured media, and its flow rate approximately equals that of the gelant produced out of the fracture outlet. Compared with the flow rate of the fracture flow or the leak off flow, the flow rate of the matrix flow is relatively weak because of the high parallel flow of the gelant in matrix, and it is uneasy to be measured. The flow ways of the gelant are closely related to the gel distribution after the gel formation.
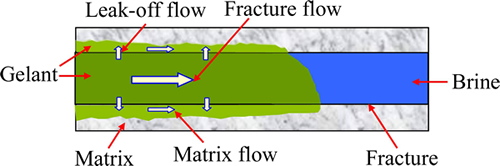
Fig. 2 Three flow ways during gelant injection into fractured core
During the gelant leak-off from fracture into matrix, a gelant leak-off layer, which consists of polymer molecules, chromium ions and water, will be formed onto the fracture face and into the adjacent matrix. The gelant leak-off layer in fractures consists of three sub-layers: the adsorption layer onto fracture surface, the aggregation layer onto adsorption layer, and the retention layer into adjacent matrix, as shown in Fig. 3. During the leak-off process of gelant, a small number of polymer molecules can entry and be entrapped into matrix pores to form a retention layer into the adjacent matrix (Fig. 3(c)), especially when the pressure differential between fracture and matrix is high [21]. Electrostatic attraction and hydrogen bonding attraction between fracture surface and charged anionic polymer molecules play dominating roles in the polymer adsorption, and it occurs between the hydroxyl groups onto sand surfaces and carboxylate groups of polymer molecules [22, 23]. The adsorption type of polymer molecules onto fracture face follows the adsorption rule of Langmuir, i.e., usually only single layer of polymer molecules can adsorb onto fracture face, and the thickness of the polymer adsorption layer is in nanometer scale [24]. After the gelant adsorption reaches the equilibrium state, polymer molecules tend to aggregate onto the adsorption layer because of the continual leak-off of gelant, and then a gelant aggregation layer will be formed onto the adsorption layer, as shown in Fig. 3(b). The gelant leak-off layer plays a role of barrier onto the fracture face and into the adjacent matrix to retard the diffusion rate of chromium ions from fracture into matrix and to slow down the leak-off rate of the subsequent gelant.
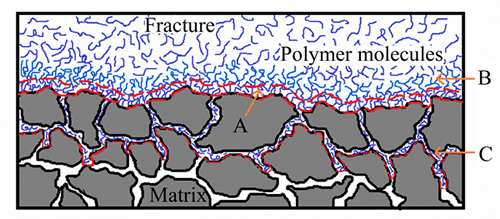
Fig. 3 Schematic of gelant leak-off layer onto fracture face and into adjacent matrix (A-Adsorption layer; B-Aggregation layer; C-Retention layer)
3.1.2 Effect of polymer on gelant leak-off
To explore the effect of polymer on the leak-off behavior of gelant, four kinds of gelants with the same chromium concentration of 357.5 mg/L and polymer concentrations of 0, 1000, 3000 and 5000 mg/L, respectively were injected into four cubic fractured cores with the same size. The flow rate of the gelant was 0.5 mL/min. The fluids produced from fracture outlets were collected in each 5 mL for the measurement of the Cr3+ concentration, and then the normalized chromium concentrations were calculated and plotted in Fig. 4. Four curves show a similar change trend which can be divided into four sub-trends: no chromium emerges in the produced fluid at the initial injection stage; with the injected fracture volume of the gelant increasing, chromium is presented but its increasing rate is low; with a further gelant injection, the chromium concentration of the produced fluid sharply increases; after certain fracture volumes of the gelant injection, curve levels off and the chromium concentration of the produced fluid is equal to that of the injected gelant. The change trend of the normalized chromium concentration curve indicates that the gelant injected at the initial stage is diluted and produced together with the formation brine, as the formation brine is gradually flooded out, the diluting effect is waning and the chromium concentration in the produced fluid is improved. The static diffusion behavior of gelant into brine is shown in Fig. 5. The gelant (3000 mg/L HPAM + 357.5 mg/L Cr3+) was first added into three transparent glass bottles, and then the brine with the same volume with gelant was added. A transitional zone which can be defined as the diffusion zone was observed because of the diffusion of gelant compositions into brine. When the static time is 0.5 min, the diffusion zone is very small and extends with the increase of the static time. It stands for the gradual diffusion of gelant into brine. The result shows that the diffusion degree of gelant into brine is significant, and it may be responsible for the advance production of some chromium and polymer molecules from the fracture outlet.
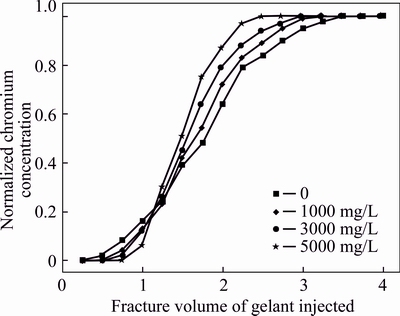
Fig. 4 Normalized chromium concentrations of produced fluids during gelant injection with different polymer concentrations of gelants

Fig. 5 Diffusion behavior of gelant in brine at different static time:
Figure 4 also illustrates that there is difference among four curves when polymer concentrations in gelants are different. When the injected volume is lower than 1.25 fracture volume (FV), the chromium concentration of the produced fluid decreases with the increase of the polymer concentration. However, after the injected gelant volume exceeds 1.25 FV, the chromium concentration in the produced fluid increases with the enhancement of the polymer concentration. The diluting effect of the formation brine, the diffusion of the chromium and the retarding effect of the polymer are responsible for the above phenomenon [18, 25]. As mentioned above, during the gelant injection, the gelant will be diluted by the formation brine at its injection front. The higher the polymer concentration is, the weaker the diluting effect shows. In addition, because of the high molecular mass of the polymer molecule, polymer can viscosify the fluid and retard the diffusion rate of the chromium. Therefore, with the increase of the polymer concentration in gelant, less chromium will diffuse into the formation brine and matrix, and thus the chromium produced at the initial injection stage is relatively low. After certain fracture volumes of the gelant is injected, the diluting effect of the formation brine becomes slight because most of the brine has been flooded out by the gelant; meanwhile, less chromium can diffuse into matrix and most of them will be produced with the viscous gelant fluid due to the retarding effect of the gelant leak-off layer [26].
Microscopic images about the gelant leak-off from fracture into adjacent matrix are obtained when polymer concentrations are 0, 1000, 3000 and 5000 mg/L, respectively, as shown in Figs. 6(a)-(d). When the gelant consisted of only chromium is injected, the matrix within the image is completely dyed by the methyleneblue; with the increase of the polymer concentration in the gelant, the dyed area of matrix obviously declines and it indicates a weak leak-off of the gelant. The average leak-off depth in each image is also measured. When only chromium solution is injected, the average leak-off depth of gelant is larger than 7.12 mm, and it sharply decreases to 4.65 mm when the polymer concentration is 1000 mg/L; with the increase of the polymer concentration to 3000 and 5000 mg/L, the average leak-off depths are correspondingly reduced to 2.92 and 1.68 mm, respectively. The main reason for this phenomenon is the retarding effect of the polymer on the gelant leak-off. As discussed above, during gelant injection, there are inaccessible pores in matrix for polymer molecules because of the high molecular mass and long molecular chain, and it results in the disproportionate leak-off between polymer and chromium (Cr3+) [20]. Namely, the main leak-off fraction is the chromium ions along with water, most of the lost polymer molecules will adsorb and aggregate on the fracture face or penetrate only a small depth into the matrix to form a gelant leak-off layer which can prevent the gelant from further leaking off into matrix [26, 27].
Besides the polymer concentration, the injection pressure may be another reason for the formation of the gelant leak-off layer. Figure 7 illustrates the change of pressure differentials when gelants with different polymer concentrations are injected. It shows that the pressure differential increases with the improvement of the polymer concentration. Microscopic images of gelant layers are shown in Figs. 8(a) and (b) when the injection pressure differentials are 20 and 100 kPa, respectively. Figure 8 shows that the gelant layer becomes thicker with the increasing pressure differential, i.e., a high pressure differential contributes to the formation of the gelant leak-off layer.

Fig. 6 Microscopic images of gelant leak-off from fracture into matrix with different polymer concentrations:
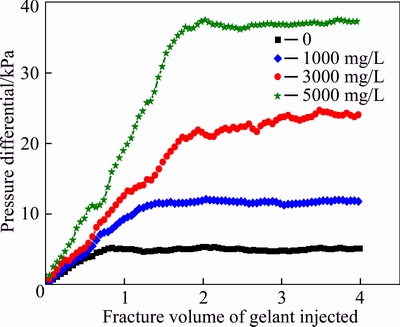
Fig. 7 Pressure differentials during gelant injection with different polymer concentrations of gelants

Fig. 8 Microscopic images of polymer film with different injection pressure differentials:
According to the analysis of the polymer effect on the gelant leak-off, to reduce the leak-off degree of the gelant, the polymer concentration of the gelant should be enhanced; however, the cost increases correspondingly. Therefore, an appropriate concentration of polymer in gelant is required. According to the field application of the gelant and the results of the present study, a fixed gelant consisting of 3000 mg/L polymer and 357.5 mg/L chromium (Cr3+) is used in the following study.
3.1.3 Leak-off behavior of a fixed gelant used for water shut-off
In this section, the leak-off behavior of the fixed gelant was tested, and the curves of cumulative flow vs injected gelant volume at fracture and matrix outlets are shown in Fig. 9. The fraction of the injected gelant that leaks off into the matrix and flows from matrix ports (ports 4, 5 and 6) is also plotted as a function of the injected gelant volume. The volume of the produced fluid collected at the fracture outlet continuously increases with the injection lasting, and the volume of the produced fluid at three matrix outlets increases at first and then levels off. Figure 9 shows that after 4.0 FV (96 mL) gelant is injected into the fractured core, the final volume of the fluid that flows from port 4 is 4.7 mL, and they are 3.4 and 1.9 mL at matrix ports 5 and 6, respectively. Data reflects that with the increase of the distance from the fracture inlet, the leak-off degree of gelant into matrix gradually declines. At the initial stage of the gelant injection, no gelant leak-off layer exists on the fracture face, and the gelant can easily leak off into the adjacent matrix; therefore, the leak-off degree is high. With lasting gelant injection, the gelant leak-off layer is formed to slow down the further loss of the water and chromium [23]. It also has been discussed that the leak-off degree decreases with the increase of the distance from the fracture inlet; therefore, the produced fluid volumes at ports 4, 5 and 6 show a decreasing trend. In addition, the change trend of the cumulative flow curves is consistent with that of pressure differential curves in Fig. 7, and both two groups of curves level off after 2.0 FV gelant are injected, and it proves that the change of the gelant leak-off degree is always accompanied with the variation of the injection pressure.
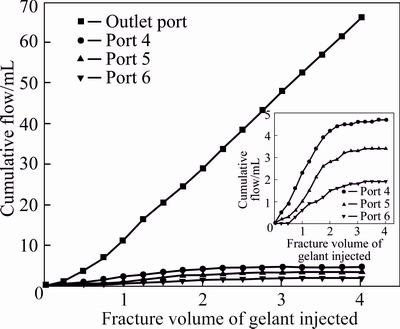
Fig. 9 Cumulative flows from fracture and matrix outlets
The leak-off ratio defined as the ratio of the cumulative leak-off volume of fluid into matrix to the injected volume of gelant is applied to evaluate the gelant leak-off, and it is calculated using the cumulative volume of the produced fluid collected at three matrix ports and the total injected volume of the gelant [28]. The changes of leak-off ratio with the gelant injection is plotted in the Fig. 10, and the fitting curve is also achieved as
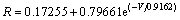 (1)
(1)
where R is the leak-off ratio and V is the fracture volume of gelant injected in FV.
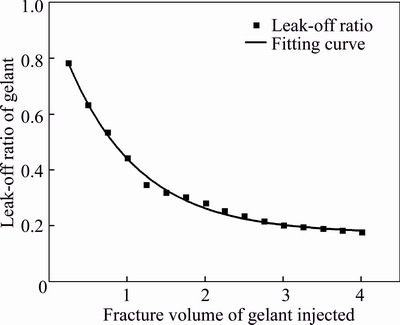
Fig. 10 Leak-off ratio of gelant from fracture into matrix during gelant injection
Two sub-curves are divided on the leak-off ratio curve using 2.0 FV as the cutting point. The initial leak-off ratio of the injected gelant is as high as 0.78, and it sharply decreases with the injected gelant volume increasing to 2.0 FV. After that, the leak-off ratio curve levels off even if the gelant is further injected, and the finally steady leak-off ratio is about 0.17. It reflects that a small volume of gelant will continually leak off from fracture into matrix after the formation of the gelant leak-off layer.
3.2 Water shut-off performance of gel in fractured media
3.2.1 Effect of gelant leak-off on gel formation
Figure 11 illustrates the effect of chromium concentration on the apparent viscosity of gel when the polymer concentration is 3000 mg/L and the temperature is 60 ��C. It shows that when the chromium concentration is lower than 150 mg/L, the apparent viscosity of formed gel is relatively low; the apparent viscosity of gel sharply increases with the chromium concentration further increasing. The gelant which is injected into fractured cores in our experiments consists of 3000 mg/L polymer and 375.5 mg/L chromium (Cr3+), and the apparent viscosity of the fixed gel formed in bottle is 10250 mPa��s. The fixed gelant is injected into three same cylindrical fractured cores with the same injection rate of 0.2 mL/min and the same injection volume of 1.0 FV, respectively. The back pressures at fracture outlets of three cores are 0, 50 and 100 kPa, respectively. It shows that the increase of the back pressure contributes to the gelant leak-off because of the increasing pressure differential between fracture and matrix. When the back pressure is 0 kPa, almost all the gelant flows through fracture and little gelant leaks off into matrix, but the gelant leak-off is significant when the back pressure is 100 kPa. Three degrees of gelant leak-off from fracture into matrix are achieved, and they are mild degree of leak-off, moderate degree of leak-off, and severe degree of leak-off, respectively. To further explore the effect of leak-off degree of gelant on the gel formation in fractures, gels are taken out of fractures, and the chromium concentrations along with apparent viscosities of three gels are measured and plotted in Fig. 12. Data show that the chromium concentrations of three gels are 328.7, 280.9 and 195.8 mg/L, respectively, and the apparent viscosities are 9785, 7969 and 2960 mPa��s, respectively. This is consistent with the results of bottle tests, and the decline of chromium concentration accounts for the reduction of gel viscosity. Compared with the apparent viscosities of gels formed in bottles, the apparent viscosities of gels formed in fractures are a little higher when chromium concentrations are the same. The disproportionate leak-off of polymer and chromium is responsible for this phenomenon. When the gelant is injected into fractured cores, the leak-off degrees of polymer molecules and chromium ions are different because of the huge difference between these two agents. The polymer tends to form a gelant leak-off layer onto fracture face and less polymer molecules will be lost in the leak-off process [29]. However, chromium ions continually leak off or diffuse from fracture into matrix along with water despite the retarding effect of the gelant leak-off layer [30]. This is the disproportionate leak-off of gelant ingredients, and it leads to the decrease of chromium concentration of gelant. On the contrary, the polymer concentration may increase due to the loss of water and it contributes to the gel formation in fractures.
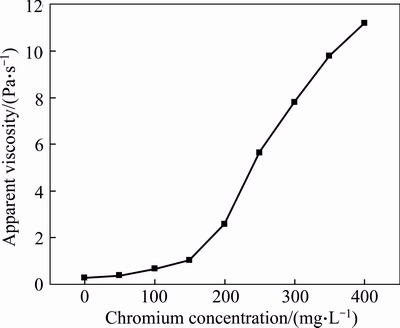
Fig. 11 Effect of chromium concentration on apparent viscosity of gel with 3000 mg/L polymer at 60 ��C
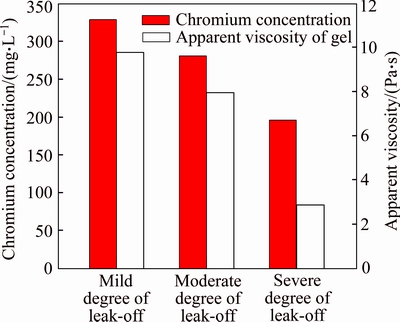
Fig. 12 Residual chromium concentration and apparent viscosity of gel formed in fractured cores after different degrees of gelant leak-off
3.2.2 Effect of gelant leak-off on water shut-off
During the experiments mentioned in section 3.2.1, all the injection volumes of gelant in three cores were 1.0 FV and no additional gelant was used to replenish the leaked-off gelant (especially Cr3+). To investigate the effect of the gelant leak-off on the water shut-off performance of gels for fractures, six volumes of gelants were injected into the same cubic fractured cores to achieve certain normalized chromium concentrations in the produced fluids which were collected at fracture outlets. Data in Table 2 shows that 2.75 FV gelant requires to be injected when the normalized chromium concentration of the produced fluid is 1.0, and it means that the chromium concentration of gelant inside the fracture is the same with that of the fixed gelant. With the reduction of injection volume of gelant, the normalized chromium concentration of the produced fluid decreases, it is only 0.4 when the injection volume of gealnt is 1.45 FV. After gel formation, the subsequent brine injection is conducted to test the water shut-off performance of gel in each fractured core, and the injection pressure differential of six cores are plotted in Fig. 13.
Figure 13 indicates that the injection pressure differential shows a changing trend that first increases and then decreases and finally levels off with the increase of the injection volume of brine. When the normalized chromium concentration is 1.0 (test No. 1), the stable injection pressure differential is 377 kPa; they are respectively 325.8, 204.5, 85.4 and 40.6 kPa when the normalized concentration decreases to 0.9 (test No. 2), 0.8 (test No. 3), 0.6 (test No. 4), and 0.4 (test No. 5), respectively. It means that the water shut-off performance of gel for fracture continually decreases with the reduction of the normalized chromium concentration, especially when the normalized chromium concentration decreases to 0.6 and 0.4. The reduction of chromium concentration of the produced fluid means the corresponding decrease of the chromium concentration of gelant that remains inside the fracture; it results in the decline of gel strength inside fracture and further reduces the water shut-off performance, and it reflects that the leak-off control of chromium is crucial when the polymer gel is applied for fracture treatment.
Table 2 Different degrees of gelant leak-off and corresponding fracture volumes of gelant injected
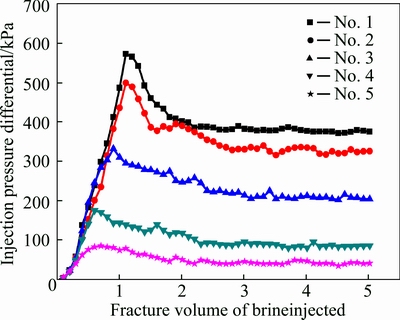
Fig. 13 Pressure differentials of subsequent brine injection obtained with different degrees of gelant leak-off in fractures cores
The water shut-off performance of gel for fractures is closely related to the resident property of gel in fractured media [31]. The general view of gel after gel formation in fractured media is shown in Fig. 14(a). It shows that there are three gel sub-states after gel formation: gel block inside fracture (Fig. 14(b)), gel layer onto fracture face (Fig. 14(c)), and dispersed gel block inside matrix pores (Fig. 14(d)). The gel block inside fracture plays a principal role for the water shut-off during the subsequent brine injection. As shown in the above experiments, because of the disproportionate leak-off of polymer and chromium, the decline of chromium concentration in gelant can greatly affect the gel strength inside the fracture; therefore, an important work is to ensure the sufficient chromium concentration during the gelant injection into fractured media. After gel formation, the ungelled gelant leak-off layer (adsorption layer) becomes into the gel layer onto the fracture face and the dispersed gel block into matrix pores. The gel layer plays a role of cement between the gel block and the fracture face, and the increase of the cementing strength contributes to the enhancement of the washout property of gel block, and then the water shut-off performance of gel can be correspondingly improved [32]. The dispersed gel block inside matrix pores is derived from the gelant retention layer because of the gleant leak-off; it can inhibit the brine channeling between fracture and matrix and improve the cementing strength of gel layer. Therefore, a proper degree of gelant leak-off is necessary to enhance the water shut-off performance of gel for fractures, and the result is consistent with that of GANGULY��s study [19, 33].

Fig. 14 Schematics of gel in fractured media:
3.2.3 Water shut-off performance of a fixed gel along fracture
A fixed gelant consisting of 3000 mg/L polymer and 357.5 mg/L chromium (Cr3+) was used to investigate the water shut-off performance of gel for fractures. After gel formation in cubic fracture core, the injection pressure differential was recorded during the subsequent brine injection and plotted in Fig. 15. It is seen that the pressure differential increases in waves with the increase of the brine injection along the opposite direction of the gelant injection. Each differential between a wave valley and next wave crest indicates the highest injection pressure of brine that needs to extrude the gel out, and it signifies that the gel inside the corresponding section is ruptured. The pressure differentials of sections 1, 2, 3 and 4 are 87.2, 135.6, 172.9 and 203.4 kPa, respectively, as shown in Fig. 15. Data present that the pressure differential along the fracture increases with the extension of the distance from the brine inlet. Because that the injection direction of the subsequent brine is opposite to that of the gelant, so the water shut-off performance of gel for fracture gradually declines with the extension of the distance from the fracture inlet (gelant inlet). The different degrees of gelant leak-off along fracture may be a crucial influencing factor. Although the normalized chromium concentration reaches 1 during the gelant injection, the gelant flow per unit area of the fracture face decreases from section 4 to section 1, and the leak-off volume of gelant gradually also decreases with the extension of the distance from the fracture inlet, as shown in Fig. 10. As mentioned in section 3.2.2, a proper degree of gelant leak-off contributes to improving the resident property of gel inside the fracture and it is favorable to the enhancement of the water shut-off performance. Therefore, the water shut-off performances of gel for sections 4 and 3 are better than those for sections 2 and 1.
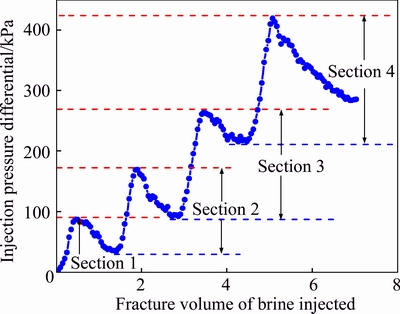
Fig. 15 Pressure differential evolution vs injected fracture volume of subsequent brine
To further testify that a proper leak-off of gelant can enhance the resident property of gel inside the fracture, 2.75 FV fixed gelant was injected into a cylindrical fractured core to ensure that the normalized chromium concentration of the produced fluid reachs 1. The injection rate is 0.2 mL/min and the back pressure is 50 kPa. After gel formation at 60 ��C, 5 FV brine is injected to extrude the gel out of the fracture; then the cylindrical fractured core is dissected into four sections at three positions as shown in Fig. 16(a), and microscopic images (magnified 500 times) of gel layers formed at three positions along the fracture are obtained, as shown in Figs. 16(b)-(d). The gel layer formed at position 1 where is 1 cm away from the fracture inlet is relatively thicker than those formed at positions 2 and 3. The result indicates that because of the contribution of gelant leak-off to the formation of gel layer, a relatively thick gel layer is formed onto the fracture face which is close to the gelant inlet. This is the main reason why the water shut-off performance of gel for fracture is gradually weakened with the extension of the distance from the fracture inlet, and it also reflects that a proper degree of gelant leak-off is conducive to the improvement of water shut-off performance of gel for fractures.

Fig. 16 Schematic and microscopic images of gel layers formed at different positions along fracture:
4 Conclusions
1) During the gelant injection into fractured media, the gelant leaks off from fracture into matrix, and it contributes to the formation of the gelant leak-off layer onto fracture face and into adjacent matrix.
2) The leak-off ratio of gelant from fracture into matrix first declines and finally levels off with the increase of the gelant injection volume because of the formation of the gelant leak-off layer.
3) The polymer concentration of gelant has a great effect on the chromium output in the fluid produced from fractures. With the increase of the polymer concentration, the chromium concentration first decreases and then increases, and the leak-off depth of gelant into matrix is gradually reduced.
4) Because of the obviously high leak-off degree of chromium, the decrease of the chromium concentration inside the fracture greatly reduces the water shut-off performance after the gel formation; therefore, an enough injection volume of gelant is essential to achieve a better water shut-off performance.
5) The water shut-off performance of gel varies along fracture, and it gradually declines with the extension of the distance from the fracture inlet (gelant inlet).
6) A proper degree of gelant leak-off contributes to improving the resident property of gel inside the fracture and it can further enhance the water shut-off performance of gel for fractures.
7) When the polymer/chromium (Cr3+) gel system is used for the water shut-off treatment of fractures, the chromium concentration control is essential because of the high leak-off degree of chromium. Moreover, a proper degree of gelant leak-off contributes to improving the water shut-off performance of gel for fractures; therefore, it is unnecessary to take measures to eliminate the gelant leak-off during its injection process, and the important measure is to ensure the enough injection volume and the concentrations of gelant constituents inside fractures.
References
[1] LIN M, ZHANG G, ZHAO H, ZHAO Q, SUN F. Conformation and plugging properties of crosslinked polymer microspheres for profile control [J]. Colloids Surf A: Physicochem Eng Asp, 2015, 475: 2-8.
[2] SHI J T, VARAVEI A, HUH C, DELSHAD M, SEPEHRNOORI K, LI X F. Viscosity model of preformed microgels for conformance and mobility control [J]. Energy Fuels, 2011, 25: 5033-5037.
[3] SERIGHT R S. Use of preformed gel for conformance control in fractured systems [C]// SPE/DOE Tenth Symposium on Improved Oil Recovery. Tulsa, USA, 1996, SPE 35351.
[4] SIDIQ H, AMIN R, KENNAIRD T. A laboratory investigation of water abatement chemicals for potential use in the Wanaea field [J]. J Petrol Sci Eng, 2009, 65: 89-92.
[5] YAO C J, LEI G L, LI L, GAO X M. Selectivity of pore-scale elastic microspheres as a novel profile control and oil displacement agent [J]. Energy Fuels, 2012, 26: 5092-5101.
[6] ABDULBAKI M, HUH C, SEPEHRNOORI K, DELSHAD M, VARAVEI A. A critical review on use of polymer microgels for conformance control purposes [J]. J Petrol Sci Eng, 2014, 122: 741-753.
[7] GOUDARZI A, ZHANG H, VARAVEI A, TAKSAUDOM P, HU Y P. A laboratory and simulation study of preformed particle gels for water conformance control [J]. Fuel, 2015, 140: 502-513.
[8] BAI B, WEI M, LIU Y. Injecting large volumes of preformed particle gel for water conformance control [J]. Oil Gas Sci Technol, 2012, 67: 941-952.
[9] AL-GHAZA M, AL-DRIWEESH S, AL-SAGR A, KRISHNAN G. A new temporary chemical packer enables efficient stimulation of a lower zone in a gas well [C]// Abu Dhabi International Petroleum Exhibition and Conference. Calgary, Canada, 2012: SPE161651.
[10] ZENG L, LIU H. Influence of fractures on the development of low-permeability sandstone reservoirs: A case study from the Taizhao district, Daqing Oilfield, China [J]. J Petrol Sci Eng, 2010, 72: 120-127.
[11] ZHAO J Z, JIA H, PU W F, LIAO R. Influences of fracture aperture on the water shutoff performance of polyethyleneimine cross-linking partially hydrolyzed polyacrylamide gels in hydraulic fractured reservoirs [J]. Energy Fuels, 2011, 25: 2616-2624.
[12] SERIGHT R S. Reduction of gas and water permeabilities using gels [J]. SPE Prod Fac, 1995, 10: 103-108.
[13] SERIGHT R S, LUNG J, SEIDAI M. Sizing gelant treatments in hydraulically fractured production wells [J]. SPE Prod Fac, 1998, 13: 223-229.
[14] SERIGHT R S. Mechanism for gel propagation through fractures [C]// Rocky Mountain Regional Meeting. Gillette, USA, 1999: SPE 55628.
[15]  M, CHIBOWSKI S, URBAN T. Impact of polyacrylamide with different contents of carboxyl groups on the chromium (III) oxide adsorption properties in aqueous solution [J]. J Hazard Mater, 2015, 283: 815-823.
M, CHIBOWSKI S, URBAN T. Impact of polyacrylamide with different contents of carboxyl groups on the chromium (III) oxide adsorption properties in aqueous solution [J]. J Hazard Mater, 2015, 283: 815-823.
[16] JIA H, PU W F, ZHAO J Z, LIAO R. Experimental investigation of the novel phenol-formaldehyde cross-linking HPAM gel system: Based on the secondary cross-linking method of organic cross-linkers and its gelation performance study after flowing through porous media [J]. Energy Fuels, 2011, 25: 727-736.
[17] STANDNES D C, SKJEVRAK I. Literature review of implemented polymer field projects [J]. J Petrol Sci Eng, 2014, 122: 761-775.
[18] LAKATOS I,  J. Diffusion of chromium ions in polymer/silicate gels [J]. Colloids Surf A: Physicochem Eng Asp, 1998, 141: 425-434.
J. Diffusion of chromium ions in polymer/silicate gels [J]. Colloids Surf A: Physicochem Eng Asp, 1998, 141: 425-434.
[19] GANGULY S. Effect of Leak-off on behavior of Chromium(III)- PHPA gel in fractured media [D]. Lawrence: University of Kansas, 2000.
[20] LE D H, DABHOLKAR D S, MAHADEVAN J, MCQUEEN K. Removal of fracturing gel: A laboratory and modeling investigation accounting for viscous fingering channels [J]. J Petrol Sci Eng, 2012, 88�C89: 145-155.
[21] LIU J X, LU X G, LIU J F, HU S Q, XUE B Q. Mechanism and gelling effects of linked polymer solution in the core [J]. Petrol Explor Develop, 2013, 40: 507-513.
[22] DANG T Q C, CHEN Z, NGUYEN T B N, BAE W. Investigation of isotherm polymer adsorption in porous media [J]. Petro Sci Technol, 2014, 32: 1626-1640.
[23] TIRAFERRI A, BORKOVEC M. Probing effects of polymer adsorption in colloidal particle suspensions by light scattering as relevant for the aquatic environment: An overview [J]. Sci Total Environ, 2015, 535: 131-140.
[24] RAN Q P, SOMASUNDARAN P, MIAO C W, LIU J P, WU S S, SHEN J. Adsorption mechanism of comb polymer dispersants at the cement/water interface [J]. J Disper Sci Technol, 2010, 31: 790-798.
[25] SENGUPTA B, SHARMA V P, UDAYABHANU G. Gelation studies of an organically cross-linked polyacrylamide water shut-off gel system at different temperatures and pH [J]. J Petrol Sci Eng, 2012, 81: 145-150.
[26] BRYANT S L, BARTOSEK M, LOCKHART T P. Propagation of Cr(III) in porous media and its effect on gelant performance [J]. J Petrol Sci Eng, 1996, 16: 1-13.
[27] PANTHI K, MOHANTY K K. Effect of alkaline prefl ush in an alkaline-surfactant-polymer flood [J]. Energy Fuels, 2013, 27: 764-771.
[28] ODUMABO S M, KARPYN Z T, AYALA H L F. Investigation of gas flow hindrance due to fracturing fluid leakoff in low permeability sandstones [J]. J Nat Gas Sci Eng, 2014, 17: 1-12
[29] HATZIGNATIOU D G, NORRIS U L, STAVLAND A. Core-scale simulation of polymer flow through porous media [J]. J Petrol Sci Eng, 2013, 108: 137-150.
[30] GUHAA H, SAIERS J E, BROOK S, JARDINE P, JAYACHANDRAN K. Chromium transport, oxidation, and adsorption in manganese-coated sand [J]. J Contam Hydrol, 2001, 49: 311-334.
[31] BAI Y R, WEI F L, XIONG C M, LI J J, JIANG R Y, XU H B, SHU Y. Effects of fracture and matrix on propagation behavior and water shut-off performance of a polymer gel [J]. Energy Fuels, 2015, 29: 5534-5543.
[32] MORADI-ARAGHI A. A review of thermally stable gels for fluid diversion in petroleum production [J]. J Petrol Sci Eng, 2000, 26: 1-10.
[33] GANGULY S. Leak-off during placement of Cr(III)-partially hydrolyzed polyacrylamide gelling solution in fractured porous media [J]. Transport Porous Med, 2010, 81: 443-460.
(Edited by FANG Jing-hua)
Cite this article as: LI Jun-jian, XIONG Chun-ming, BAI Ying-rui, JIANG Ru-yi, WEI Fa-lin, ZHANG Miao. Leak-off behavior and water shut-off performance of a polymer/chromium (Cr3+) gel in fractured media [J]. Journal of Central South University, 2017, 24(6): 1418-1429. DOI: 10.1007/s11771-017-3546-1.
Foundation item: Project(51404280) supported by the National Natural Science Foundation of China; Project(2014D-5006-0203) supported by the PetroChina Innovation Foundation, China
Received date: 2015-10-23; Accepted date: 2016-04-22
Corresponding author: BAI Ying-rui, PhD; Tel: +86-18562567163; E-mail: smart-byron@163.com

