J. Cent. South Univ. Technol. (2010) 17: 936-942
DOI: 10.1007/s11771-010-0580-7
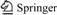
A new multifunctional polymer: Synthesis and characterization of mPEG-b-PAA-grafted chitosan copolymer
LIU Cheng-bin(���б�)1, 2, WANG Xiao-jian(��С��)2, LIU Rong-hua(���ٻ�)1, 2,
WU Yu-lin(������)2, LUO Sheng-lian(��ʤ��)1, 2
1. State Key Laboratory of Chemo/Biosensing and Chemometrics, Hunan University, Changsha 410082, China;
2. College of Chemistry and Chemical Engineering, Hunan University, Changsha 410082, China
? Central South University Press and Springer-Verlag Berlin Heidelberg 2010
Abstract: A new multifunctional mPEG-b-PAA-grafted chitosan copolymer possessing amino and carboxyl groups, mPEG-b-PAA-g-CHI (compound 6), was designed for a potential application in gene/drug delivery and synthesized by the methods of reversible addition-fragmentation chain transfer (RAFT) polymerization of acrylic acid (AA) and grafting reaction of a biodegradable chitosan (CHI) derivative. Completion of the reactions and characterization of the resulting compounds were demonstrated by 1H NMR, FTIR and gel permeation chtomatography (GPC) studies. The results show that the molar ratio of amino groups to carboxyl groups in the copolymer (compound 6) is 0.41?0.59.
Key words: chitosan; polyacrylic acid (PAA); polyethylene glycol (PEG); multifunction polymer; synthesis; characterization
1 Introduction
The synthesis of well-defined block or star copolymers requires a controlled/��living�� polymerization technique. Controlled/��living�� radical polymerization methods including nitroxide-mediated polymerization [1], atom transfer radical polymerization (ATRP) [2], and reversible addition-fragmentation chain transfer (RAFT) polymerization [3] are effective for preparing complex architectures such as blocks and stars. Most particularly, RAFT is arguably the most versatile due to its compatibility with a wide range of vinyl-based monomers, including functional monomers under conditions similar to conventional free radical polymerization [4]. Controlled/��living�� radical poly- merization methods rapidly move to the forefront in construction of drug and gene delivery vehicles [5]. The RAFT technique allows an unprecedented latitude in the synthesis of water soluble or amphiphilic architectures with precise dimensions and appropriate functionality for attachment and targeted delivery of diagnostic and therapeutic agents.
Polyethylene glycol (PEG) (or polyethylene oxide, PEO) is an important hydrophilic polymer because of its unique properties of excellent solubility, chain flexibility, and nontoxicity. PEG-containing polymers have numerous applications in biochemical [6-7] and biomedical [8-9] fields. Particularly, PEO-based drug delivery systems can improve the solubility of water-insoluble drugs, stabilize and protect drugs from being subjected to the surrounding environment, to reduce the nonspecific uptake by the reticuloendothelial system (RES) and prolong the circulation time in the body [10-11]. There are limited reports about the synthesis of the block copolymers containing PEG block through RAFT polymerization method. LI et al [12] reported the synthesis of PEO-b-(DMA-s-NAS)-b- NIPAM thermally responsive triblock copolymers through the method of RAFT polymerization using a PEO-based macro-chain transfer agent (macro-CTA). However, all the block segments from RAFT polymerization of vinyl-based monomers such as N,N- dimethylacrylamide (DMA), N-acryloxysuccinimide (NAS) and N-isopropylacrylamide (NIPAM) are nonbiodegradable and cytotoxic, which limits their biomedical application. Recently, it is suggested that nonbiodegradable polymers accumulate in the nucleus and interact with the gene of the host cells [13]. This effect may cause long-term toxicity. The cytotoxicity of nonbiodegradable blocks strongly depends on their relative molecular mass. To overcome this type of toxicity, the control of the polymerization degree of monomers in RAFT polymerization is very important [14]. But the polymer blocks with a short chain length have not adequate reactive sites for conjugating the diagnostic and therapeutic agents. Thus, the relative molecular mass of the polymers should be selected in consideration of the balance between the cytotoxicity and the functionality [15].
Herein, the block copolymer of mPEG-b-PAA was prepared by the RAFT polymerization of acrylic acid (AA), using an mPEG-based macromolecular chain transfer agent, mPEG-CTA. The resulting diblock copolymer of mPEG-b-PAA, which can be defined as an end-functionalized mPEG with multifunctional groups at one end, was then grafted with a biodegradable chitosan (CHI) derivative, creating a multifunctional copolymer of mPEG-b-PAA-g-CHI (compound 6) possessing amino and carboxyl groups.
2 Experimental
2.1 Materials and synthetic procedure for mPEG-b- PAA-g-CHI (compound 6)
1-Ethyl-3-(3-dimethyllaminopropyl)carbodiimide
(EDC), N-hydroxysuccinimide (NHS), and poly(ethylene glycol) methyl ether (mPEG, Mn=1 900 g/mol, Mw/Mn= 1.10, Mw is weight-average molecular mass, and Mn is number-average molecular mass) were purchased from Alfa Aesar in USA. 2, 2-Azobis-isobutyronitrile (AIBN, 98%, Fluka in USA) was recrystallized twice from ethanol. Carbon disulfide, magnesium turnings, chloroacetyl chloride, and AA (AR grade) were purchased from Aldrich in USA. CHI with a degree of deacetylation of 92% and a number-average molecular mass of 46 000 was purchased from Jinan Haienbei Marine Bioengineering Corporation of China. Other reagents were of the highest commercially available quality and purified before use. The synthetic procedure for mPEG-b-PAA-g-CHI (compound 6) is shown in Fig.1.
2.2 Synthesis of mPEGylated ��-haloesters (mPEG- CA, compound 2)
Poly(ethylene glycol) methyl ether (1.90 g, 1.0 mmol), triethylamine (TEA) (0.5 mL, 3 mmol), and chloroacetyl chloride (0.4 mL, 4 mmol) were mixed in a round- bottom flask using dried tetrahydrofuran (THF) (40 mL) as a solvent, and the solution was refluxed at atmospheric pressure for 2 d. The solvent was then removed, and chloroform (40 mL) was added. The resulting solution
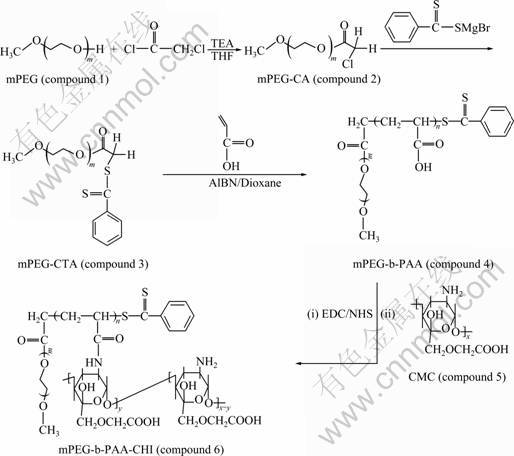
Fig.1 Synthetic procedure for mPEG-b-PAA-g-CHI (compound 6)
was washed with saturated sodium bicarbonate solution, and the yellow organic phase was dried over anhydrous magnesium sulfate. The solvent was then removed in vacuum and further purified by precipitation in cold diethyl ether to give a yellowish solid, an ester of chloroacetic acid (mPEG-CA, compound 2, 2.10 g, yield of 91%).
2.3 Synthesis of mPEG-based chain transfer agent, mPEG-CTA (compound 3)
A THF solution (10 mL) with bromobenzene (2.5 mL, 24 mmol) was added dropwise to a mixture of Mg (0.60 g, 25 mmol) in THF (40 mL) and a crystal of I2 in a two-necked round-bottom flask equipped with a condenser and a magnetic stirrer under a nitrogen atmosphere. The mixture was refluxed for 4 h and then cooled to room temperature. Carbon disulfide (2.0 mL, 32 mmol) in 10 mL of THF was added dropwise, and the solution was stirred at 50 �� for 1 h. This solution was then added at room temperature to 40 mL of THF solution of mPEG-CA (compound 2, 1.90 g, 1 mmol) and refluxed overnight. The orange-red reaction product was precipitated in cold diethyl ether to give mPEG-based chain transfer agent (CTA), mPEG-CTA (compound 3, 2.05 g, yield of 92%).
2.4 Synthesis of block copolymers of mPEG-b-PAA (compound 4)
AA (1.1 mL, 16 mmol), mPEG-CTA (1.69 g, 0.9 mmol), and AIBN (0.03 g, 0.18 mmol) were added along with 1, 4-dioxane (10 mL) to an ampoule. The ampoule was sparged with nitrogen for 30 min and then placed in a preheated oil bath at 70 ��. The reaction was terminated after 24 h by cooling in an ice bath then exposed to air. The crude mPEG-b-PAA block copolymer was then precipitated in cold diethyl ether (2.13 g, conversion 40%, Mw=3 380, Mw/Mn=1.4). The number of acrylic acid unit is 7 calculated from the conversion and the relative molecular mass. The resulting block copolymer can be designated as mPEG43-b-PAA7.
2.5 Synthesis of carboxymethyl chitosan, CMC (compound 5)
The synthesis of carboxymethyl chitosan (CMC, compound 5) was previously reported in Ref.[16]. A general procedure is given as follows: 4.60 g of chitosan (CH2OH: 28 mmol) in a 250-mL flask was activated with 100 mL of aqueous solution of NaOH (1.50 g) at room temperature for 2.5 h. 100 mL of dimethylsulfoxide solution of chloroacetic acid (11.50 g, 122 mmol) was added dropwise into the above activated chitosan solution at 0 �� for 0.5 h, and then hexadecyl trimethyl ammonium bromide (0.25 g) as catalyst was added. The mixture was heated to 50 �� and refluxed for 6 h. The resulting reaction mixture was poured into a beaker containing 200 mL of water, and the pH of the mixture was adjusted to 7.0 with acetic acid to yield a precipitate. The precipitate was purified by filtration, dialysis, and cryodesiccation to get a bright yellow powder, CMC (compound 3, 3.77 g, yield of 80%).
2.6 Synthesis of mPEG-b-PAA-g-CHI (compound 6)
The mixture containing mPEG-b-PAA (compound 4, 0.70 g, 2 mmol of COOH), EDC (0.45 g, 2.4 mmol), and NHS (0.35 g, 3 mmol) in 50 mL of deionized water was refluxed at room temperature for 24 h to get a carboxyl-activated compound 4 solution. Compound 5 (1.10 g, 5.2 mmol of ��NH2) solution in 80 mL of chloroform was added dropwise at room temperature for 0.5 h into the above carboxyl-activated compound 4 solution, and the resulting mixture continued to being stirred at room temperature overnight to obtain a yellowish solid. The solid was further purified by means of column chromatography using Sephadex-25, dialysis and cryodesiccation to get a yellowish powder, mPEG-b-PAA-g-CHI (compound 6, 1.67 g, yield of 92%).
2.7 Characterization
1H nuclear magnetic resonance (1H NMR) spectra were recorded by using a Varian Mercury 400 MHz NMR spectrometer (USA). FTIR spectra were recorded on a Bio-Rad FTS 135 spectrophotometer (USA). To avoid the strong and broad absorption band of moisture at 3 450 and 1 635 cm-1, all specimens were made by mixing and milling the fine powdered polymer with liquid paraffin to a pasty mass. The relative molecular mass and its distribution of the polymers were determined by gel permeation chromatography (GPC) with Waters 1515 GPC (USA).
3 Results and discussion
In most cases, CTAs for RAFT polymerization are not commercially available, so their design and synthesis are very important to efficiently control the polymerization. The preparation of dithiobenzoate-based CTA through the use of ��-haloesters (Fig.2) is remarkable as it often requires a one-step reaction, under a facile reaction condition. Group R is expected to be a sufficiently good leaving group and needs to form a radical that is not only stable but also reactive enough to be added onto the double bond of the monomers. The
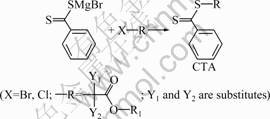
Fig.2 General synthetic procedure of CTA from ��-haloesters
variation of substituents Y1 and Y2 on C atom in �� of the carbonyl group leads to generated radicals of various stability and polymerization control. In the case of small organic CTA with Y1=Y2=H, the stability of the generated radical is poor, resulting in the low monomer conversion and small polymerization degree for the polymerization of styrene and acrylates [17]. So, the macromolecular chain transfer agent of Y1=Y2=H, mPEG-CTA (compound 3), was synthesized and used for the synthesis of PAA blocks with prospectively low polymerization degree.
The dithioester RAFT agent, mPEG-CTA (compound 3), was synthesized by the method similar to that reported by LI et al [12]. First, the precursor of mPEG-ylated ��-haloesters, i.e. mPEG-CA (compound 2), was facilely synthesized through the esterification reaction between poly(ethylene glycol) methyl ether and chloroacetyl chloride with a high yield of 91%. Then, the precursor of mPEG-CA further reacted with dithiobenzoate Grignard agent to get dithioester RAFT agent, mPEG-CTA (compound 3). Their 1H NMR and FTIR spectra are shown in Figs.3 and 4, respectively. There appears the characteristic chemical shift at 3.5-3.7 (m, ��O��CH2CH2��O��) and 4.3 (s, ��CH2Cl) in the 1H NMR spectrum of mPEG-CA (Fig.3(a)). In comparison with the 1H NMR spectrum of compound 2, some new characteristic chemical shifts appear at 7.4, 7.5 and 8.0 (m, Ar��H) in the 1H NMR spectrum of compound 3 (Fig.3(b)). Additionally, in comparison with FTIR spectrum of mPEG (Fig.4(a)), the FTIR spectrum of mPEG-CTA (Fig.4(b)) shows obviously characteristic absorption bands of substituted phenyl group at 1 500 and 1 600 cm-1 (��C=C), 690 and 770 cm-1 (��Ar��H), and 1 740 cm-1 (��C=O). The results obtained thus indicate that the macromolecular chain transfer agent, mPEG-CTA, is obtained.
The RAFT polymerization of AA in 1, 4-dioxane utilizes AIBN as the initiator with mPEG-CTA in a facile

Fig.3 1H NMR spectra of mPEG-CA (a), mPEG-CTA (b), mPEG-b-PAA (c) and mPEG-b-PAA-g-CHI (d)
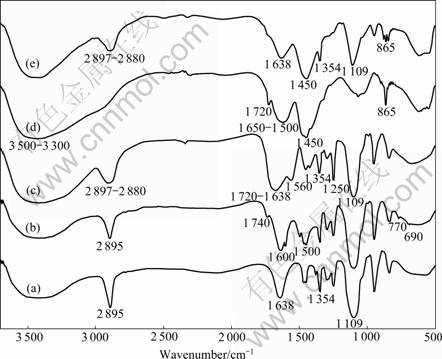
Fig.4 FTIR spectra of mPEG (a), mPEG-CTA (b), mPEG-b-PAA (c), CMC (d), and mPEG-b-PAA-g-CHI (e)
manner at 70 ��. Our attempts to conduct the poly- merization at the temperature lower than 65 �� are not successful due to unacceptable low monomer conversions. However, the polymerization temperature over 70 �� leads to primary radical coupling and AIBN decomposition. In all the cases, the highest monomer conversion of 40% is obtained at 70 ��. The low monomer conversions are probably due to the formation of macromolecular radicals with poor reactivity. The relative molecular mass and its distribution of mPEG-b- PAA block polymers are shown in Fig.5 representing their GPC curves. The resulting mPEG-b-PAA block polymer presents an obviously bimodal and broad relative molecular mass distribution of 1.4, which is possibly attributed to the residual mPEG-CTA. The polymerization degree of AA monomer is 7, satisfying the prospectively limited polymerization degree. So it is a commonly efficient approach to obtain end-functionalized PEGs through RAFT polymerization of monomers with desired functionalities. The resulting mPEG-b-PAA copolymer can be hydrolyzed in the aqueous medium to produce low relative molecular mass PAA blocks with nearly neglectable toxicity. The structure of block copolymer of mPEG-b-PAA was determined by 1H NMR and FTIR spectra shown in Figs.3 and 4, respectively. Compared with the 1H NMR spectrum of mPEG-CTA, there exist the new characteristic chemical shifts at 1.9 (m, CH2 in ��CH2��CH(COOH)��), 2.9 (m, CH in ��CH2��CH(COOH)��) and 9.95 (s, ��COOH) in the 1H NMR spectrum of mPEG-b-PAA (Fig.3(c)). Also, there are some new characteristic FTIR absorption bands: 1 720 cm-1 (��C=O of ��COOH), 2 897-2 880 (��C��H in ��CH2��CH(COOH)��), and 1 250 cm-1 (��C��O and ��O��H of COOH), retaining the characteristic FTIR absorption bands of mPEG-CTA (Fig.4(b)) in the FTIR spectrum of mPEG-b-PAA shown in Fig.4(c).
Compound 6, mPEG-b-PAA-g-CHI, can be facilely synthesized by the acylation reaction between carboxymethyl chitosan (CMC, compound 5) and mPEG-b-PAA (compound 4) with carboxyl groups activated with EDC and NHS. The molar ratio of amino groups to the carboxyl groups in compound 6 is 0.41?0.59 calculated with the method in Ref.[18]. As specially pointed, the content of free amino groups of compound 6 can be adjusted through controlling the reaction molar ratio of compound 4 to compound 5. The 1H NMR and FTIR spectra of compound 6 are shown in Fig.3(d) and Fig.4(e), respectively. There exist the characteristic chemical shifts: 3.2-3.8 (m, mPEG��H), 2.457-4.913 (m, CMC��H) [16], 9.96 (s, CMC�� COO��H), 2.0 (m, CH2 in PAA), and 2.9 (m, CH in PAA) in the 1H NMR spectrum of compound 6. Compared with the FTIR spectrum of compound 4 (Fig.4(c)) and compound 5 (Fig.4(d)) with strong characteristic absorption bands of ��C=O at 1 720 cm-1, ��N��H at 1 500-1 650 cm-1, and ��N��H at 3 300-3 500 cm-1, in the FTIR spectrum of compound 6 (Fig.4(e)) a reduced absorption appears at 3 300-3 500 cm-1 (��N��H), due to the sacrifice of free amino to form acyl amine through the acylation reactions between free amino groups in compound 5 and carboxyl groups in compound 4. Also, the FTIR spectrum of compound 6 retains the intrinsically characteristic absorption bands of compound 4 at 2 880-2 897, 1 354 and 1 109 cm-1, and those of compound 5 at 1 450 and 865 cm-1.
Though polyethylenimine (PEI) is most popularly employed as cationic gene carriers due to its high transfection efficiency [19-20], its acute cytotoxicity is a serious problem. Non-toxic and biodegradable sugar-based materials, in which the primary and secondary amines can be protonated in a physiological milieu, are more and more popularly used in gene delivery with comparable transfection efficiencies to that of PEI [21-23], while the sugar-based materials are widely used in other biomedical and biochemical fields [24-25].
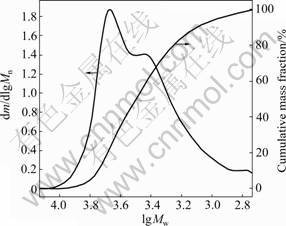
Fig.5 GPC spectra of mPEG-b-PAA (dm is mass change of mPEG-b-PAA)
Most attractively, the as-synthesized copolymers of mPEG-b-PAA-g-CHI (compound 6) possess amino and carboxyl groups. The carboxyl groups may conjugate drugs to form pH-sensitive chemical bonds for pH-controlled release of drugs, and the amino groups may be the potential sites to bind gene, which may achieve the gene or drug delivery [26].
4 Conclusions
(1) A new multifunctional copolymer of mPEG-b- PAA-g-CHI compound (6) possessing amino and carboxyl groups is synthesized by using the well-adopted techniques of RAFT and EDS activation.
(2) The amount of functional groups in compound (6) can be facilely adjusted.
(3) Due to the existence of amino and carboxyl groups in compound (6), the as-synthesized polymer will have a potential in gene or drug delivery.
Acknowledgements
Authors thank Professor NIE Shu-ming (Department of Biomedical Engineering and Chemistry, Emory University and Georgia Institute of Technology, Atlanta) for the discussion and suggestions.
References
[1] Hawker C J, Bosman A W, Harth E. New polymer synthesis by nitroxide mediated living radical polymerizations [J]. Chemical Reviews, 2001, 101: 3661-3688.
[2] Matyjaszewski K, Xia J. Atom transfer radical polymerization [J]. Chemical Reviews, 2001, 101: 2921-2990.
[3] Boyer C, Bulmus V, Davis T P, Ladmiral V, Liu J, Perrier S. Bioapplications of RAFT polymerization [J]. Chemical Reviews, 2009, 109: 5402-5436.
[4] Lowe A B, McCormick C L. Reversible addition�Cfragmentation chain transfer (RAFT) radical polymerization and the synthesis of water-soluble (co)polymers under homogeneous conditions in organic and aqueous media [J]. Progress in Polymer Science, 2007, 32: 283-351.
[5] York A W, Kirkland S E, McCormick C L. Advances in the synthesis of amphiphilic block copolymers via RAFT polymerization: Stimuli-responsive drug and gene delivery [J]. Advanced Drug Delivery Review, 2008, 60: 1018-1036.
[6] Bae J W, Lee E, Park K M, Park K D. Vinyl sulfone-terminated PEG-PLLA diblock copolymer for thiol-reactive polymeric micelle [J]. Macromolecules, 2009, 42: 3437-3442.
[7] Workman H, Flynn P F. Stabilization of RNA oligomers through reverse micelle encapsulation [J]. Journal of the American Chemical Society, 2009, 131: 3806-3807.
[8] YI X, Batrakova E, Banks W A, VINOGRADOV S, KABANOV A V. Protein conjugation with amphiphilic block copolymers for enhanced cellular delivery [J]. Bioconjugate Chemistry, 2008, 19: 1071-1077.
[9] Bae Y, Nishiyama N, Kataoka K. In vivo antitumor activity of the folate-conjugated pH-sensitive polymeric micelle selectively releasing adriamycin in the intracellular acidic compartments [J]. Bioconjugate Chemistry, 2007, 18: 1131-1139.
[10] Fukushima S, Miyata K, Nishiyama N, Kanayama N, Yamasaki Y, Kataoka K. PEGylated polyplex micelles from triblock catiomers with spatially ordered layering of condensed pDNA and buffering units for enhanced intracellular gene delivery [J]. Journal of the American Chemical Society, 2005, 127: 2810-2811.
[11] Peracchia M T, Vauthier C, Passirani C, Couvreur P, Labarre D. Complement consumption by poly(ethylene glycol) in different conformations chemically coupled to poly(isobutyl 2-cyanoacrylate) nanoparticles [J]. Life Sciences, 1997, 61: 749-761.
[12] Li Y, Lokitz B S, McCormick C L. RAFT synthesis of a thermally responsive ABC triblock copolymer incorporating N-acryloxysuccinimide for facile in situ formation of shell cross-linked micelles in aqueous media [J]. Macromolecules, 2006, 39: 81-89.
[13] Godbey W T, Wu K K, Mikos A G. Tracking the intracellu-lar path of poly(ethylenimine) /DNA complexes for gene delivery [J]. Proceedings of the National Academy Science USA, 1999, 96: 5177-5181.
[14] Park T G, Jeong J H, Kim S W. Current status of polymeric gene delivery systems [J]. Advanced Drug Delivery Review, 2006, 58: 467-486.
[15] Kakizawa Y, Kataoka K. Block copolymer micelles for delivery of gene and related compounds [J]. Advanced Drug Delivery Review, 2002, 54: 203-222.
[16] WU Yun-dong, LIU Cheng-bin, ZHAO Xiang-yun, XIANG Jian-nan. A new biodegradable polymer: PEGylated chitosan-g-PEI possessing a hydroxyl group at the PEG end [J]. Journal of Polymer Research, 2008, 15: 181-185.
[17] Farmer S C, Patten T E. (Thiocarbonyl-��-thio)carboxylic acid derivatives as transfer agents in reversible addition-fragmentation chain-transfer polymerizations [J]. Journal of Polymer Science (Part A): Polymer Chemistry, 2002, 40: 555-563.
[18] Muzzarelli R A A. N-(carboxymethylidene)chitosans and N-(carboxymethyl)chitosans: Novel chelating polyampholytes obtained from chitosan glyoxylate [J]. Carbohydrate Research, 1982, 107: 199-214.
[19] Thomas M, Klibanov A M. Enhancing polyethylenimine��s delivery of plasmid DNA into mammalian cells [J]. Proceedings of the National Academy Science USA, 2002, 99: 14640-14645.
[20] Thomas M, Lu J J, Ge Q, Zhang C, Chen J, Klibanov A M. Full deacylation of polyethylenimine dramatically boosts its gene delivery efficiency and specificity to mouse lung [J]. Proceedings of the National Academy Science USA, 2005, 102: 5679-5684.
[21] Pun S H, Bellocq N C, Liu A, Jensen G, Machemer T, Quijano E, Schluep T, Wen S, Engler H, Heidel J, Davis M E. Cyclodextrin-modified polyethylenimine polymers for gene delivery [J]. Bioconjugate Chemistry, 2004, 15: 831-840.
[22] Koping-Hoggard M, Tubulekas I, Guan H, Edwards K, Nilsson M, Varum K M, Artursson P, Chitosan as a nonviral gene delivery system: Structure�Cproperty relationships and characteristics compared with polyethylenimine in vitro and after lung administration in vivo [J]. Gene Therapy, 2001, 8: 1108-1121.
[23] Lee M, Nah J W, Kwon Y, Koh J J, Ko K S, Kim S W. Water-soluble and low molecular weight chitosan-based plasmid DNA delivery [J]. Pharmaceutical Research, 2001, 18: 427-431.
[24] TU Xiang, SU Ben-sheng, LI Xiao-ning, ZHU Jian-rong. Characteristics of extracellular fluorescent substances of aerobic granular sludge in pilot-scale sequencing batch reactor [J]. Journal of Central South University of Technology, 2010, 17(3): 522-528.
[25] Zhang R, Tang M, Bowyer A, Eisenthal R, Hubble J. A novel pH- and ionic-strength-sensitive carboxy methyl dextran hydrogel [J]. Biomaterials, 2005, 26: 4677-4683.
[26] Wang Y, Gao S, Ye W H, Yoon H S, Yang Y Y. Co-delivery of drugs and DNA from cationic core-shell nanoparticles self- assembled from a biodegradable copolymer [J]. Nature Materials, 2006, 5: 791-796.
(Edited by CHEN Wei-ping)
Foundation item: Project(20704011) supported by the National Natural Science Foundation of China; Project(09JJ3027) supported by the Natural Science Foundation of Hunan Province, China; Project(50725825) supported by the National Science Foundation for Distinguished Young Scholars
Received date: 2009-12-11; Accepted date: 2010-03-04
Corresponding author: LIU Cheng-bin, Associate professor; Tel: +86-731-88823805; E-mail: chem_cbliu@hnu.cn