Trans. Nonferrous Met. Soc. China 24(2014) 1905-1911
Influence of Phanerochaete chrysosporium on degradation and preg-robbing capacity of activated carbon
Qian LIU, Hong-ying YANG, Lin-lin TONG
School of Materials and Metallurgy, Northeastern University, Shenyang 110819, China
Received 9 June 2013; accepted 6 September 2013
Abstract: The effect of Phanerochaete chrysosporium on degradation and preg-robbing capacity of activated carbon, which was used as a substitute of carbonaceous matter in carbonaceous gold ores, was studied. After 14 d treatment with Phanerochaete chrysosporium, the degradation rate of activated carbon reached 27.59%. The XRD and FTIR analyses indicate that Phanerochaete chrysosporium can distort the micro-crystalline structure of activated carbon, increase the number of oxygen-containing groups and aliphatics and make the aromatic structures be oxidized and exfoliated. The gold-adsorption tests show that Phanerochaete chrysosporium can reduce the preg-robbing capacity of activated carbon by 12.88%. This indicates that Phanerochaete chrysosporium is an available microorganism, and it can be employed to reduce the preg-robbing capacity of carbonaceous matter and improve the gold leaching rate. The combined effect of passivation, alkalization and oxidation of biological enzymes-free radicals of Phanerochaete chrysosporium on carbonaceous matter was also discussed.
Key words: Phanerochaete chrysosporium; activated carbon; degradation; carbonaceous gold ores; preg-robbing capacity
1 Introduction
The carbonaceous gold ores are refractory since dissolved gold is robbed by carbonaceous matter in the cyaniding process, and this phenomenon is termed as preg-robbing [1-3]. A large number of carbonaceous gold ores have been discovered around the world, which distribute mainly in Navada of America and southwest of China [4,5]. With the increasing exhaustion of easy-leached gold ores, carbonaceous refractory gold ores have become the main minerals for extracting gold.
At present, the pretreatment methods of carbonaceous gold ores mainly include high-temperature roasting, bio-oxidation, chemical oxidation, competitive adsorption, barrier inhibition, microwave roasting, and so on. Bio-oxidation method has been paid more and more attention recently because of its advantages such as mild conditions, simple process, low energy consumption, selective oxidation and environment-friendly. BRIERLEY and KULPA [6,7] firstly oxidized sulfides in gold ores with Thiobacillus ferrooxidans, and made the gold extraction rate increase from 0 to 55.50%. Afterwards, they deactivated carbonaceous matter using microbial consortium which is comprised of Pseudomonas maltophilia, Pseudomonas oryzihabitans, Achromobacter species and Arthrobacter species, and made the leaching rate of gold increase from 55.50% to 74.40%. However, the addition of hydrocarbons such as kerosene and hexadecane could result in the difficulty of solid-liquid separation after pretreatment, and the formed organic membrane could disturb the gold leaching. YEN et al [8] used Trametes versicolor to pre-treat refractory gold ores. Firstly, Trametes versicolor culture medium (with the fungal agent) was used to deactivate the preg-robbing carbonaceous components, and the gold extraction rate was 54.10%-64.50%. Secondly, the refractory sulfides of the gold ores were decomposed by Trametes versicolor culture medium (without the fungal agent). The gold extraction rate increased to 87.00%-95.25% when the ores samples were treated by a combination of bio-treatment and bio-oxidation with Trametes versicolor. AMANKWAH et al [9] treated double refractory gold concentrates with two-stage bio-oxidation. The chemolithotrophic bacteria were used to oxidize sulfides firstly, and 81.10% of gold extraction rate occurred during cyanidation. Secondly, the carbonaceous matter was destroyed by Streptomyces setonii, and the cyanide leaching rate of gold was increased from 81.10% to 94.70%.
Recently, some researchers have studied the influence of fungi and bacteria on the preg-robbing capacity of lignite, sub-bituminous coal, bituminous coal and anthracite which were used as substitute of carbonaceous matter, and obtained good research results [10,11]. Carbonaceous matter is mainly composed of elemental carbon, organic carbon and hydrocarbons. Among them, the most important carbonaceous preg-robber of gold is elemental carbon which is a kind of natural activated carbon between anthracite and graphite. The elemental carbon has been discovered in almost all carbonaceous gold ores, and its preg-robbing behavior is similar with activated carbon [12]. Thus, the activated carbon, chosen as substitute of carbonaceous matter in carbonaceous gold ores, was used to study the capacity of Phanerochaete chrysosporium on degrading and reducing preg-robbing of carbonaceous matter.
2 Experimental
2.1 Materials
The nut shell activated carbon, a kind of wooden activated carbon, was chosen as the experimental material in this study. Its moisture content was 3.40%, ash content was 3.10% and apparent density was 0.53 g/mL. In addition, the granularity distribution of the milled activated carbon is listed in Table 1. The contents of main chemical elements are listed in Table 2. The carbon accounted for 84.70% in activated carbon. The X-ray diffraction (XRD) spectrum of activated carbon is shown in Fig. 1. Two broad diffraction peaks are observed around 24° and 44°. The C002peak is the strongest diffraction peak, and the C100 peak is the second one. Generally, the stacking situation of aromatic rings was characterized by C002 peak, and the size of aromatic rings was obtained by analyzing the C100peak [13-16].
Table 1 Particle size distribution of activated carbon

Table 2 Content of main chemical elements in activated carbon (mass fraction, %)

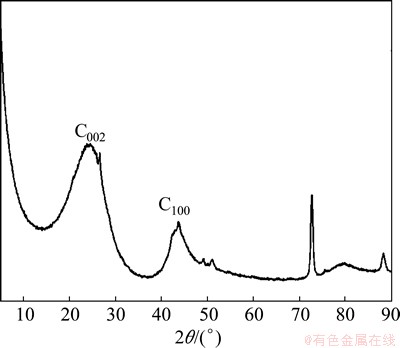
Fig. 1 XRD pattern of activated carbon
2.2 Strain and culture medium
Phanerochaete chrysosporium, model strain of White-rot fungi, was chosen as the experimental strain in this study. The nitrogen limited culture medium was used since excessive nitrogen source could inhibit the formation of ligninolytic enzymes system in Phanerochaete chrysosporium [17,18].
2.3 Determination of degradation rate
Under the condition of 30 °C, 150 r/min shake culture for 14 d, the sample was filtered to obtain brown filtrate, and a lot of flocculent precipitates were produced in filtrate when it was adjusted to pH>12. As for the residue from activated carbon, it was washed with distilled water to remove the medium. The fungal degradation residue and water soluble alkaline precipitate were dried, weighed and the degradation rate (η) was calculated by [13]:
η=(m0-m1)/m0×100% (1)
where m0 is the initial mass of activated carbon; m1 is the mass of fungal degradation residue.
2.4 Analyses of XRD, FTIR and SEM
Activated carbon is a typical amorphous material, but it has crystallite structure on the basis of graphite layers, and only its orderliness is worse than graphite. Crystallite is stacked of some aromatic rings layers which are composed of aromatic nuclei, branched-chain hydrocarbons and various functional groups. The difference of crystallite structural parameters (d, La and Lc) can quantitatively characterize micro-structural change of activated carbon [14,19]. The phase analyses of samples were carried out on an X-ray diffractometer (PW3040/60). The infrared analyses of samples were performed using an Fourier transform infrared spectrometer (VECTOR-22, Bruker). The surface morphologies of samples were observed by a scanning electron microscope (SEM-SSX-550).
2.5 Gold-adsorption experiment
Gold-bearing cyanide solution (17 μg/mL) was used for testing the gold adsorption capacity of activated carbon, fungal degradation residue and water soluble alkaline precipitate at pH=12. Three kinds of samples were weighed 1 g respectively, and then put in 100 mL gold solution at 150 r/min shake culture for 24 h. At the end of the experiment the residual solution was filtered, and the content of gold was measured in the filtrate. The change of preg-robbing capacity of activated carbon could be evaluated by the different gold concentrations in cyanide solutions before and after treatment with Phanerochaete chrysosporium.
3 Results and discussion
3.1 Bio-degradation of Phanerochaete chrysosporium on activated carbon
Phanerochaete chrysosporium can make the macromolecular structures of activated carbon depolymerize into small molecular soluble substances by its own metabolic activity. After 14 d incubation with Phanerochaete chrysosporium, the degradation rate of activated carbon reached 27.59% as shown in Table 3, and the carbon content of activated carbon was reduced from 84.70% to 77.60%. This may be attributed to the following reasons: 1) some components of activated carbon are transformed to soluble ingredients into solution; 2) some carbon is metabolized since Phanerochaete chrysosporium is a heterotroph. Moreover, the sulfur content was reduced from 0.84% to 0.27%, which indicated that Phanerochaete chrysosporium could partly remove sulfur of activated carbon [13,20]. The water soluble alkaline precipitate was filtrated and dried in a kind of dark brown solid which is similar to activated carbon. The results indicated that the dark brown solid is insoluble in alcohol but soluble in hydrochloric acid. The chemical analysis of dark brown solid showed that the carbon content is 79.20%, sulfur content is 4.80% and there are other elements such as Ca, Mg, Si, Fe and Na.
Table 3 Bio-degradation rate of activated carbon
3.2 Effect of Phanerochaete chrysosporium on microcrystal structure of activated carbon
Figure 2 shows the XRD patterns of the activated carbon (sample 1), fungal degradation residue (sample 2) and water soluble alkaline precipitate (sample 3). Compared with the diffraction peaks of sample 1. The peak positions of samples 2 and 3 were shifted to low-angle region, which indicated that the microcrystalline structure of activated carbon was distorted. The La (0.578 nm) of sample 3 was lower than that of sample 1 (0.941 nm), which showed that the diameter of aromatic layers of sample 3 was reduced due to the decrease of the number of aromatic rings. The Lc values of samples 2 and 3 were 0.763 nm and 0.378 nm respectively, which were much less compared with sample 1 (1.142 nm). This indicated that the stacking height and the number of aromatic layers were decreased while the structure of some macromolecules was destroyed in activated carbon. Compared with the interlayer spacing d002(0.347 nm) of sample 1, the d002(0.363 nm) of sample 2 and d002(0.423 nm) of sample 3 slightly increased, which showed that Phanerochaete chrysosporium could increase the interlayer distance of microcrystal layers and reduce the condensation degree of aromatic nuclei in samples 2 and 3. This change is consistent with that of Lc.
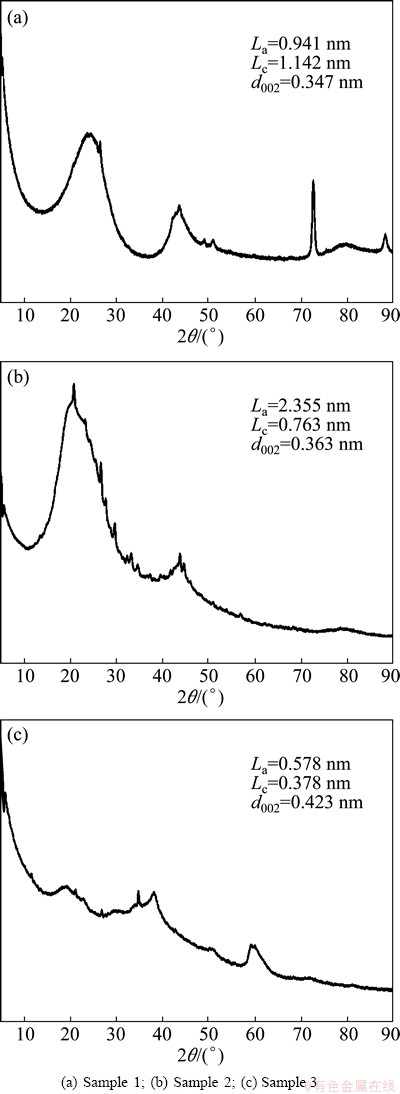
Fig. 2 XRD patterns of samples
3.3 Effect of Phanerochaete chrysosporium on functional groups of activated carbon
According to the Lambert-Beer law, the relative intensity of the FTIR spectrum peaks can partly reflect the concentration of included substances and functional groups in qualitative analysis [21]. The peak shape and trend of FTIR spectra of samples 1-3 were basically same as shown in Fig. 3, which indicated that Phanerochaete chrysosporium had little effect on the structure and compositional distribution of functional groups in activated carbon. The difference of three samples was mainly focused on the peak and their intensity of some functional groups [21-26].
A broadening peak was observed at 3350 cm-1 in sample 2, which should be attributed to the stretching vibration of associated―OH in phenol, alcohol, carboxylic acid and peroxides due to the low nitrogen content of activated carbon, but this peak was not found in samples 1 and 3. Compared with the samples 1 and 3, the stretching vibration peak of C=O appeared at 1650 cm-1 in sample 2. The increase of oxygen-containing groups, such as ―OH and C=O, made more reaction sites of activated carbon, which could promote the degradation of Phanerochaete chrysosporium on activated carbon.
The stretching vibration peaks of aromatic and aliphatic C―H were located in the regions of 3000-3100 cm-1and 2800-3000 cm-1, respectively. FTIR spectrum of sample 1 presented two weak stretching vibration peaks at 3010 cm-1and 2890 cm-1, which illustrated that the organic structure of sample 1 contained a certain amount of aromatic and aliphatic structures [27]. Aromatic C―H stretching vibration of sample 2 disappeared, but a strong stretching vibration peak of aliphatic C―H appeared at 2920 cm-1. These indicated that Phanerochaete chrysosporium could significantly decrease the content of aromatic structures and increase the number of aliphatic structures of activated carbon.
Compared with sample 1, the relative intensity of the kerogen O―C―O group at 1550 cm-1diminished drastically in sample 2, which proved that Phanerochaete chrysosporium could reduce the metamorphic grade of activated carbon and the condensation degree of aromatic rings by metabolic activity.
The stretching vibration peak of S―S was observed at 600 cm-1 in the FTIR spectrum of sample 3, which indicated that Phanerochaete chrysosporium could remove the sulfur of activated carbon. The phenomenon is consistent with the decrease of sulfur content.

Fig. 3 FTIR spectra of sample 1 (a), sample 2 (b) and sample 3 (c)
3.4 Influence of Phanerochaete chrysosporium on preg-robbing capacity of activated carbon
The carbonaceous materials, such as activated carbon, lignite and bituminous coal, have gold- adsorption capacity. The gold-adsorption experiment could reflect the effect of Phanerochaete chrysosporium on the preg-robbing capacity of activated carbon [10]. The research showed that samples 1-3 could adsorb aurocyanide, but the difference of gold-adsorption capacity occurred obviously as shown in Table 4. The gold adsorption rates of samples 1-3 were 99.35%, 86.47% and 84.29%, respectively, and the preg-robbing capacity of activated carbon was decreased by 12.88%, which indicated that Phanerochaete chrysosporium can reduce the gold-adsorption capacity of activated carbon.
Table 4 Preg-robbing capacity of samples 1-3
4 Effect mechanism of Phanerochaete chrysosporium on activated carbon
Presently, the effect mechanism of Phanerochaete chrysosporium on carbonaceous matter includes mainly two ways: passivation mechanism and bio-degradation mechanism. The gold adsorption of graphite-like carbonaceous matter mainly depends on the activity sites related to adsorption because its maturity is similar to the anthracite [10]. The SEM analysis indicated that the untreated activated carbon with extensive micropores (20-40 μm) presented strong activity and adsorption capacity as shown in Fig. 4(a). In the leaching system with Phanerochaete chrysosporium, mycelium and metabolites could adsorb and cover on the surface of activated carbon to reduce preg-robbing capacity by changing physicochemical properties of minerals as shown in Fig. 4(b). After 14 d incubation with Phanerochaete chrysosporium, the pH value of culture medium increased from 4 to 8-9, which indicated that Phanerochaete chrysosporium could produce and secrete nitrogenous bases such as polypeptide and polyamine which could involve in the degradation of carbonaceous matter by increasing pH of the reaction system [28]. The results of XRD showed that Phanerochaete chrysosporium could damage the macromolecular structures of activated carbon as presented in Fig. 2, which was probably related to the chain reactions based on the free radicals by catalysis of the lignin degradation enzymes system, and the specific mechanism is shown in Fig. 5 [10,29]. Firstly, the bio-enzymes-P(Fe3+) loses two electrons (2e) to form positive ions free radicals P(O=Fe4+・) by the reaction with hydrogen peroxide (H2O2). Secondly, P(O=Fe4+・) reacts with a substrate molecule RH, which results in electron transfer from RH to P(O=Fe4+・), thus free radicals R・ and P(O=Fe4+) forms. Finally, P(O=Fe4+) returns to the initial state by reaction with another molecule RH. These reactions produce massive free radicals which can reduce the preg-robbing capacity by destroying the structure of carbonaceous matter. The adsorption characteristic of activated carbon depends on the pore structure and the surface chemical groups. FTIR analyses showed that Phanerochaete chrysosporium could increase the number of oxygen-containing functional groups as shown in Fig. 3, which could reduce the gold adsorption capacity of activated carbon [11]. The function of Phanerochaete chrysosporium on carbonaceous matter is the result of combined effect of passivation, alkalization and oxidation of biological enzymes-free radicals.
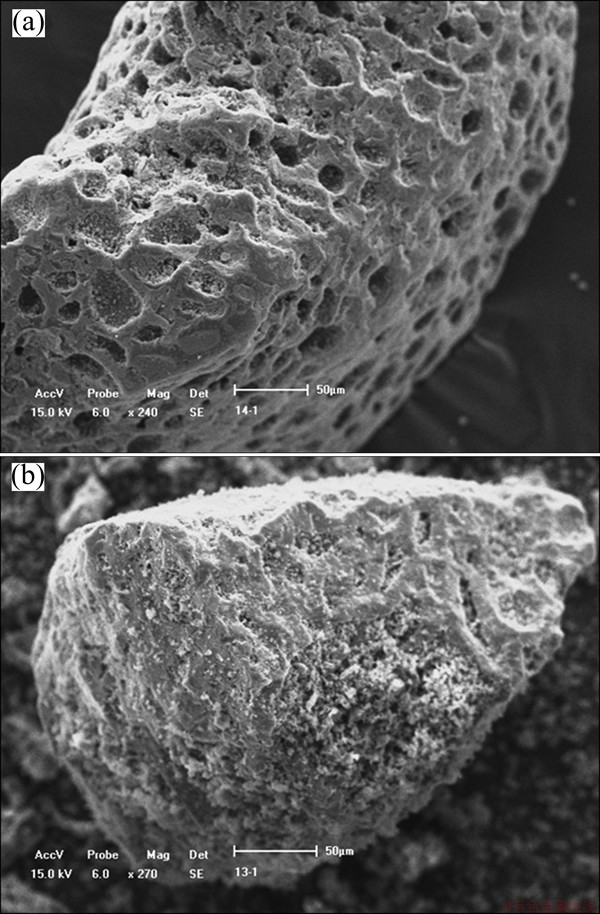
Fig. 4 SEM images showing surface morphologies of activated carbon before (a) and after (b) treatment with Phanerochaete chrysosporium

Fig. 5 Effect mechanism of biological enzymes on carbonaceous matter
5 Conclusions
1) Phanerochaete chrysosporium is an available microorganism to degrade activated carbon. Phanerochaete chrysosporium can not only degrade carbonaceous matter, but also partly remove sulfur in the carbonaceous matter.
2) Phanerochaete chrysosporium can effectively inhibit the preg-robbing capacity of carbonaceous matter by destroying the structure and passivating the active sites for gold adsorption in carbonaceous gold ores.
3) Phanerochaete chrysosporium can destroy the microcrystalline and macromolecular structures, which increases the number of aliphatic structures and oxygen-containing groups, and decreases the content of aromatic structures and the condensation degree of aromatic nucleus.
4) The effect mechanism of Phanerochaete chrysosporium on carbonaceous matter is the combined action of passivation, alkalization and oxidation of biological enzymes-free radicals.
References
[1] ABOTSI G M K, OSSEO-ASARE K. Surface chemistry of carbonaceous ores I. Characterization of the carbonaceous matter and adsorption behavior in aurocyanide solution [J]. International Journal of Mineral Processing, 1986, 18(3-4): 217-236.
[2] REES K L, van DEVENTER J S J. Preg-robbing phenomena in the cyanidation of sulphide gold ores [J]. Hydrometallurgy, 2000, 58(1): 61-80.
[3] HAUSEN D M, BUCKNAM C H. Study of preg-robbing in the cyanidation of carbonaceous gold ores from Carlin, Nevada [C]// Applied Mineralogy: Proceedings of the 2nd International Congress on Applied Mineralogy in the Minerals Industry. Los Angeles, California, USA: Metallurgical Society of AIME, 1985: 833-856.
[4] FANG Zhao-heng. Mineral characteristic and extractive technology of carbonaceous gold ores [J]. Gold Science and Technology, 2003, 11(6): 28-35. (in Chinese)
[5] LUO Zhen-kuan, MIAO Lai-cheng, GUAN Kang, CHEN Ge. Geotectonic evolution and Au metallogeny in China [J]. Contributions to Geology and Mineral Resources Research, 1998, 13(3): 24-34. (in Chinese)
[6] BRIERLEY J A, KULPA C F. Microbial deactivation of preg-robbing carbon in gold ore [C]//Bio-hydrometallurgical Technologies, vol 1: Proceedings of an International Bio-hydrometallurgical Symposium. Jackson Hole, Wyoming, USA: Minerals, Metals & Materials Society, 1993: 427-435.
[7] BRIERLEY J A, KULPA C F. Microbial consortium treatment of refractory precious metal ores: US 5127942 [P]. 1992-07-07.
[8] YEN W T, AMANKWAH R K, CHOI Y. Microbial pre-treatment of double refractory gold ores: US 20090158893 Al [P]. 2009-06-25.
[9] AMANKWAH R K, YEN W T, RAMSAY J A. A two-stage bacterial pretreatment process for double refractory gold ore [J]. Minerals Engineering, 2005, 18(1): 103-108.
[10] OFORI-SARPONG G, TIEN M, OSSEO-ASARE K. Myco- hydrometallurgy: Coal model for potential reduction of preg-robbing capacity of carbonaceous gold ores using the fungus, Phanerochaete chrysosporium [J]. Hydrometallurgy, 2010, 102(1-4): 66-72.
[11] AMANKWAH R K, YEN W T. Effect of carbonaceous characteristics on biodegradation and preg-robbing behavior [C]//Proceeding of the 23rd International Mineral Processing Congress. Istanbul, Turkey: Istanbul Technical University, 2006: 1445-1451.
[12] STENEBRATEN J F, JOHNSON W P, BROSNAHAN D R. Characterization of goldstrike ore carbonaceous material part 1: Chemical Characteristics [J]. Minerals and Metallurgical Processing, 1999, 16(3): 37-43.
[13] WANG Long-gui. Microbial conversion and utilization of coal [M]. Beijing: Chemical Industry Press, 2006. (in Chinese)
[14] TIAN Cheng-sheng, ZENG Fan-gui. Analysis of structure between vitrain and fusain X-ray diffraction analysis [J]. Journal of Taiyuan University of Technology, 2001, 32(2): 102-105. (in Chinese)
[15] SHARMA A, KYOTANI T, TOMITA A. Comparison of structural parameters of PF carbon from XRD and HRTEM techniques [J]. Carbon, 2000, 38(14): 1977-1984.
[16] ASO H, MATSUOKA K, SHARMA A, TOMITA A. Structural analysis of PVC and PFA carbons prepared at 500-1000 °C based on elemental composition, XRD, and HRTEM [J]. Carbon, 2004, 42(14): 2963-2973.
[17] SATO S, LIU F, KOC H, TIEN M. Expression analysis of extracellular proteins from Phanerochaete chrysosporium grown on different liquid and solid substrates [J]. Microbiology, 2007, 153(9): 3023-3033.
[18] GARG S K, MODI D R. Decolorization of pulp-paper mill effulent by white-rot fungi [J]. Critical Reviews in Biotechnology, 1999, 19(2): 85-112.
[19] WANG Qing-bo, ZHU Wen-kai, QU Jing-yuan, CHENG Jin-xing, ZHOU Bai-chang. Structural studies of some activated carbons with different radon adsorption ability by X-ray diffraction techniques [J]. Nuclear Electronics and Detection Technology, 2010, 30(10): 1279-1282. (in Chinese)
[20] XU Fu-ming, YANG Ling-xiao, ZHOU Shen-fan, TANG Wan-ying. Study on coal desulfurization by white rot fungus [J]. Journal of China Coal Society, 1999, 24(4): 424-428. (in Chinese)
[21] LI Qing-zhao, LIN Bai-quan, ZHAO Chang-sui, WU Wei-fang. Chemical structure analysis of coal char surface based on Fourier-transform infrared spectrometer [J]. Proceedings of the Chinese Society for Electrical Engineering, 2011, 31(32): 46-52. (in Chinese)
[22] SUN Xu-guang, CHEN Jian-ping, HAO Duo-hu. Micro-FTIR spectroscopy of macerals in coals from the Tarim basin [J]. Acta Scientiarum Naturalium Universitatis Pekinensis, 2001, 37(6): 832-838. (in Chinese)
[23] YANG Hai-ping, YAN Rong, CHEN Han-ping, LEE D H, ZHENG Chu-guang. Characteristics of hemicellulose, cellulose and lignin pyrolysis [J]. Fuel, 2007, 86(12-13): 1781-1788.
[24] GUO Ying-ting, BUSTIN R M. Micro-FTIR spectroscopy of liptinite macerals in coal [J]. International Journal of Coal Geology, 1998, 36(3-4): 259-275.
[25] PETERSEN H I, ROSENBERG P, NYTOFT H P. Oxygen groups in coals and alginite-rich kerogen revisited [J]. International Journal of Coal Geology, 2008, 74(2): 93-113.
[26] DROBNIAK A, MASTALERZ M. Chemical evolution of miocene wood: Example from the Belchatow brown coal deposit, central Poland [J]. International Journal of Coal Geology, 2006, 66(3): 157-178.
[27] ZHANG Ke, YAO Su-ping, HU Wen-xuan, FANG Hong-feng. Analysis on infrared spectra characteristic of coal and discussion of coalification mechanism [J]. Coal Geology and Exploration, 2009, 37(6): 8-13. (in Chinese)
[28] STRANDBERG G W, LEWIS S N. Factors affecting coal solubilization by the bacterium Streptomyces setonii 75Vi2 and by alkaline buffers [J]. Applied Biochemistry and Biotechnology, 1988, 18(1): 355-361.
[29] XU Jing-yao. Study on modified new culture genetic engineering in biotransformation of the coal [D]. Huainan Institute of Globe and Environment, Anhui University of Science and Technology, 2009: 143-153. (in Chinese).
Phanerochaete chrysosporium对活性炭的降解及劫金能力的影响
刘 倩,杨洪英,佟琳琳
东北大学 材料与冶金学院,沈阳 110819
摘 要:研究黄孢原毛平革菌(Phanerochaete chrysosporium)对碳质金矿中碳质物的替代物――活性炭的降解转化及劫金能力的影响。经Phanerochaete chrysosporium作用14 d后,活性炭的降解率高达27.59%。XRD和FTIR分析表明,Phanerochaete chrysosporium可使活性炭的微晶结构发生一定的畸变;含氧官能团和脂族结构的含量增多,一些芳烃结构氧化脱落。金吸附性试验表明,经Phanerochaete chrysosporium作用后,活性炭的吸金能力明显降低,可达12.88 %。这说明Phanerochaete chrysosporium在降解转化活性炭类碳质物及降低其劫金能力方面是一种有效的微生物,可用于处理碳质金矿中碳质物,以降低其劫金性,提高金浸出率。探讨了Phanerochaete chrysosporium对碳质物的联合作用机理(钝化机理、碱作用机理和生物酶-自由基作用机理)。
关键词:Phanerochaete chrysosporium;活性炭;降解;碳质金矿;劫金能力
(Edited by Xiang-qun LI)
Foundation item: Projects (51174062, 51104036) supported by the National Natural Science Foundation of China; Project (2012AA061502) supported by the National Hi-tech Research and Development Program of China; Project (2012BAE06B05) supported by the National Science and Technology Support Program of China during the 12th Five-Year Plan Period; Projects (N120602006, N110302002, N110602005) supported by Fundamental Research Funds for the Central Universities of China
Corresponding author: Hong-ying YANG; Tel: +86-24-83680373; E-mail: yanghy@smm.neu.edu.cn
DOI: 10.1016/S1003-6326(14)63270-2