
Catalytic activity of Au/Fe-PILC and Au/Fe-oxide catalysts for catalytic combustion of formaldehyde
LI Chang-yan (李常艳)1, SHEN Yue-nian (沈岳年)1, HU Rui-sheng (胡瑞生)1,
LI Pei-pei (李沛培)2, ZHANG Jun (张 军)1
1. College of Chemistry and Chemical Engineering, Inner Mongolia University, Huhhot 010021, China;
2. Lanzhou Institute of Chemical Physics, Chinese Academy of Sciences, Lanzhou 730050, China
Received 15 July 2007; accepted 10 September 2007
Abstract: Iron polymeric hydroxygroups pillared clays (Fe-PILC) were prepared by Na+-montmorillonite with iron pillaring agent. 2.01Au/Fe-PILC catalyst was obtained by deposited-precipitation (DP) method. 2.52Au/Fe-oxide catalyst was prepared by co-precipitation method. The catalytic activity of these catalysts was measured by catalytic combustion of formaldehyde. The catalyst of 2.01Au/Fe-PILC exhibits the high catalytic activity. The catalytic combustion reaction of formaldehyde proceeds at considerable rates at 20 ℃ and complete burn-off of formaldehyde is achieved at 120 ℃. The structure of catalysts, the valence state of gold and the size of gold particles were investigated by means of X-ray powder diffractometry, X-ray photoelectron spectroscopy and transmission electron microscopy. The results show that gold atoms with partially positive charge exist in the catalyst and play an important role in the catalytic activity. In addition, nano-sized, well-dispersed gold particles and good adsorption properties of support are necessary to obtain high activity Au catalysts for catalytic combustion of formaldehyde.
Key words: Au/Fe-PILC catalyst; catalytic combustion of formaldehyde; gold with positive charge; gold nanocrystals
1 Introduction
Formaldehyde (HCHO) is a common indoor contaminant which is widely used in the production of isolation materials, furniture, disinfectants, water-based paints (as additive), and exists in tobacco smoke, exhaust gases and even wood. Chronic exposure to the air polluted by HCHO with low concentration may cause adverse effects on health, such as skin disease, tracheitis and even lung cancer. In general, the techniques for the removal of the low concentration HCHO can be classified into two major classes: recycling and combustion of the formaldehyde[1-2]. Although it is technically feasible for the recycling of the low concentration HCHO, it is not an economic approach. Accordingly, catalytic combustion at relatively low temperature appears to be the potential solution for this environmental problem by virtue of its low energy cost.
Noble metals usually exhibit superior catalytic activity for the removal of formaldehyde/methanol[3]. However, noble metals are expensive and vulnerable to poisoning and the applications of these catalysts in commercial scale have been seriously impeded by these serious concerns. Some metal oxides[4] and transition metal perovskites[5] were investigated as substitutes for the noble metals, but their activities for catalytic combustion were unsatisfactory. Consequently, developing new catalysts, which do not contain noble metals and are able to catalyze the HCHO at moderate temperatures, is vital for HCHO combustion technique.
Naturally existing montmorillonite clays are layered alumni-silicates, which are held together by weak electrostatic force. New classes of materials have been recently developed by exchanging the small Na+ ions that are normally found between these layers with inorganic metal ions of larger size and charge, such as iron, complexes, etc[6]. Fe-PILC was adapted because of good properties on catalysis and adsorption, which is few publicly reported.
In the present work, active species gold was supported by deposited-precipitation (DP) method. The catalytic activity of Au/Fe-PILC and Au/Fe-oxide catalysts for catalytic combustion of formaldehyde was measured.
2 Experimental
2.1 Catalyst preparation
Smectite used in this work came from Inner Mongolia, China. Ca-montmorillonite was obtained after washing, removing excess salt, centrifuging and drying. Then Ca-montmorillonite (10 g) was dispersed in 0.5 mol/L Na2CO3 solution (1 L) for 5 h at 65 ℃ in order to exchange Na+ and get Na+-montmorillonite[7].
0.5 mol/L Na2CO3 solution was slowly dipped into 0.1 mol/L Fe(NO3)3 solutions at pH=2.2 [8]. The iron pillaring agent was kept for 24 h at room temperature, then dipped into 1% Na+-montmorillonite (pH=9.5). Through dispersing and washing, Fe-PILC was obtained. The centrifuged Fe-PILC was separated into two parts. One was used as reference. The other was added into 2.5 mmol HAuCl4 solution and reacted to get Au/Fe-PILC catalyst after washing, drying and calcimine. Au/Fe- oxide catalysts were prepared by a co-precipitation approach[9].
2.2 Catalyst characterization
The structures of the catalysts were characterized by X-ray powder diffractometry (XRD, Phillips PW 1700X, Cu Kα, 40 kV, 20 mA). The morphologies of the catalysts were obtained by transmission electron microscopy (TEM, JEM2010, 120 kV).
The bonding energy were measured by X-ray photoelectron spectroscopy (XPS, VG ESCALAB MK-2, Al Kα=1 486.6 eV, voltage 12.5 kV, power 250 W, E(C1S)=284.6 eV, vacuum 2×10-8 Pa).
The content of Au was measured by atomic absorption spectroscopy (AAS, Analyst 200 USA). In samples, Au contents were found to be 2.01% and 2.52% (mass fraction), respectively. The catalysts were denoted as 2.01Au/Fe-PILC and 2.52Au/Fe-oxide. The samples without gold were also denoted as reference, named Fe-PILC and Fe-oxide.
2.3 Catalytic activity measurement
The activity of the catalysts for catalytic combustion of formaldehyde was tested in a conventional continuous flow U-shape glass (d 8 mm) reactor at atmosphere pressure. Catalyst (0.2 g, 100-175 μm) was loaded. The gaseous reactant mixture was fed to the reactor by an air flow from a cylinder, which passes through a container filled with formalin (37% formaldehyde). In order to keep the temperature of formalin at 0 ℃, the formalin container was immersed in a mixture of water and ice. The flow rate passing through the reactor was controlled at 180 mL/min by a mass-flow meter. This flow rate is equivalent to 6.25 mg/m3 of formaldehyde. The effluent gases were analyzed on-line by a gas chromatograph (GC-8A, Shimadzu) equipped with thermal conductivity detector (TCD) and packed with GDX-403. Column temperature was 110 ℃ and TCD detection temperature was 160 ℃. Current was kept at 80 mA. Helium was used as carrying gas (30 mL/min) for the GC analysis.
3 Results and discussion
3.1 Structural features of catalysts
Fig.1 illustrates the XRD patterns for the pillared products and Fe-oxide. The X-ray basal spacings (d001) are described in Table 1.
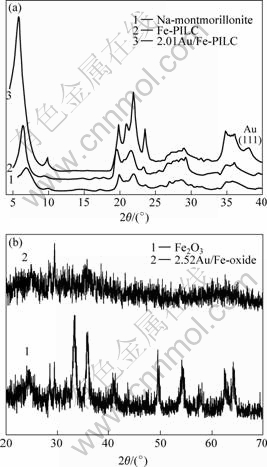
Fig.1 XRD patterns of 2.01Au/Fe-PILC(a), 2.52Au/Fe-oxide(b) catalysts and some references
Table 1 d001-basal spacing values

The low 2θ peak of the basal spacing, d001, was initially positioned near 7.091?, and the peak was relatively strong, indicating that the effect of sodium exchange montmorillonite was distinguished[10]. The basal spacing of Fe-PILC was equal to that of Na+-montmorillonite. As for 2.01Au/Fe-PILC, the peak of the low 2θ shifted to higher basal spacing and increased in intensity, which indicated nano-gold particle intercalated into the interlayer. Other major peaks in Fig.1(a) are assigned as follows. The peaks at 2θ of 19.6? and 34.9? are assigned to the two-dimensional diffraction. Weak peaks of metallic gold (Au0) at 2θ values of 38.2? is attributed to diffraction from (111) lattice plane. When gold is loaded in ion states, it is highly dispersed in small size so that its diffraction is too weak to be observed. Fig.1(b) shows the XRD patterns of 2.52Au/Fe-oxide catalyst. From the reference, The high peaks at 2θ of 33.36? and 35.79? are confirmed to be the phase of Fe2O3 [11]. When gold was supported, no sharp peaks for either iron oxide or metallic gold could be observed, indicating that their crystal sizes are very small [12-13].
3.2 Catalytic activity
Fig.2 and Fig.3 show the TEM photographs and the size distribution of gold particles. The spherical gold nanoparticles were observed as dark spots, which were homogeneously dispersed on the surfaces of Fe-PILC support (Fig.2). The mean particle size of gold was determined in the narrow range of 2-5 nm. While in the 2.52Au/Fe-Oxide sample, the iron oxide component of the catalyst consisted of aggregates of fine particles with sizes up to several tens of nanometers. The mean particle size of gold was determined to be in the range of 10-15 nm (Fig.3).
CO2 and H2O were the products of the catalytic HCHO combustion. The experimental results (Fig.4 and Fig.5) indicated clearly that 2.01Au/Fe-PILC catalyst showed excellent catalytic behavior. The burn-off temperature of 2.01Au/Fe-PILC is lower by 40 ℃ than that of 2.52Au/Fe-Oxide catalyst. Based on TEM image, 2-5 nm gold particles were prerequisite for high-activity gold catalysts[14]. Moreover the Fe-PILC was interlayer support and had much micro pores, which resulted in active species gold homogeneously dispersed on the surfaces of Fe-PILC support and over aggregates of nanogold particles were got. The more the value of d001 is, the better the adsorption is.
3.3 Oxidation state of gold
XPS analyses were carried out in order to determine the valence state of gold on 2.01Au/Fe-PILC and 2.52Au/Fe-oxide catalysts. The existing forms and the states of the gold in the catalyst were crucial for explaining the good catalytic activity. It has been
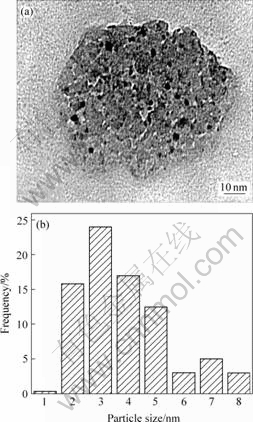
Fig.2 TEM photograph(a) and gold particle size distribution(b) on 2.01Au/Fe-PILC catalyst
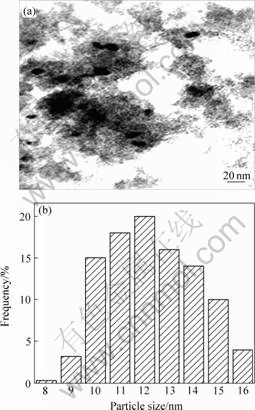
Fig.3 TEM photograph(a) and gold particle size distribution(b) on 2.52Au/Fe-Oxide catalyst

Fig.4 Catalytic combustion of formaldehyde on 2.01Au/Fe- PILLC catalyst

Fig.5 Catalytic combustion of formaldehyde on 2.52Au/Fe- Oxide catalyst
suggested that gold species existing in states with different charges, such as Au3+, Au1+. Auδ+ (0<δ<1) and Au0 on the support are the active components for catalytic combustion of VOCs [9, 15-16].
The XPS spectra of 2.01Au/Fe-PILLC and 2.52Au/Fe-Oxide catalysts in the Au 4f region are illustrated in Fig.6 and Fig.7, respectively. There are two peaks for gold species corresponding to the Au 4f7/2 and Au 4f5/2 transitions, but the positions of the peaks vary with the state of the gold species in the samples. In the 2.01Au/Fe-PILLC catalyst, two major peaks in XPS spectra are from different gold species (Au3 and Au0). The peak of Au 4f5/2 is higher than that of the Au 4f7/2, compared with the metallic gold Au0 (83.8 and 87.45 eV) [17]. It is highly possible that Au(OH)3 coexists with the metallic gold. Moreover, Au3+(Au 4f7/2, 86.5 eV)species is more than any other gold species[18-20]. It can be seen that metallic gold Au0 (83.7.0 and 87.45 eV) and gold species Auδ+ (84.9 and 88.6 eV) account for most of the gold species in the 2.52Au/Fe-Oxide catalyst[20-21].
The difference between the binding energies of Au 4f of samples and those of bulk metallic gold can be attributed to partial flow of the 5d electrons of gold into the 3d orbitals of iron or, to some extent, into 6s orbital
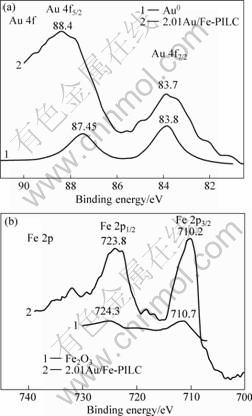
Fig.6 XPS spectra of 2.01Au/Fe-PILC catalyst in Au 4f region(a) and Fe 2p region(b)

Fig.7 XPS spectra of 2.52Au/Fe-oxide catalyst in Au 4f region(a) and Fe 2p region(b)
of gold itself. Thus, the nuclear attraction in gold atoms is increased, resulting in the increase in the binding energies of Au 4f. The outer orbitals of gold atoms with partial charge have electronic configuration similar to those of Pt element, thus making them active for catalysis. Moreover, the binding energies of Fe 2p decrease slightly and this is attributed to the flow of some electrons into some empty Fe 3d orbitals. The coexistence of different gold species in one catalyst is an interesting phenomenon, which could be attributed to the interaction between the gold clusters and the support.
According to the analysis of activity for catalytic combustion of formaldehyde, active species gold was in state of Au3+ and Au0 coexisting in the 2.01Au/Fe-PILC catalyst, while, active species gold were in state of Auδ+ and Au0 coexisting in the 2.52Au/Fe-oxide catalyst. By comparing these two catalysts, the higher activity of 2.01Au/Fe-PILC catalyst demonstrates again that small Au nanoparticles coexisting with a certain number of Au3+ ions are more active for catalytic combustion formaldehyde[14, 22]. It is not difficult to understand that the activity of 2.52Au/Fe-oxide is less than that of 2.01Au/Fe-PILC, even if the content of gold in 2.52Au/Fe-oxide catalyst is more than that of 2.01Au/Fe-PILC catalyst.
4 Conclusions
1) The catalytic activity of 2.01Au/Fe-PILC for formaldehyde catalytic combustion is attributed to nano-sized (2-5 nm), highly dispersed gold particles and good adsorption properties of support.
2) Gold species with partial positive charge are active sites, contributing to the catalytic activity. Furthermore, XPS analysis results show that small Au nanoparticles coexisting with a certain number of Au3+ ions are more active for catalytic combustion formaldehyde than Au nanoparticles coexisting with Auδ+.
References
[1] YANG Xu-zhang, SHEN Yue-nian, YUAN Zang-Fu, ZHU Hai-yong. Ferric ions doped 5A molecular sieves for the oxidation of HCHO with low concentration in the air at moderate temperatures[J]. Journal of Molecular Catalysis A: Chemical, 2005, 237: 224-231.
[2] JIA Mei-lin, SHEN Yue-nian, LI Chang-yan, BAO Zhao-rigetu, SHENG Shi-shan. Effect supports on the gold catalyst activity for catalytic combustion of CO and HCHO[J]. Catalysis Letters, 2005, 99(3/4): 235-239.
[3] de la PE?A O’SHEA V A, ALVAREZ-GALVAN M C, REQUIES J, BARRIO V L, ARIAS P L, CAMBRA J F, G?EMEZ M B, FIERRO J L G. Synergistic effect of Pd in methane combustion PdMnOx/Al2O3 catalysts[J]. Catalysis Communications,2007,8: 1287-1292.
[4] HARUTA M, TSUBOTA S, KOBAYASHI T, KAGEYAMA H, GENET M J, DELMON B. Low-temperature oxidation of CO over gold supported on TiO2, α-Fe2O3, and Co3O4 [J]. Journal of Catalysis,1993, 144: 175-192.
[5] SPINICCI R, FATICANTI M, MARINI P, DE ROSSI S, PORTA P. Catalytic activity of LaMnO3 and LaCoO3 perovskites towards VOCs combustion[J]. Journal of Molecular Catalysis A: Chemical,2003, 197: 147-152.
[6] P?LINK? I, L?Z?R K, KIRICSI I. Cationic mixed pillared layer clays: infrared and M?ssbauercharacteristics of the pillaring agents and pillared structures in Fe, Al and Cr, Al pillared montmorillonites[J]. Journal of Molecular Structure, 1997, 410/411: 547-550
[7] ZHU H Y, ZHU Z H, LU G Q. Controlled doping of transition metal cations in alumina pillared clays[J]. J Phys Chem B, 2000, 104: 5674-5680.
[8] MISHRA T, PARIDA K M, RAO S B. Transition metal oxide pillared clay: 1. A comparative study of textural and acidic properties of Fe(Ⅲ) pillared montmorillonite and pillared acid activated montmorillonite[J]. Journal of Colloid and Interface Science,1996, 183: 176-183.
[9] MINIC? S, SCIR? S, CRISAFULLI C, MAGGIORE R. Catalytic combustion of volatile organic compounds on gold/iron oxide catalysts[J]. Appl Catal B, 2000, 28(3/4): 245-251.
[10] NGUYEN-THANH D, BLOCK K, BANDOSZ T J. Adsorption of hydrogen sulfide on montmorillonites modified with iron[J]. Chemosphere, 2005, 59: 343-353
[11] CAUDO S, CENTI G, GENOVESE C, PERATHONER S. Copper- and iron-pillared clay catalysts for the WHPCO of model and real wastewater streams from olive oil milling production[J]. Applied Catalysis B: Environmental, 2007, 70: 437-446.
[12] LIN H Y, CHEN Y W. Low-temperature CO oxidation on Au/FexOy catalysts[J]. Ind Eng Chem Res, 2005, 44(13): 4569-4576.
[13] LIU C T, SHI P F, ZHANG J X. Preparation of Au/Fe2O3 catalyst and its catalytic performance for selective oxidation of CO in hydrogen-rich gas[J]. Chinese Journal of Catalysis, 2004, 25(9): 697-701.
[14] YAN Wen-fu, PETKOV V, MAHURIN S M, OVERBURY S H, DAI Sheng. Powder XRD analysis and catalysis characterization of ultra-small gold nanoparticles deposited on titania-modified SBA-15[J]. Catalysis Communications, 2005(6): 404-408.
[15] ZHANG Xin, SHI Hui, XU Bo-Qing. Comparative study of Au/ZrO2 catalysts in CO oxidation and 1,3-butadiene hydrogenation[J]. Catalysis Today, 2007, 122: 330-337.
[16] SALVATORE S, SIMONA M, CARMELO C, CRISTINA S, ALESSANDRO P. Catalytic combustion of volatile organic compounds on gold/cerium oxide catalysts[J]. Applied Catalysis B: Environmental, 2003, 40: 43-49.
[17] WAGNER C D, RIGGS W M, DAVIS L E. Handbook of X-Ray photoelectron spectroscopy[M]. USA: Perkin-Elmer Corporation Physical Electronic Division, 1979: 154.
[18] WANG Shu-ping, ZHANG Tong-ying, WANG Xiao-ying, ZHANG Shou-min, WANG Shu-rong, HUANG Wei-ping, WU Shi-hua. Synthesis, characterization and catalytic activity of Au/Ce0.8Zr0.2O2 catalysts for CO oxidation[J]. Journal of Molecular Catalysis A: Chemical, 2007, 272: 45-52.
[19] CENTENO M A, HADJIIVANOV K, VENKOV TZ, KLIMEV HR, ODRIOZOLA J A. Comparative study of Au/Al2O3 and Au/CeO2-Al2O3 catalysts[J]. Journal of Molecular Catalysis A: Chemical , 2006, 252: 142-149
[20] PARK E D, LEE J S. Effects of pretreatment conditions on CO oxidation over supported Au catalysts[J]. J Catal, 1999, 186(1): 1-11.
[21] LI Chang-yan, SHEN Yue-nian, HU Rui-sheng, JIA Mei-lin. The state of active Au species in Au/Fe-O catalytic during CO catalytic oxidation[J]. Chinese Journal of Catalysis, 2006, 27(3): 259-262.
[22] BULUSHEV D A, YURANOV I, SUVOROVA E I, BUFFAT P A, KIWI-MINSKER L. Highly dispersed gold on activated carbon fibers for low-temperature CO oxidation[J]. Journal of Catalysis, 2004, 224: 8-17.
(Edited by YANG Bing)
Foundation item: Projects (20263001, 20601012) supported by the National Natural Science Foundation of China; Project (ND412) supported by Inner Mongolia University Youth Foundation, China
Corresponding author: LI Chang-yan; Tel: +86-13804745744; E-mail: Li.changyan00@163.com