


Trans. Nonferrous Met. Soc. China 22(2012) 694-699
Preparation of metal zinc from hemimorphite by vacuum carbothermic reduction with CaF2 as catalyst
XIONG Li-zhi1, 2, CHEN Qi-yuan1, YIN Zhou-lan1, ZHANG Ping-min1, DING Zhi-ying1, LIU Zhi-xiong1, 2
1. School of Chemistry and Chemical Engineering, Central South University, Changsha 410083, China;
2. College of Biology and Environment Sciences, Jishou University, Jishou 416000, China
Received 12 January 2011; accepted 30 December 2011
Abstract: Zn reduction was investigated by the vacuum carbothermic reduction of hemimorphite with or without CaF2 as catalyst. Results indicate that CaF2 can catalyze the carbothermic reduction of zinc silicate, decrease the reaction temperature and time. The lower the reaction temperature and the more the amount of CaF2, the better the catalytic effect. The optimal process condition is obtained as follows: the addition of about 10% CaF2, the reaction temperature of 1373 K, the molar ratio of C to ZnTotal of 2.5, the pressure of system lower than 20 kPa, the reaction time of about 40 min. Under the optimal process condition, the zinc reduction rate is about 93% from hemimorphite.
Key words: metal zinc; calcium fluoride; catalysis; carbothermic reduction; hemimorphite
1 Introduction
Hemimorphite is one of the most valuable zinc oxide ores as well as the final product when zinc silicate comes into being from sphalerite by weathering over a long period of time. The proportion of hemimorphite to zinc oxide ore is next to that of smithsonite, but the floatability of the former is far less than that of the latter [1]. Due to changeable surface properties and complicated structure, it is difficult to extract metal zinc from hemimorphite. There were few researches on zinc silicate such as hemimorphite. There were different opinions on hemimorphite leaching behaviors. Many researchers [2,3] held that hemimorphite can not be dissolved, which makes the low leaching ratio of zinc oxide ore. Some researchers [4] thought that the silica with high content usually entered solution as silica gel with zinc when hydrometallurgical method was used, and the formation of silica gel made filtration difficult. In pyrometallurgical process, due to the differences of structure and properties between zinc oxide and zinc silicate formed from the dehydration of hemimorphite [5], the reduction of hemimorphite needs high temperature, thus the energy consumption increases greatly. Consequently, it is urgent to find an effective method to exploit and utilize hemimorphite. Vacuum metallurgical technology has high-efficiency on improving metal recovery and shortening process flow, and meets the environmentally-friendly and resource-saving requirements [6-8]. On the basis of the advantages of vacuum metallurgy technology, CaF2 is added as catalyst to cut down energy consumption and increase the Zn recovery.
2 Experimental
2.1 Materials
Hemimorphite from Lanping of Yunnan Province, China, was employed. Tables 1 and 2 show chemical composition and phases of the original mineral, respectively.
The content of zinc is 49.13% and the major mineral is hemimorphite (Zn4Si2O7(OH)2��H2O) in the original mineral. Coking coal from Douli Mountain of Lianyuan, Hunan Province in China��was used as not only the reductant but also the binder in this study. The chemical composition of coal is shown in Table 3. Other chemical reagents such as CaO, CaF2 and NaF are analytically pure.
Table 1 Chemical composition of original mineral (mass fraction, %)

Table 2 Zn content in different phases of original mineral
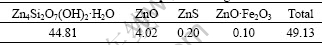
Table 3 Chemical composition of coking coal

2.2 Facilities
The reduction was carried out in a vacuum furnace with classification condensation, which was made by Hunan Acme Technology Co., Ltd. The vacuum furnace was designed according to the characteristics of vacuum carbothermic reduction of hemimorphite and utilization of the waste heat for the condensation classification of vapour. It is composed of vacuum furnace (Fig. 1), furnace temperature control system, vacuum acquisition system, water-cooling system and gas-supply system.
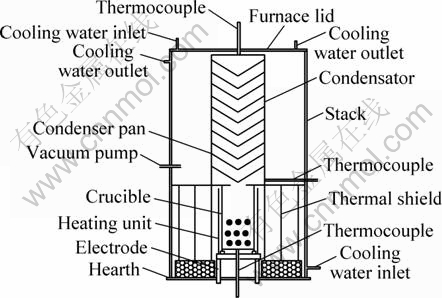
Fig. 1 Sketch map of vacuum furnace with classification condensation
2.3 Experiment process
The experiment process is described as:
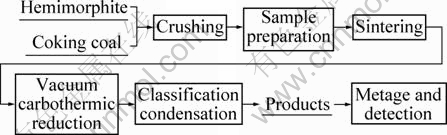
Hemimorphite and coking coal were sifted through a sieve with sieve pore size of 0.214 mm (70 mesh). Hemimorphite, coal and water (sometimes adding additive) were well mixed and made into cylinder samples (the radius was 10 mm, the height was 10 mm). The dried samples were sintered at 823 K for 30 min to ensure the sample strength. Then the samples were fed into the crucible of vacuum furnace and quickly heated to the experimental temperature at the required pressure. In this process, zinc silicate was reduced to metal zinc, then vapour of zinc and other volatile substances entered into a classification condenser. The classification condenser consisted of many bottom-up condenser pans where the temperature decreased from 1123 K to 300 K in the experiment process and the substances could be separated, purified and condensed in a suitable place of classification condenser to obtain pure zinc (purity was above 99.95% [9]), pure cadmium and other products. At last, products and slag were weighed and detected. The relevant experimental conditions are listed in Tables 4 and 5.
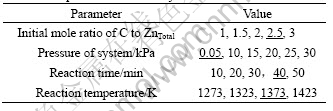
Table 4 Processing parameters in vacuum carbothermic reduction experiments without catalyst
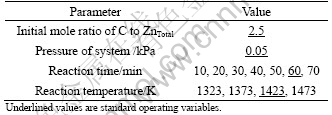
Table 5 Processing parameters in vacuum carbothermic reduction experiments with catalyst
3 Results and discussion
3.1 Calculation of Zn reduction rate and analysis of zinc content
Zinc reduction rate, ��, can be calculated according to:
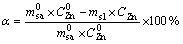 (1)
(1)
where t is the reaction time, min; 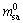 represents the mass of sample at t=0; ms1 is the mass of slag at t;
represents the mass of sample at t=0; ms1 is the mass of slag at t;  is the content of Zn in sample at t=0; CZn is the content of Zn in slag at t, g/g.
is the content of Zn in sample at t=0; CZn is the content of Zn in slag at t, g/g.
The contents of Zn in the samples and slag were analyzed according to GB/T 8151.1��2000. When the content of Zn in the slag was very low, it was determined by ICP-OES (Intrepid II XSP, American Hermo Electron Corporation).
3.2 Effect of conditions of vacuum carbon reduction without additives
3.2.1 Effects of reaction time
Figure 2 shows the effect of reaction time from 10 min to 70 min on the zinc reduction rate. It can be seen that the zinc reduction rate increases with prolonging the reaction time. As the reaction time is over 40 min, the growth rate of zinc reduction rate gets less. The results indicate that the decrease of the content of zinc silicate during the reaction process slows down the reduction reaction.
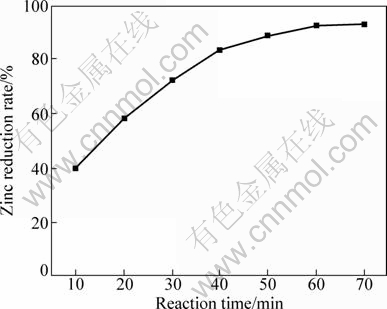
Fig. 2 Effect of reaction time on zinc reduction rate without catalyst
3.2.2 Effects of reaction temperature
Experiments were conducted at four different temperatures ranging from 1323 to 1473 K, and the results are shown in Fig. 3. It is found that the increase in reaction temperature accelerates the zinc reduction rate significantly. Taking into account of power consumption and production costs, the most suitable reaction temperature is about 1423 K, almost 250 ��C higher than that of zinc oxide which is about 1173 K [9,10]. The critical reaction temperature of zinc silicate reduction is about 50 ��C higher than that of zinc oxide [5]. The reduction temperature may be decreased by the addition of some reagent.
3.3 Effects of additives
To select suitable additive, many tentative experiments were carried out. Experiment results show that fluorine salt could enhance zinc reduction rate in the reduction reaction.
Figure 4 shows the effect of adding 10% CaO, 10% CaF2 and 10% NaF on the zinc reduction rate, respectively. It is discovered that not only CaF2 but also NaF can enhance zinc reduction rate greatly, but adding 10% CaO has little effect on the zinc reduction rate. The experimental results indicate that F-ion helps to promote carbothermic reduction of zinc silicate.
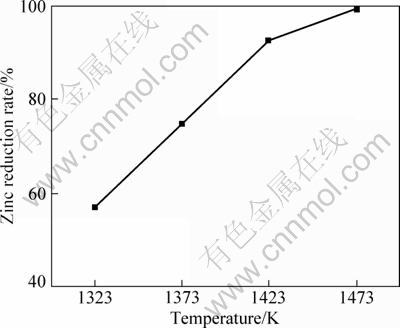
Fig. 3 Effect of reaction temperature on zinc reduction rate without catalyst
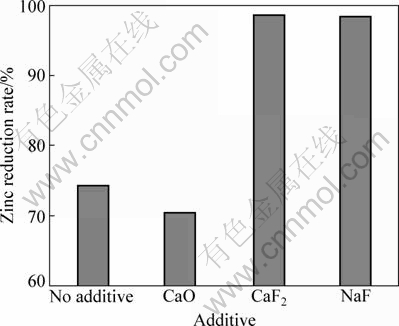
Fig. 4 Effect of different additives on zinc reduction rate
To explore functions of F-ion in the reduction process, an experiment was carried out with CaF2 as additive. Samples consisting of 10% CaF2 and 90% dehydrated ore were distilled in vacuum at 1373 K for 60 min. The experiment results from the cooling slag by XRD analysis are shown in Fig. 5. Samples with hemimorphite, coking coal (molar ratio of C to ZnTotal was 2.5) and 10% CaF2 were distilled and reduced in vacuum at 1373 K for 30 min. The experiment results from the cooling slag by XRD are shown in Fig. 6.
From Fig. 5 it can be seen that the slag is mainly composed of zinc silicate and CaF2, and the ratio of their mass changes very little. If zinc silicate was decomposed into ZnO and SiO2, then ZnO would evaporate in the experiment process [5], and the slag should contain more SiO2. So, CaF2 can not promote decomposition of zinc silicate. From Fig.6 it is found that there is a large amount of SiO2 and a small amount of Zn2SiO4 and CaF2 without other fluoride in the slag, namely, CaF2 can catalyze the carbothermic reduction of zinc silicate.
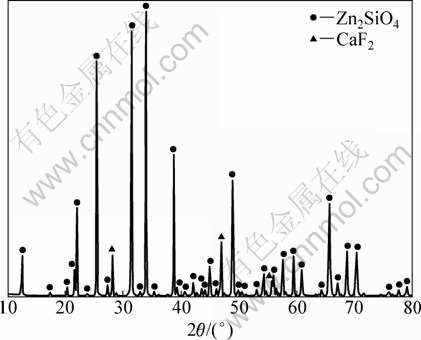
Fig. 5 XRD pattern of slag from dehydrated ore containing CaF2 by distillation of vacuum
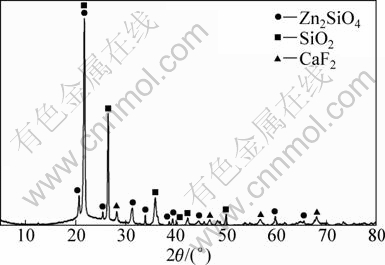
Fig. 6 XRD pattern of slag from hemimorphite containing CaF2 by vacuum carbothermic reduction
3.4 Catalytic effects of temperature
To study catalytic effects of temperature, the reduction experiments were conducted at four different temperatures (1273 K, 1323 K, 1373 K and 1423 K), on the conditions of no adding CaF2, adding 10%CaF2, respectively, and the results are shown in Fig. 7. The zinc reduction rate can quickly rise up by increasing the reaction temperature, CaF2 between 0 and 10%. It is found that CaF2 has a remarkable catalytic effect at a low temperature. This is because reaction temperature has great influence on reduction of zinc silicate. At a lower temperature, the speed of reduction reaction is slow, and the reduction of zinc silicate by coal has a notable effect by adding CaF2. The speed will increase significantly with the rise of reaction temperature, so catalytic effect will become weak. To enhance catalytic effect reaction should be carried out at lower temperature, but the slow speed would influence the production efficiency. Thus the suitable reaction temperature is nearly 1373 K with CaF2 as catalyst.
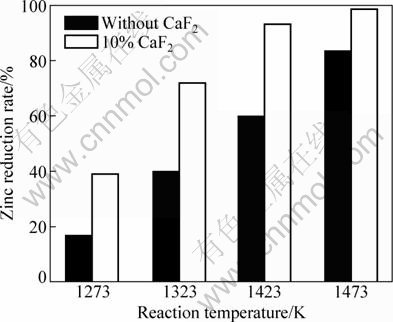
Fig. 7 Comparison of effect of catalyst on reduction reaction at different temperatures
3.5 Effects of vacuum carbon reduction with CaF2 as catalyst
3.5.1 Effect of mass of CaF2
Figure 8 shows the effect of different amount of CaF2 added in the samples on the zinc reduction rate. It can be seen that the zinc reduction rate rises with increasing the amount of CaF2 in the samples. And when the addition of CaF2 is over 10%, the reduction rate increases slowly. So, the suitable addition of CaF2 is about 10%.
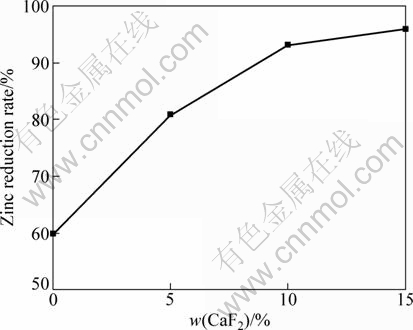
Fig. 8 Effect of amount of CaF2 added on zinc reduction rate with CaF2 as catalyst
3.5.2 Effect of initial molar ratio of C to ZnTotal
The experimental results of the initial molar ratio of C to Zn are shown in Fig. 9. The higher the initial molar ratio of C to ZnTotal is, the higher the reduction rate is. A higher carbon content in the solid samples, which means a higher initial molar ratio of C to ZnTotal, provides more contact surface area for C and Zn2SiO4, then accelerates reaction, produces more Zn and CO which enhance reaction rate of Zn2SiO4 reduction [11-17]. But when the initial molar ratio of C to ZnTotal surpasses 2.5, the reduction rate is enhanced slowly. To reduce power consumption and production costs, the initial molar ratio of C to ZnTotal should be nearly 2.5.
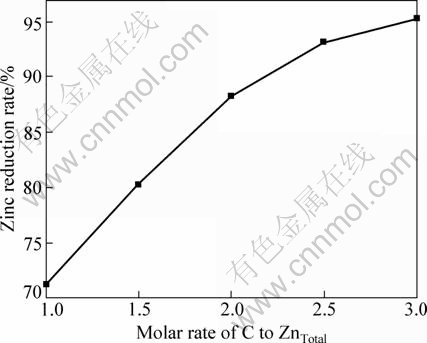
Fig. 9 Effect of initial molar ratio of C to ZnTotal on zinc reduction rate with CaF2 as catalyst
3.5.3 Effect of pressure of system
The effect of system pressure is presented in Fig. 10 (the pressure of system was controlled by gas-supply system). It can be seen that the higher the pressure of system over 20 kPa, the lower the reduction rate. Because system pressure becomes large, higher pressure on the samples brings about and the diffusion resistance of Zn vapor is increased in the samples and hence a low reduction rate of zinc is afforded [6]. The variance of zinc reduction rate may be ignored when system pressure is ranges from 50 Pa to 20 kPa. There is a critical pressure (pc) in zinc evaporation at some temperature. As p>pC (p is pressure of system), the rate of evaporation decreases with increasing system pressure; as p
C, the rate of evaporation does not change with increasing or decreasing system pressure [6,18]. The pressure of system should not exceed 20 kPa by carbothermic reduction of zinc silicate at 1373 K.
3.5.4 Effect of reaction time
Figure 11 shows the effect of reaction time on the zinc reduction rate. It is similar to case of no catalyst(Fig. 2) in which the zinc reduction rate increased with prolonging the reacting time. But there is somewhat difference: with CaF2 as catalysis, the reaction temperature is 50 ��C lower than that without CaF2, and the reaction time is only roughly two-thirds of that without CaF2.
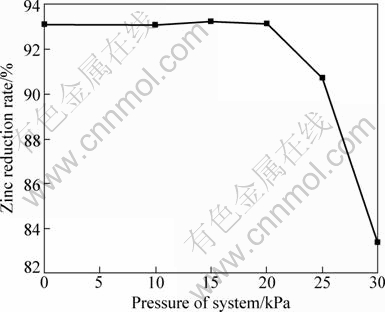
Fig. 10 Effect of system pressure on zinc reduction rate with CaF2 as catalyst
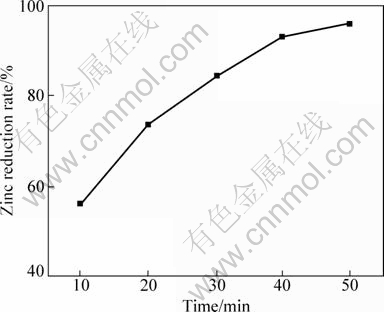
Fig. 11 Effect of reaction time on zinc reduction rate with CaF2 as catalyst
3.5.5 Effect of reaction temperature
In order to investigate the influence of the reaction temperature with add ion of 10% CaF2 on the zinc reduction rate, experiments were performed at 1273, 1323, 1373 and 1423 K respectively. The results are shown in Fig. 12. It indicates that as temperature increases, the zinc reduction rate is notably enhanced. Similar results are observed from Fig. 3 without CaF2. When the reaction temperature is 1373 K, the zinc reduction rate reaches about 93% at 40 min with the addition of 10% CaF2, but it is only about 92% at 1423 K at 60 min when CaF2 is not added. The obtained results show that catalysis of CaF2 can remarkably decrease reduction temperature and reaction time.
3.6 Main characterization of condensates by XRD
Figure 13 shows the XRD pattern of the main condensate. From Fig. 13 it is found that the main condensate is metal zinc.
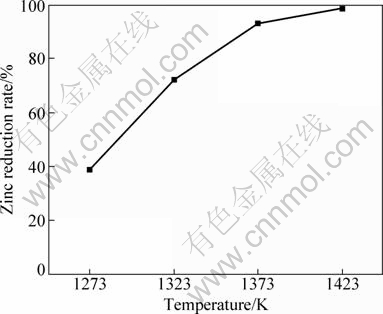
Fig. 12 Effect of reaction temperature on zinc reduction rate with CaF2 as catalyst
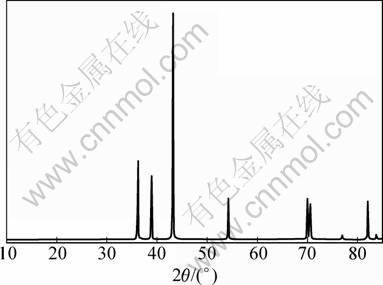
Fig. 13 XRD pattern of main condensate
4 Conclusions
1) CaF2 could catalyze effectively the carbothermic reduction of zinc silicate.
2) Not only reaction temperature but also the reaction time is decreased by adding CaF2.
3) The optimal process conditions are obtained as follows: the addition of CaF2 is about 10%, the molar ratio of C to ZnTotal is 2.5, the reaction temperature is 1373 K, the pressure of system is lower than 20 kPa, and the reaction time is about 40 min. Under the optimal process condition, the zinc reduction rate is about 93% from hemimorphite.
References
[1] CHEN Ye, CHEN Jian-hua, LI Yu-qiong. Experimental research on flotation of hemimorphite ore [J]. Metal Mine, 2008, 379(1): 67-69, 120. (in Chinese)
[2] ALETAN G. Ammonia leaching of oxidized lead-zinc ores of Iran [J]. Geol Survey of Iran, 1969, 15: 19-66.
[3] BRETHERTON S E. Preparation of ore containing zinc for the recovery of other metals such as silver, gold, copper and lead by the elimination and subsequent recovery of the zinc as a chemically pure product [J]. Transition Metal Chemistry, 1914, 47: 82-90.
[4] HUA Y, LIN Z, YAN Z. Application of microwave irradiation to quick leach of zinc silicate ore [J]. Minerals Engineering, 2002, 15: 451-456.
[5] XU Cai-dong, LIN Rong. Physics chemistry of zinc metallurgy [M]. Shanghai: Shanghai Science and Technology Press, 1979: 40-66, 143-150. (in Chinese)
[6] DAI Yong-nian, YANG Bin. Vacuum metallurgy of nonferrous materials [M]. Beijing: Metallurgical Industry Press, 2000: 7-60. (in Chinese)
[7] DAI Yong-nian, ZHAO Zhong. Vacuum metallurgy [M]. Beijing: Metallurgical Industry Press, 1998. (in Chinese)
[8] HAN Long, YANG Bin, DAI Yong-nian, LIU Da-chun, YANG Bu-zheng. Applications of vacuum metallurgical technology in recovery of secondary zinc-based resources [J]. Vacuum, 2008, 45(2): 20-22. (in Chinese)
[9] XIONG Li-zhi, CHEN Qi-yuan, YIN Zhou-lan, ZHANG Ping-min. Theoretical analysis and experimental research on treatment of zinc oxide ore using vacuum carbothermic reduction [J]. Chinese Journal of Vacuum Science and Technology, 2010, 30(3): 321-324. (in Chinese)
[10] XIONG Li-zhi, CHEN Qi-yuan, YIN Zhou-lan, ZHANG Ping-min. Thermodynamic analysis and experimental research on treatment of low-grade Pb/Zn oxide ore by vacuum carbothermic reduction [J]. Mining and Metallurgical Engineering, 2009, 29(4): 64-66. (in Chinese)
[11] YANG Xue-min, GUO Zhan-cheng. Research on the reduction mechanism of iron ore pellets containing graphite [J]. The Chinese Journal of Process Engineering, 1995, 16(2): 118-126. (in Chinese)
[12] SUN S, LU W K. Mathematical modeling of reactions in iron/coal composites [J]. ISIJ International, 1993, 33(10): 1062-1069.
[13] DING Y L, WARNER N A. Kinetics and mechanism of reduction of carbon-chromite composite pellets [J]. Ironmaking and Steelmaking, 1997, 24(3): 224-229.
[14] XIONG Li-zhi, CHEN Qi-yuan, YIN Zhou-lan, ZHANG Ping-min. Vacuum carbothermal reduction kinetics of zinc oxide ore [J]. The Chinese Journal of Process Engineering, 2010, 10(1): 133-137. (in Chinese)
[15] HUANG C H, LIN C I, CHEN H K. Kinetics of the carbothermic reduction of zinc sulfide in the presence of calcium oxide [J]. Journal of the Chinese Institute of Chemical Engineers, 2007, 38: 143-149.
[16] HUANG H C, LIN C I, CHEN H K. Zinc recovery from spent ZnO catalyst by carbon in the presence of calcium carbonate [J]. Metallurgical and Materials Transactions B, 2004, 35: 55-63.
[17] HUANG C H, LIN C I, CHEN H K. Carbothermic reduction of zinc sulfide in the presence of sodium carbonate [J]. Journal of the Chinese Institute of Chemical Engineers, 2008, 39: 457-465.
[18] CHEN Wen, HU Cui. Studies on the critical pressure of bismuth in the vacuum distillation process [J]. Non-ferrous Mining and Metallurgy, 2000, 16(3): 25-29. (in Chinese)
CaF2�����̼�Ȼ�ԭ�켫���Ʊ�����п
����֥1, 2������Ԫ1��������1����ƽ��1������Ӣ1����־��1, 2
1. ���ϴ�ѧ ��ѧ����ѧԺ����ɳ 410083��2. ���״�ѧ ������Դ�뻷����ѧѧԺ������ 416000
ժ Ҫ��ͨ���켫�����̼�Ȼ�ԭ���飬�о�����CaF2�Ͷ�̼�Ȼ�ԭ����п�ĵ�Ӱ�졣���������CaF2�ܴ�����п��̼�Ȼ�ԭ�����ͻ�ԭ�¶ȣ����̷�Ӧʱ�䣻�¶�Խ�ͣ���Ч��Խ�ã�CaF2��������Խ�࣬��Ч��Խ���ԡ�CaF2�����̼�Ȼ�ԭ�켫��Ľϼѹ��������ǣ�CaF2��������Լ10%����ԭ�����¶�1373 K��C/Zn�������ʵ�����2.5��ϵͳѹǿ����20 kPa����Ӧʱ��Լ40 min���ڽϼѹ��������£��켫����Լ93%��п����ԭ���������
�ؼ��ʣ�����п��CaF2�������ã����̼�Ȼ�ԭ���켫��
(Edited by YANG Hua)
Foundation item: Project (2007CB613601) supported by the National Basic Research Program of China; Project (51142001) supported the National Natural Science Foundation of China; Project (11B099) supported the Scientific Research Fund of the Human Provincial Education Department, China; Project (2011TT2038) supported the Scientific Research Fund of the Human Provincial Scientific and Technological Department, China
Corresponding author: CHEN Qi-yuan; Tel: +86-731-88877843; E-mail: cqy@mail.csu.edu.cn
DOI: 10.1016/S1003-6326(11)61233-8