
One-step synthesis of antimony-doped tin dioxide
nanocrystallites and their property
ZHENG Min(֣ ��), WANG Bao(�� ��)
College of Textiles and Clothing Engineering, Suzhou University, Suzhou 215021, China
Received 9 May 2008; accepted 28 August 2008
Abstract: Antimony-doped tin dioxide(ATO) nanoparticles with primary diameter in the range of 9-10 nm were rapidly synthesized via a novel combustion technique, starting with antimony trichloride and tin tetrachloride as metal sources and self-assembly compounds as fuels. The combustion phenomena and characteristics of products were controlled by assembling components in fuel compounds according to appropriate molar ratio. The as-synthesized products were characterized by XRD, SEM, TEM and XPS, respectively. The electrical conductivity was evaluated through measuring the antistatic property of polyester fiber treated by the as-synthesized products. The results show that a mild combustion phenomena without release of smoke can be taken on and perfect azury rutile ATO crystal with complete substitution can be formed rapidly under the appropriate synthetic conditions. The antistatic property of the polyester fiber treated by the as-synthesized ATO products is enhanced remarkably. The triboelectricity voltage below 1.0 kV, half life below 1.0 s and surface resistance below 1.0��106 �� can be attained.
Key words: antimony-doped tin dioxide; combustion; self-assembly; antistatic
1 Introduction
Recently, much attention has been focused on the modification of metal oxide by doping or substituting with special atoms[1-4]. Among these doping systems, antimony-doped tin dioxide(ATO) has attracted considerable attention owing to its potential applications as gas sensor, solar battery, transparent electrode, electricity-conducting coatings and so on[5-6]. Therefore, different methods including solid blend, chemistry co-deposition, sol-gel, metal alkoxide hydrolyzation and hydrothermal technologies have been proposed to prepare ATO[7-12]. Although conventional chemistry co-deposition method is the common method because of its economy and controllability, it is difficult to prepare excellent ATO powder and the technology is very unfriendly to the environment. Other methods cannot be applied widely because they are usually time-consuming, expensive and special equipment is needed usually. For example, high-pressure reactor and long time high-temperature treatment are essential in hydrothermal technology. The major challenges in preparing nano-ATO are the control of the hydrolyzation process and the genuine substitution of Sb5+ for Sn4+ in tin dioxide. It is quite difficult to render substitution reaction at low temperature in short time. While the conventional substituting process at high temperature in long time results in the rapid growth and agglomeration of particles inevitably, which has important negative effects on the characteristics of ATO. Although the hydrothermal technology is reported to prepare perfect and monodisperse ATO nanoparticles, long time heat-treatment (about 12 h) is essential and washing process repeatedly with deionized water is still inevitable[13], which make the technology limited in practical application.
Combustion method is well known as an effective technology for producing a mass of simple and complex inorganic materials having high chemical purity and excellent physical and mechanical properties[14-17]. The reaction based on molecule level assures the complete crystallographic rearrangement, and the ultrashort reaction time (20-30 s) controls the growth of particles. However, there are limited reports about the preparation of substituting system via combustion method and there exist many intricate problems on combustion theory and technology. Furthermore, the poor quality, low product yield and safety in con- ventional combustion reaction also restrict the practical application of the technology.
ZHANG and GAO[18] reported a kind of combustion method called Pechini to prepare nano-ATO. However, the sol-gel process and heat-treating at high temperature (600 ��) for long time (2 h) are still inevitable.
In present work, we report the synthesis of rutile nanometer ATO by a rapid and safe method called self-assembly combustion technique(SAC). The key idea is that, the fuel components are chosen carefully and assembled in an appropriate molar ratio (hereafter termed as��), which ensures the stability of the reaction course and thereby enhances the characteristics and product yield. More importantly, the reaction course is mild and safe without a release of smoke. The corresponding technique has been reported in our patent[19]. This paper focused on the microcosmic and antistatic property of ATO synthesized by SAC technique.
2 Experimental
All reagents were commercially available and used without further purification. In a typical experiment, firstly, SnCl4 was dissolved in distilled water to attain 0.5-2.0 mol/L solutions, and citric acid was added dropwise to the SnCl4 solution until a pH value of 3-4 was reached. The chosen fuels, including ammonium perchorate (AP), ammonium nitride (AN), urea (U) and salvolatile (S), were assembled in an appropriate��, to form self-assembly fuel compounds. Then, a suitable amount of SbCl3 modificated and self-assembly fuel compounds were mixed well with the above SnCl4 solution by stirring to attain a ropy paste (hereafter termed as precursor). This precursor was put into a resistance furnace pre-heated to the given temperature. After going through boiling, evaporating and concentrating, the precursor suddenly foamed up and deflagrated, leaving a very finely azury flocculent powder like sponge.
The crystalline phase was determined by X-ray diffraction (XRD-6000, model D/Max-��B, Shimadzu) instrument with Cu K�� radiation (��=0.15418 nm). The scanning rate of 0.05(?)/s was applied to record the pattern in the 2�� range of 20?-80?. The crystal size was calculated with Scherrer��s equation.
The morphological feature of the products was investigated by scanning electron microscope(SEM) (Japan S-570) and transmission electron microscope (TEM) (JEM��200 CX).
The analysis of element content was done with Thermo ESCALAB 250 XPS instrument (America) using Al K�� radiation (h��=1 486.6 eV).
The antistatic property of the as-synthesized sample was estimated according to the following approach. ATO aqueous dispersion was prepared by adding 0.2-1.0 g of the as-synthesized sample and 0.2 g of polymer surfactant polyvinylpyrrolidone(PVP) into 100 mL of distilled water with pH value adjusted to 8.5, then keeping stirred at 60 �� for 30 min. 5.0 g of polyester fiber was washed before use for several times in acetone for 20 min using an ultrasonic bath to remove surface impurities. After being dried, it was impregnated for 30 min in a treatment solution containing the ATO aqueous dispersion, 0.5 g binder agent and 1.0 g silicone softener, then was padded twice (take-up 70%) on a laboratory padding mangle. After they were padded, the polyester fiber was immediately dried at 100 �� for 2.0 min and cured at 160 �� for 1.0 min. The triboelectricity voltage, half life and surface resistance of the treated polyester were measured by Static Honestmeter S-5109 (environment pressure 5 kV) and Surface Resistance Instrument SIMCO.
3 Results and discussion
3.1 Effect of fuel
ATO powder can be synthesized by combustion reaction using single fuel and fuel compounds. However, it is noted that the nature of combustion and the appearance of the as-synthesized samples are completely different with the different fuels. Using single fuel, such as ammonium perchorate(AP), ammonium nitride(AN) and urea(U), results in rough powder including yellow and navy blue sheet substances, companied with dazzling firelight and amounts of light-brown smoke during the initial period of combustion reaction. Whereas using fuel compounds assembled according to appropriate molar ratio as fuel, perfect azury ATO powder like flocculent can be attained and controllable combustion reaction course is presented, going through a series of phenomena such as boiling, evaporating and concentrating, foaming up and deflagrating. Fig.1 demonstrates two typical appearances of the as-synthesized samples using ammonium perchorate and fuel compounds (�� value 4/2/3/1) as fuels respectively at igniting temperature of 500 ��, which were screened by digital camera.

Fig.1 Typical appearances of as-synthesized samples using ammonium perchorate (a) and fuel compounds (�� value 4/2/3/1) (b) as fuels
3.2 Effect of mass ratio of fuel compounds to tin tetrachlorine
The mass ratio of fuel compounds to tin tetrachlorine plays an important role in controlling the morphology and particle size of the as-synthesized powder. Fig.2 displays the typical XRD patterns of the as-synthesized samples under different mass ratio of fuel compounds to tin tetrachlorine.
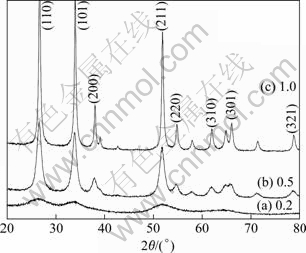
Fig.2 XRD patterns of samples prepared under different mass ratio of fuel compounds to tin tetrachlorine
In Fig.2(a), the diffraction peaks are very weak and wide, which indicates that the sample synthesized under
low mass ratio (0.2) is amorphous. When the mass ratio is increased to 0.5, the strong diffraction peaks in Fig.2(b) can be indexed as (110), (101) and (211) planes of a standard tetragonal phase rutile tin dioxide crystal (JCPDS21-1250) and the other planes such as (200), (220), (310) and (301) are also visible. The strong and sharp reflection peaks suggest that the samples prepared are well crystallized. There are no other crystalline phases such as Sb2O3 and Sb2O5 in the X-ray diffraction pattern, which indicates the even and complete antimony-doping in SnO2. Antimony ions bring into the crystal lattice of tin dioxide to substitute for tin ions and don��t bring about a new object phase, which is consistent with the traditional doping theory. The broadening of the diffraction peaks is due to the small size domino effect. The crystalline size is calculated to be 9.24 nm according to Scherrer��s equation. A further increase in mass ratio results in sharper and stronger diffraction peaks, and the corresponding primary crystalline size is calculated to be 26.06 nm (seen in Fig.2(c)).
The difference of diffraction peaks and crystalline size under different mass ratio of fuel compounds to tin tetrachlorine can be attributed to the energy released by fuel compounds during combustion. When the mass ratio is lower (0.2), the energy evolved is not enough and the local temperature remains low, causing incomplete substitution reaction and low crystallization agree. However, too much fuel compounds (mass ratio is 1.0) will result in high combustion temperature, which is prone to promote the crystallite growing and the active primary particles sintering, although high combustion temperature is in favor of substitution of antimony ions for tin ions in tin dioxide crystal lattice. Furthermore, the combustion reaction degree is too violent to be controlled under higher mass ratio and a majority of product is carried away following the gases liberated during reaction. The practical experimental results show that the samples with well degree of crystallization and small size are obtained when the mass ratio of fuel compounds to tin tetrachlorine is 0.5, following with a controllable reaction phenomena and high product yield (above 90%).
3.3 Effect of environment temperature
During the combustion process, the amount of energy afforded by the compound fuels decomposing is not enough to initiate ignition. Therefore, sufficient heat should be supplied from external heating source. In our study, the external temperature to initiate ignition is from 400 �� to 700 ��. Fig.3 shows the typical SEM photographs of the as-synthesized samples prepared at the temperature from 400 �� to 700 ��. The microstructure of the sample synthesized at 400 �� is entirely composed of porous shards with agglomerated fine particles (Fig.3(a)). When the temperature is increased to 500 ��, the sample takes on regular spherecity and the size is very small and uniform ranging from 40 nm to 50 nm (Fig.3(b)), which indicates that the as-synthesized ATO crystal grains are well-dispersed. A further increase in temperature results in larger crystalline size and obvious conglomeration (seen in Figs.3(c) and (d)). The difference of morphology and size at different temperatures shows that environment temperature above 500 �� results in crystal growth and agglomeration. Whereas lower environment temperature below 500 �� cannot afford enough energy to assure complete substitution reaction. Therefore, the appropriate environment temperature should be 500�� to assure that product has perfect morphology and high purity.

Fig.3 SEM images of samples prepared under different external temperatures: (a) 400 ��; (b) 500 ��; (C) 600 �� (d) 700 ��
Fig.4(a) shows the complete XPS spectrum of antimony-doped tin dioxide(ATO) nanocrystals synthesized under the optimum technology condition. In Fig.4(a), the peaks corresponding to C (contamination), Sn, Sb and O can be observed. Fig.4(b) shows the XPS spectra of O 1s and Sb 3d transition region obtained between 520 eV and 544 eV. The Sb transition(3d3/2) has been employed to obtain the oxidation state of antimony. The peak (centered at 540.0 eV) can be separated into two contributions: one corresponds to antimony with an oxidation state of Sb(��), while the other corresponds to Sb(��). The analysis results for the XPS spectrum areas show that the relative amount of Sb in ATO nanocrystals is 1.99%.
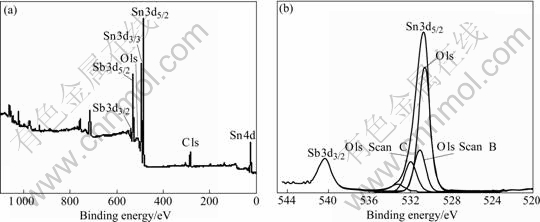
Fig.4 Complete XPS spectrum (a) of ATO nanocrystals synthesized under optimum technology condition and XPS spectrum of O 1s and Sb 3d (b)
Fig.5 displays the TEM morphology of antimony- doped tin dioxide(ATO) nanocrystals synthesized under the optimum technology condition. The sample in Fig.5 takes on regular spherical morphology and well- dispersed property. The mean diameter is about 50 nm, which agrees well with the result obtained from scanning electron microscope.

Fig.5 TEM photograph of sample prepared under optimum technology condition
3.4 Antistatic property of ATO nanocrystallite on polyester fiber
The antistatic property of the sample prepared under the optimum technology condition is estimated through measuring the triboelectricity voltage, half life and surface resistance of the polyester fiber treated by the corresponding sample with different concentrations. The results are listed in Table 1.
Table 1 Antistatic properties of polyester fiber treated by as-synthesized samples with different concentrations
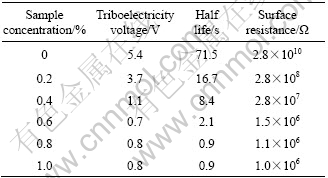
The results show that the triboelectricity voltage, half life and surface resistance decrease remarkably compared with those of the untreated polyester fiber. The lowest triboelectricity voltage, half life and surface resistance of the former are 0.7 kV, 0.9 s and 1.0��106, respectively, whereas the latter are 5.4 kV, 71.5 s and 2.8 ��1010. The high antistatic properties of the polyester fibre reveal the eminent conductive property of the as-synthesized sample.
4 Conclusions
1) Antimony-doped tin dioxide(ATO) nano- crystallines were successfully synthesized by a novel self-assembly combustion technique in short time. The mass ratio of components in fuel compounds was 4/2/3/1 and the mass ratio of fuel compounds to the tin chlorine was 0.5. The environment temperature schedule was 500 �� to initiate ignition.
2) The primary diameter of the as-synthesized ATO was 9-10 nm and the ATO took on fully crystalline cubic phase. The formation of the ATO with small size and even distribution was contributed to the integrative action of different fuels in the self-assembly fuel compounds.
3) The polyester fiber treated by the as-synthesized sample under the optimum technology condition obtained triboelectricity voltage of 0.7 kV and half life of 0.9 s, which revealed the eminent conductive property of the as-synthesized sample.
References
[1] KIM D W, KIM D S, KIM Y G, KIM Y C, KIM S G. Preparation of hard agglomerates free and weakly agglomerated antimony doped tin oxide(ATO) nanoparticles by coprecipitation reaction in methanol reaction medium [J]. Material Chemistry Physics, 2006, 97(2/3): 452-457.
[2] ZHANG X J, LIANG H B, GAN F X. Novel anion exchange method for exact antimony doping control of stannic oxide nanocrystal powder [J]. Journal of America Chemistry Society, 2006, 89(3): 792-798.
[3] DOUY A. Polyacrylamide gel: An efficient tool for easy synthesis of muticomponent oxide precursors of ceramics and glass [J]. International Journal of Inorganic Materials, 2001, 3(7): 699-702.
[4] GUO Yu-zhong, WANG Jian-hua, HUANG Rui-an. Electrical and optical properties transparent and conductive Sb-doped SnO2 films [J]. Journal of Inorganic Materials, 2002, 17(1): 131-138. (in Chinese)
[5] MARCEL C, HEGDE M S. Electrochromic properties of antimony tin oxide(ATO) thin films synthesized by pulsed laser deposition [J]. Electrochemica Acta, 2001, 46(23): 2097-2104.
[6] JAIN G, KUMAR R. Electrical and optical properties of tin oxide and antimony doped tin oxide film [J]. Optical Materials, 2004, 26(1): 27-31.
[7] ZHANG Jian-rong, GAO Lian. Hydrothermal synthesis of antimony- doped tin oxide(ATO) nanoparticles and electrical properties [J]. Acta Chimica Sinica, 2003, 61(10): 1679-1681.
[8] LI Qin-shan, ZHANG Jin-zhao, SONG Li. Influence of heat-treatment on the particle size and properties of conductive nanorneter antimony-doped tin dioxide powders [J]. Journal of Inorganic Materials, 2002, 17(3): 490-495. (in Chinese)
[9] MIAO Hong-yan, DING Chang-sheng, LUO Hong-jie. Antimony-doped tin dioxide nanometer powders prepared by the hydrothermal method [J]. Microelectronic Engineering, 2003, 66: 142�C146.
[10] JIANG Ming-xi, YANG Tian-zu, GU Ying-ying, DU Zuo-juan, LIU Jian-ling. Preparation of antimony doped nanoparticles by hydrothermal method [J]. Trans Nonferrous Met Soc China, 2005, 15(3): 702-705.
[11] BAI Fan-fei, HE Yun, HE Ping, TANG Yi-wen, JIA Zhi-jie. One-step synthesis of monodispersed antimony-doped tin oxide suspension [J]. Materials Letters, 2006, 60: 3126-3129.
[12] YANG Hua-ming, HU Yue-hua, QIU Guan-zhou. Preparation of antimony-doped SnO2 nanocrystallites [J]. Material Research Bulletin, 2002, 37(15): 2453-2458.
[13] LI Li-li, MAO Li-ming, DUAN Xue-chen. Solvothermal synthesis and characterization of Sb-doped SnO2 nanoparticles used as transparent conductive films [J]. Material Research Bulletin, 2006, 41: 541-546.
[14] LI Wen-xia, YIN Sheng, WANG Hui. Micro powders of multiphase PSZ by low temperature combustion synthesis [J]. Journal of the Chinese Ceramic Society, 2000, 28(6): 534-537. (in Chinese)
[15] HWANG C C. WU T Y. Synthesis and characterization of nanocrystalline ZnO powders by a novel combustion synthesis method [J]. Material Science Engineering B, 2004, 111(2/3): 197-206.
[16] PUROHIT R D, SHARMA B P, PATIL K T. Ultrafine ceria powders via glycine-nitrate combustion [J]. Material Research Bulletin, 2001, 36(15): 2711-2721.
[17] DONG Y C, EIL H L. Microwave-induced combustion synthesis of Ce1-xSmxO2-x/2 powder and its characterization [J]. Journal of Alloys Compound, 2004, 374(1/2): 69-73.
[18] ZHANG Jian-rong, GAO Lian. Synthesis of antimony-doped tin oxide(ATO) nanoparticles by the nitrate-citrate combustion method [J]. Material Research Bulletin, 2004, 39: 2249-2255.
[19] ZHENG Min, ZHU Ya-wei. Rapid synthesis of antimony-doped tin dioxide nanoparticles. China patent 200710020429 [P]. 2007. (in Chinese)
Foundation item: Project(08KJD430006) supported by Natural Science Foundation of Jiangsu Province University
Corresponding author: ZHENG Min; Tel: +86-13915571419; +86-512-68998646; E-mail: zhengmin@suda.edu.cn
DOI: 10.1016/S1003-6326(08)60286-1
(Edited by YUAN Sai-qian)