�����������ǵ�������֬����������ͽ���
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2012���11��
�������ߣ�Meral YURTSEVER İ. Ayhan ŞENGİL
����ҳ�룺2846 - 2854
�ؼ��ʣ���������������������ѧ������Ag+���ӣ������������ǵ������֬
Key words��biosorption; desorption; kinetics; silver; Ag+ ions; absorbent; valonia tannin; resin
ժ Ҫ�������ǵ��������ȩ�ۺ����γɵ�������Ϊ����������Ч����ȥˮ��Һ�е������ӡ��о��˲�ͬ�����ӳ�ʼŨ�ȡ���Һ�¶ȡ�pHֵ���������ǵ�������֬�ĸ��ͽ�����Ϊ���о����йؾ��鶯��ѧģ�͵���Ӧ�ԡ�α��������ѧģ�ͱ��������ӵ������Ǻܿ�ġ���FTIR��SEM�������ͽ��������Ӻ������ͮ��������ǵ�������֬���б�����������������������֬�ϵ��������������ӳ�ʼŨ�ȵ����Ӷ����ӣ�������Һ�¶ȵ����߶����١��ڵ�pHֵ��H2SO4��HNO3��HCl��Һ�н��н���ʵ�顣���ǵ�������֬��pH2.0~7.0��Һ�ж������ӱ��ֳ��ߵ������������������ӳ�ʼŨ��100.0 mg/L���¶�296 K��pH5.0�������´ﵽ����������97.08 mg/g����1 mol/L HCl+1%������Һ�������ӵ������ʴ�99.6%��
Abstract: Valonia tannin (VT) was gelated through polymerization with formaldehyde to prepare an adsorbent, which was found effective to remove Ag+ from aqueous solution. The adsorption-desorption behaviors of valonia tannin resin (VTR) were investigated under various initial Ag+ concentrations, solution temperatures, pH values etc. The applicability of empirical kinetic models was also studied. The pseudo-second-order model studies revealed the Ag+ sorption was very rapid. VT and VTR were characterized using FTIR and SEM before and after adsorption. The Ag+ biosorption on VTR increased with a rise in initial concentration of Ag+ and with a decrease in temperature. Desorption experiments were conducted at low pH values and the solutions of H2SO4, HNO3 and HCl were used for desorption. The VTR shows high adsorption capacity to Ag+ in a wide pH range of 2.0-7.0, and a maximum adsorption capacity of 97.08 mg/g was obtained at pH 5.0 and 296 K when the initial concentration of Ag+ was 100.0 mg/L. Ag+ ion desorption could reach 99.6% using 1 mol/L HCl+1% thiourea (NH2CSNH2) solution. By utilizing such characteristics of VTR, it is expected that it can be applied to recovering Ag+ efficiently and simply with low cost.
Trans. Nonferrous Met. Soc. China 22(2012) 2846-2854
Meral YURTSEVER,  . Ayhan
. Ayhan 
Department of Environmental Engineering, Engineering Faculty, Sakarya University, 54187 Sakarya, Turkey
Received 2 February 2012; accepted 24 May 2012
Abstract: Valonia tannin (VT) was gelated through polymerization with formaldehyde to prepare an adsorbent, which was found effective to remove Ag+ from aqueous solution. The adsorption�Cdesorption behaviors of valonia tannin resin (VTR) were investigated under various initial Ag+ concentrations, solution temperatures, pH values etc. The applicability of empirical kinetic models was also studied. The pseudo-second-order model studies revealed the Ag+ sorption was very rapid. VT and VTR were characterized using FTIR and SEM before and after adsorption. The Ag+ biosorption on VTR increased with a rise in initial concentration of Ag+ and with a decrease in temperature. Desorption experiments were conducted at low pH values and the solutions of H2SO4, HNO3 and HCl were used for desorption. The VTR shows high adsorption capacity to Ag+ in a wide pH range of 2.0-7.0, and a maximum adsorption capacity of 97.08 mg/g was obtained at pH 5.0 and 296 K when the initial concentration of Ag+ was 100.0 mg/L. Ag+ ion desorption could reach 99.6% using 1 mol/L HCl+1% thiourea (NH2CSNH2) solution. By utilizing such characteristics of VTR, it is expected that it can be applied to recovering Ag+ efficiently and simply with low cost.
Key words: biosorption; desorption; kinetics; silver; Ag+ ions; absorbent; valonia tannin; resin
1 Introduction
Silver has the highest electrical and thermal conductivity among metals. It exhibits superior corrosion and oxidation resistance. Silver is a nonessential trace metal. The monovalent silver ion is more toxical for fish than copper or mercury, and it is an extremely effective bactericide [1]. Silver is not a dietary requirement for organisms. Antimicrobial agents such as silver are used in commercial textiles and they damage the cell wall or alter cell membrane permeability, denature proteins, inhibit enzyme activity or inhibit lipid synthesis, all of which are essential for cell survival [2]. The presence of pathogen microorganism in water and environment is one of the perennial problems. Recently, nano-silver- containing composite materials have been used in studies to remove bacteria, fungus, viruses and odor from water and environment [3-6]. The experimental studies showed that the dope loaded nano-silver particles have high practicality. In view of these notable properties, the industrial and technical usage of silver in photographic materials, electrical and electronic products, brazing alloys, batteries, silver painted bearings of aircraft, diesel locomotives and gas turbine engines, mirrors and in dental amalgams has exceeded its applications as a decorative and ornamental metal [7].
In literature, many natural organic and inorganic materials have been used as adsorbents. HANZLIK et al [8] studied the removal of Ag(I), Cd(II) and Cu(II) ions by adsorption by ��natural carbonaceous materials (spruce wood, pine bark, cork, peat, fusinite, lignite, oxidised lignite, bituminous coal and anthracite)�� [8]. Since metal ions have toxic effects on the environment, many researchers suggested cost-effective organic biosorbents such as living [9] and non-living biomass [10,11] for removing dissolved metals from wastewaters.
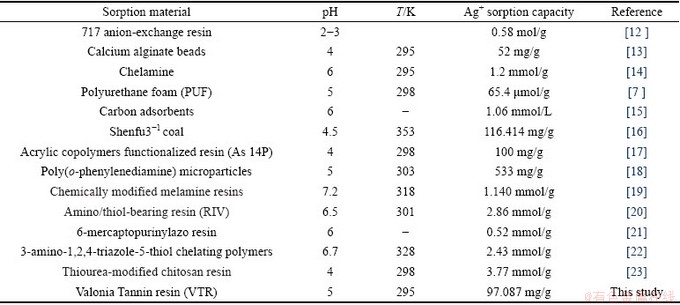
Recently, various sorbents have been used in studies to remove Ag ions and the heavy metals from water and waste water by sorption. Several studies have been proposed in the literature about the use of new sorbents, in relation with silver ions sorption from water solutions. These materials have some comparative advantages in contrast to common sorbent materials. In the last decade, researches have prepared new Ag sorbents using various materials by miscellaneous methods. Some remarkable studies can be seen in Table 1.
As seen in Table 1, there are researches all over the world, on the understanding of the mechanisms of removing Ag ions through new sorbents. Tannin adsorbents are used for metal sorption as powder or granules in direct use [24], and some studies have used them fixed onto a constant membrane. In many studies, developed tannin resins were used for uptake platinum(IV), palladium(II) [25], and bismuth(III) [26] ions as adsorbent on their own and in others adsorbent mechanisms are studied on tannins immobilized collagen fiber membrane.
Table 1 Silver sorption studies
For removal of many elements such as Hg2+, Zn2+, Pb2+, Cr2+, Cd2+, Ni2+, Bi3+, Th4+, V, U and precious metals such as Au3+, Pt4+, Pd2+, tannin sorbents have been employed in literature [27]. However, no studies on Ag+ removal with the tannin biosorbents were found. Recovery of silver as a precious metal from industrial wastewater is important from economical and environmental perspectives. The aim of the present work was to study the detailed sorption isotherms and kinetics of less-researched Ag(I) metal from aqueous solutions by tanin resins and the desorption behavior. Experiments were performed as a function of adsorption time, initial Ag+ concentration in the solution, and the temperature. Our objectives were (i) to determine and compare the isotherms of Ag+ adsorption on VTR with various physico-chemical properties, (ii) to determine and compare the sorption kinetics and mechanism of Ag+ adsorption on VTR, and (iii) to investigate the sorption�Cdesorption hysteresis phenomena and its possible mechanism.
2 Materials and methods
2.1 Preparation of VTR
Valonia tannin was selected as the raw material to synthesize the resin. Tannin was provided from the Tuzla Organized Leather Industry (Turkey). A certain amount of tannin powder was dissolved in ammonia solution at room temperature. Then, tannin was gelated through polymerization with formaldehyde (37%) at 343 K. After gelation at 343 K for 14 h, the obtained tannin resin was crushed and sieved to produce particles of 38-53 ��m in diameter. They were washed successively with distilled water and HNO3 solution (0.5 mol/L) to remove unreacted substances, and finally rinsed with distilled water again.
2.2 Batch equilibrium studies
The concentration of the silver solution was determined by an atomic absorption spectrophotometer (Shimadzu, AA-6200 type). The adsorbed Ag+ concentrations were obtained from the difference between total initial Ag+ concentration and finally detected Ag+ concentration. The maximum adsorption capacity (Qm) was calculated from isotherm data. The amount of Ag+ ion sorption onto VTR can be calculated by [28]
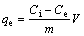 (1)
(1)
where qe is the silver ion adsorbed onto the VTR (mg/g); Ci is the initial silver ion concentration (mg/L); Ce is the final silver ion concentration in the solution (mg/L), V is the volume of the solution (L); m is the mass of used VTR adsorbent (g). Batch adsorption studies were conducted to determine the relationship between adsorbent and adsorbate, varying the amounts of adsorbate. In the study, VTR particle size of 38-53 ��m was used. Agitation rate on the Ag uptake was studied by using 350 r/min stirring speed. All experiments were repeated three times and the results given here are the average of there values.
In the all batch experiments, contact time was 180 min. Parameters such as temperature and pH are quite important on adsorption. The pH of the solution was adjusted to desired values with 0.1 mol/L HNO3 and 0.1 mol/L NaOH. Adsorption studies were performed by mixing 1 g VTR with various initial Ag solution concentrations ranging from 10 to 150 mg/L at (296��2) K and pH 5.0.
2.3 Physical and chemical characterization of adsorbent
Characterization of the adsorbents was performed in four ways: raw VT, VTR and Ag adsorbed VTR were analyzed by scanning electron microscopy (SEM), infrared spectroscopy (FT-IR) by determining of multi- point surface area and Zeta potential measurement. The SEM enables the direct observation of the changes in the surface structures of the resin due to the modifications. In order to understand the surface structure of VTR, morphological analysis of the VT and VTR was performed by SEM using a Jeol JSM-6060LV. Energy dispersive spectrophotometry (EDS) analysis was performed with EXRF SYSTEMS  nc 500 Digital processing. Fourier transform infrared (FTIR) spectra of VT and VTR were obtained by using a FTIR spectroscope (Mattson 60R). The dry beads (about 0.1 g) were thoroughly mixed with KBr and pressed into a pellet and then the FTIR spectrum was recorded. Surface area is a main characteristics that shows the adsorption ability of an adsorbent. The multi-point BET surface area analysis of VTR powder was performed with quantachrome autosorb automated gas sorption system (Vers.1.27). The analysis was carried out through an adsorption of N2 gas (gas effluent temperature=75 ��C; bath temperature=77.35 ��C). Zeta potential was measured with a Zeta potential-mobility measurement system (MALVERN Nano ZS90).
nc 500 Digital processing. Fourier transform infrared (FTIR) spectra of VT and VTR were obtained by using a FTIR spectroscope (Mattson 60R). The dry beads (about 0.1 g) were thoroughly mixed with KBr and pressed into a pellet and then the FTIR spectrum was recorded. Surface area is a main characteristics that shows the adsorption ability of an adsorbent. The multi-point BET surface area analysis of VTR powder was performed with quantachrome autosorb automated gas sorption system (Vers.1.27). The analysis was carried out through an adsorption of N2 gas (gas effluent temperature=75 ��C; bath temperature=77.35 ��C). Zeta potential was measured with a Zeta potential-mobility measurement system (MALVERN Nano ZS90).
2.4 Desorption studies
Desorption of the adsorbed silver ions from the VTR was studied in a batch system. Firstly, 1 g of VTR was used for adsorbing Ag+ ions in 1000 mL of Ag+ solution (50 mg/L) at pH 5 and then the Ag adsorbed VTR was used for desorption studies. Ag+ binding on VTR was washed with deionized water to remove any unabsorbed Ag+ ions. Then, desorption experiments were carried out using various concentrations of H2SO4, HNO3, HCl, thiourea (NH2CSNH2) and thiourea�CHCl solutions with a constant stirring (130 r/min) at room temperature for 30 and 10 min. Thiourea is an alternative nonpolluting reagent for extracting precious metals such as silver, gold, etc [29]. It is possible to take advantage of different fundamental parameters for the separation of Ag+ and Cu2+ in the presence of thiourea [30], and indeed it works in the optimal conditions. The desorption ratio was calculated from the number of metal ions adsorbed on the VTR and the final metal ion concentration in desorption medium, according to the following equation:
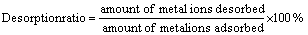 (2)
(2)
3 Results and discussion
3.1 Characteristics of resin
3.1.1 Microscopic analysis of VTR
Scanning electron micrographs were obtained on raw tannin and resin samples either before or after sorption of Ag ions (Fig. 1). As shown in Figs. 1(a) and (b), many small pores and particles with diameter <1 mm are seen on the surface of raw VT and VTR. In Fig. 1(c) the pores of VTR are partially covered with bright Ag ions.

Fig. 1 SEM images of raw VT (a), VTR (38-53 ��m in particle size) (b), Ag adsorbed VTR at (366��2) K (38-53 ��m in particle size, ��o=30 mg/L) (c)
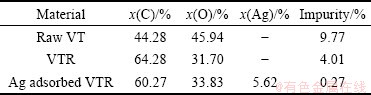
A look at Table 2 indicates that VT is not pure and it involves 9.77% impurity (S, Na, Mg and Ca ions) according to EDS (energy dispersive spectrophotometry) results. When valonia tannin is transformed into VTR, the impurity of S, Na, Mg and Ca ions decreases to 4.01%. This may be due to the chemicals added when tannin is turned into resin.
Table 2 Average surface components of raw VT and VTR by using EDS
3.1.2 Fourier transform infrared spectroscopy
FT-IR spectra of raw VT and VTR are shown in Fig. 2. Bold line in Fig. 2 illustrates the spectrum of the raw tannin before VTR. When looking at IR bands, it is seen that wide peaks in the range of 3200-3600 cm-1 show that phenolic ��OH groups are intensively present within the nature of raw tannin. In VTR, it is seen that this wide band has undergone some changes and broadened extensively. This suggests the existence of intense hydrogen bonds. Between 3000-3200 cm-1 aromatic C��H peaks have decreased in contrast to raw valonia tannin. Conversely, between 2900-3000 cm-1 aliphatic C��H groups in VTR have increased. The peaks between 1740-1750 cm-1 show the presence of ketone. The peaks between 1600-1650 cm-1 indicate the ��C=C�� bonds in aromatic nature. The peak around 1190 cm-1 shows the presence of ether (C��O��C) groups.
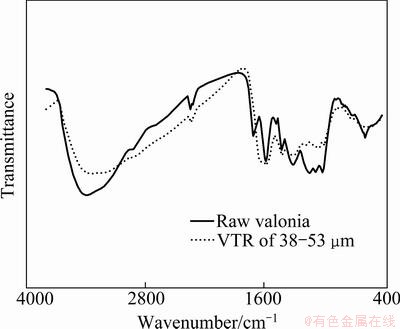
Fig. 2 FTIR spectra of raw VT and VTR
3.1.3 BET surface area
The BET specific surface area of VTR was measured to be 3.821 m2/g. In comparing the surface area of VTR and common activated carbon, it is seen that the VTR is smaller in size. The surface area of activated carbon ranges between 200-1500 m2/g [31]. Though the surface area of resin is not large, its adsorption capability to metal ions is high. This shows that functional groups in resin are more efficient than surface area in sorption process.
3.1.4 Zeta potential
Figure 3 shows Zeta potential measured in certain pH range. As seen in Fig. 3, the isoelectric point of resin corresponds to very low pH (pH=2.20). The resin has a negative surface when it is high above its pH 2 value.
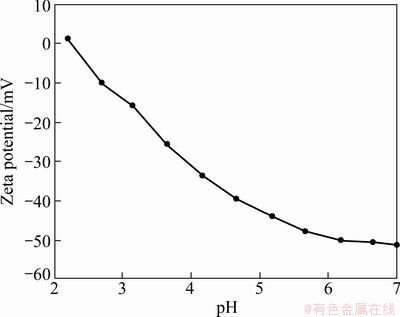
Fig. 3 Isoelectric titration graph of VTR
3.2 Influence of initial pH
In sorption study, knowledge of pH is important because the pH of solution strongly affects the sorption capacity of the resin regarding metal ions. The effect of pH on the Ag+ removal of VTR was determined over the pH ranging 2 to 7. Figure 4(a) indicates the effects of pH on the silver adsorption onto VTR. Since the surface charge of an adsorbent could be modified by changing the pH of the solution, pH is one of the most important parameters affecting the adsorption process of Ag+ ions. In order to understand the influence of this parameter on the adsorption, VTR was contacted with Ag metal ion solution at different pH values changing from 2 to 7 for 3 h. Following the equilibrium, remaining metal concentrations were determined for each solution. The results of pH experiments are presented in Fig. 4(a). It may be seen from Fig. 4(a) that adsorption of Ag+ ions increased by pH on VTR. Clearly, the maximum lead uptake by the VTR was found at pH 5. VTR contains three adjacent hydroxyl groups for binding of metal ions. Ag+ ions react with phenolic groups of the tannin resins to release protons with their anion sites to displace an existing Ag+. The adsorption of silver ions onto VTR is a pH-dependent process due to both the chemistry of silver ions in solution and the protonation of functional groups. The VTR has a negative surface at pH 5 (Eq. (3)). Silver ions can be adsorbed onto VTR by ionic interaction (Eq. (4)).
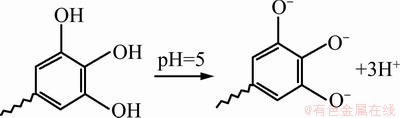 (3)
(3)
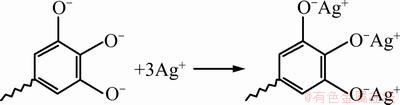 (4)
(4)
3.3 Influence of temperature and thermodynamic considerations
VTR was contacted with Ag metal ion solution at various temperatures for 3 h. Temperature studies were performed at about 296, 306, 326, 346 and 366 K. The results are shown in Fig. 4(b). Figure 4(b) shows the effect of temperature on adsorption of Ag+ ions onto VTR at pH 5.0 with an initial Ag+ concentration of 30 mg/L. It can be seen from Fig. 4(b) that the adsorption capacity of Ag+ ion decreases as the temperature rises, thus the Ag sorption process on VTR is exothermic.
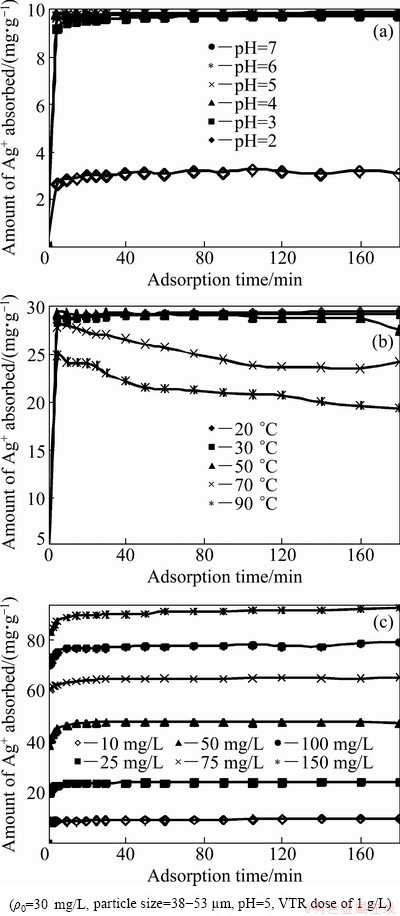
Fig. 4 pH influence (a), temperature influence (b), initial concentration influence (c) on Ag+ biosorption
The knowledge of the relevant thermodynamic parameters is needed to establish if the removal process would be able to occur spontaneously or not. Hence, the standard changes of Gibbs free energy ��G�� (kJ/mol), enthalpy ��H�� (kJ/mol), and entropy ��S�� (J��mol-1��K-1) have been estimated to evaluate the feasibility of the adsorption process. If enthalpy (��H��) is negative, the reaction is exothermic, that is the overall decrease in enthalpy is achieved by the generation of heat. A negative value of entropy (��S��) indicates a decrease in entropy, and the system becomes less random. The Gibbs free energy change of the process is related to the Kc by the following equations [32]:
��G��=-RTlnKc (5)
 (6)
(6)
where Kc is the distribution coefficient for the adsorption, Ca is the amount of adsorbate (mg) adsorbed on the adsorbent per liter of the solution at equilibrium and Ce is the equilibrium concentration (mg/L) of solution.
The thermodynamic parameters such as ��G��, ��H�� and ��S�� were determined by using the following equation:
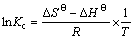 (7)
(7)
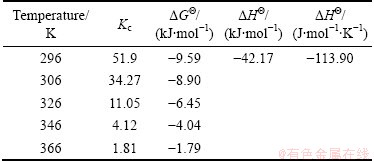
To determine thermodynamic parameters, batch experiments were carried out at different temperatures in the range of 296-366 K for Ag+ adsorption. The values of ��H�� and ��S�� were calculated from the slope and intercept of the plots of ln Kc versus 1/T. The obtained values are given in Table 3. The Gibbs free energy change (��G��) values are found to be decreasingly negative with temperature [33], which indicates the feasibility and spontaneity of the adsorption process of Ag+ ions on VTR. The negative standard enthalpy and the entropy changes showed the exothermic nature of Ag+ adsorption.
Table 3 Thermodynamic parameters for Ag+ adsorption on VTR (��o=30 mg/L, pH=5, particle size of 38-53 ��m and agitation rate of 350 r/min)
3.4 Influence of initial Ag+ concentration
The effect of initial silver ion concentration in the solution for six different concentrations of Pb (10, 25, 50, 75, 100 and 150 mg/L) on the adsorption is shown in Fig. 4(c). It can be seen from Fig. 4(c) that there was decrease in adsorption of silver with increasing silver ion concentration. Within the first 10 min, a rapid increase in Ag adsorption took place because of the large number of sites available for the sorption, and no less than 90% of total Ag+ ion was removed. After this period, owing to a reduction in the number of available sites, the silver ions took more time to access the least accessible sites. So, the sorption decelerated and reached the equilibrium approximately after 20 min.
3.5 Isotherm models for Ag+ biosorption by VTR
Four isotherm equations have been applied for this study: Langmuir [34], Freundlich [35], Temkin [36] and Dubinin-Radushkevich [37] isotherms.
The well-known linearized isotherm equation of Langmuir was reported below [34]:
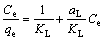 (8)
(8)
where qe is the amount of Ag(I) adsorbed at equilibrium (mg/g), and Ce is the equilibrium concentration of the adsorbate (mg/L). The empirical constants KL and aL for Langmuir model are related to the maximum capacity (L/g) and bonding strength (L/mg), respectively. The theoretical monolayer capacity is Qo and numerically equal to KL/aL.
In addition to the experimental data, the linearized form of Langmuir, Freundlich, Temkin and Dubinin- Radushkevich isotherms were used for Ag ion removal by VTR. The outcome values of parameters aL, KL, Qo, KF, n, B, A, qm, �� and R2 for all the experiments with pH of solution equal to 5 for removal of Ag+ are presented in Table 4.
The Langmuir constants can be seen from Table 4. The isotherm data fits the Langmuir equation well (R2=0.998). The value of maximum monolayer adsorption capacity (Qo) of the VTR which was used at 296 �� 2 K was found to be 97.087 mg/g. The Langmuir equation is valid for monolayer sorption onto a completely homogeneous surface with a finite number of identical sites. The results suggested that the Langmuir model was the best choice among the other isotherm models to describe the adsorption behavior of Ag+ onto VTR.
3.6 Kinetic modeling of Ag+ biosorption by VTR
In kinetic studies, intra particle diffusion model [38], Lagergren pseudo-first-order [39], Pseudo-second-order [40] and Elovich [41] models were used to test the experimental data and their formulas.
The second-order equation [40] is in the following form:
 (9)
(9)
where k2 is the rate constant of the second-order equation (g��mg-1��min1); qt the amount of adsorption at time t (mg/g); qe is the amount of adsorption equilibrium (mg/g).
Table 4 Langmuir, Freundlich Temkin and Dubinin-Radushkevich isotherm constants for Ag+ adsorption onto VTR (at (296��2) K, pH=5)

Table 5 Kinetic parameters for sorption of Ag+ on VTR
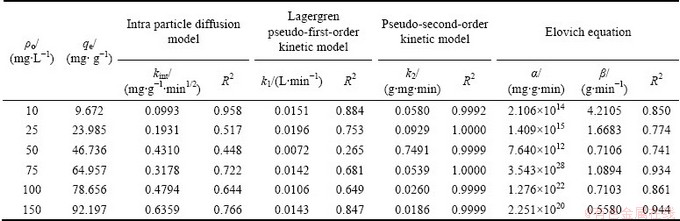
The calculated qt values were not in agreement with the experimental qe values, suggesting that the adsorption of silver ion does not follow the intra particle diffusion model, pseudo-first-order, and Elovich kinetics. In order to find a more reliable description of the kinetics, a pseudo-second-order kinetic model was applied to the experimental data. R2 and several parameters obtained from the kinetic models are shown in Table 5. The experimental data fitted well to the pseudo-second-order kinetic model with a high correlation coefficient, which indicates that the external mass transfer limitations in the system can be neglected and the chemical sorption is the rate-limiting step. Hence, the adsorption kinetics could be approximated more favorably by pseudo-second-order kinetic model for Ag+ onto VTR. A similar trend was observed in similar systems [17].
The correlation coefficients, R2, showed that the pseudo-second-order model fits better the experimental data (R2=1) than the intra particle diffusion model (R2 is 0.09-0.635), the pseudo-first-order model (R2 is 0.265-0.884) and Elovich equation (0.741-0.944).
3.7 Desorption of Ag
Samples prepared for the adsorption studies were used in the desorption studies. Desorption experiments were conducted at low pH values and the solutions of H2SO4, HNO3, HCl and thiourea-HCl were used for desorption. Initial batch desorption experiments were performed using 100 mL of 2.5 mol/L H2SO4, 5 mol/L HNO3, 5 mol/L HCl solutions with constant stirring (130 r/min) at room temperature for 30 minutes. Then the mixture was filtered and the concentration of Ag+ in filtrate was determined, and then desorption extent was calculated. Figure 5(a) shows the amount of the desorbed Ag+ in the different acid solutions. Results demonstrate that experiments conducted with 5 mol/L of acid solutions were not more efficient. Next desorption experiments were performed using 100 mL of 1 mol/L HNO3, 5 mol/L HNO3, 10 mol/L HNO3 solutions with constant stirring (130 r/min) at room temperature for 30 min. Figure 5(b) shows the amount of the desorbed Ag+ in the different HNO3 concentrations.
Results show that experiment conducted with 1 mol/L HNO3 was very efficient.
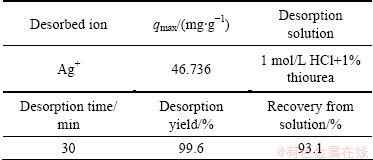
The last experiments were carried out with different HCl concentrations in the presence of thiourea. Figure 5(c) indicates the amount of the desorbed Ag+ in the different HCl-thiourea concentrations. It was found that HCl-thiourea solution is very effective for desorption of Ag+ from VTR. It can be concluded from Fig. 5(c) and Table 6 and Table 7 that 1 mol/L HCl+1% thiourea solution is the most effective solution in this desorption study. Desorption experiments for Ag were fast similar to the adsorption reaction. For all Ag desorption experiments, a greater quantity of Ag was desorbed in the presence of thiourea. In batch experiments, the adsorbed Ag on VTR can be almost completely desorbed by thiourea�CHCl solution. Studies reveal that VTR is great potential to be used as low-cost and reusable adsorbent for the removal of Ag from wastewater.
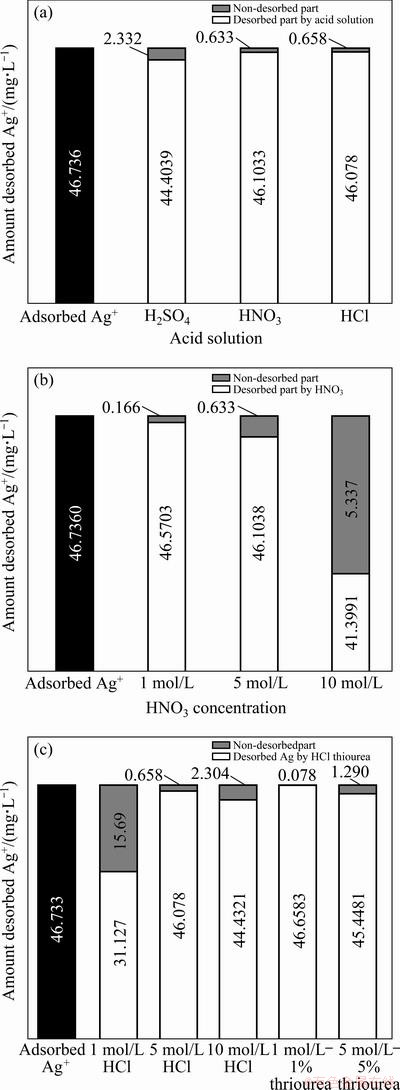
Fig. 5 Ag+ desorption in different acid solutions (a), in different HNO3 concentrations (b), in different HCl-thiourea concentrations (c)
Table 6 Ag+ desorption in 5 mol/L HCl+5% thiourea
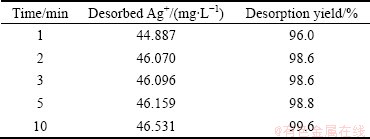
Table 7 Ag+ desorption and recovery
4 Conclusions
1) The adsorption-desorption behaviors of valonia tannin resin (VTR) were investigated under various initial Ag+ concentrations, solution temperatures, pH values etc. The results suggested that the sorption process was dependent on contact time, initial metal ion concentration, solution pH and temperature. At pH 5 of Ag+-VTR system, the adsorbent yielded a maximum Ag+ biosorption. The variation of sorption with temperature yields the values of ��G��=-9.59 kJ/mol at (296��2) K, ��H��=-43.17 kJ/mol and ��S��=-113.90 J/(mmol��K), with a correlation factor R2=0.998. The negative standard enthalpy and the entropy changes showed the exothermic nature of Ag+ biosorption. The BET specific surface area of VTR was measured as 3.821 m2/g. Zeta potential of VTR was measured and the isoelectric point of resin was corresponded to very low pH (pH=2.20). Based on the experimental data, sorption equilibrium and sorption kinetics of Ag (I) removal by VTR followed a Langmuir isotherm and pseudo-second-order kinetics. The maximum adsorption capacity of Ag+ was obtained as 97.087 mg/g at (296��2) K.
2) Desorption for Ag was fast similar to the adsorption reaction. Desorption experiments were conducted at low pH values. To get low pH values the solutions of H2SO4, HNO3 and HCl were used. It was observed that a greater quantity of Ag was desorbed in the presence of thiourea. The 1 mol/L HCl+1% thiourea (NH2CSNH2) solution can effectively desorb Ag+ ions from the VTR (99.6% efficiency). The results suggest that tannin gel is very useful as an adsorbent in a novel recovery system for silver. As a kind of low-cost, high effective and reusable adsorbent, the practical application of this novel adsorbent can be expected for the removal of Ag+ from aqueous solution. Investigation with an adsorption column is also recommended before large scale implementation.
Acknowledgements
This work has been supported by The Scientific and Technological Research Council of Turkey. The authors would like to present their deepest thanks TUBITAK for its financial support.
References
[1] JACOBSON A R, MCBRIDE M B, BAVEYE P, STEENHUIS T S. Environmental factors determining the trace-level sorption of silver and thallium to soils [J]. Sci Total Env, 2005, 345: 191-205.
[2] GAO Y, CRANSTON R. Recent advances in antimicrobial treatments of textiles [J]. Textile Res J, 2008, 78(1): 60-72.
[3] REDDY M P, VENUGOPAL A, SUBRAHMANYAM M. Hydroxyapatite-supported Ag-TiO2 as Escherichia coli disinfection photocatalyst [J]. Water Res, 2007, 41: 379-386.
[4] OLGUN U, TUNC K, OZASLAN V. Preparation of antimicrobial polycaprolactonesilica composite films with nanosilver rods and triclosan using roll-milling method [J]. Polym Adv Technol, 2011, 22: 232-236.
[5] BALAGNA C,  M, VERNE E, CANUTO R A,
M, VERNE E, CANUTO R A,  G. Biocompatibility and antibacterial effect of silver doped 3D-glass-ceramic scaffolds for bone grafting [J]. J Biomaterials Appl, 2011, 25(6): 595-617.
G. Biocompatibility and antibacterial effect of silver doped 3D-glass-ceramic scaffolds for bone grafting [J]. J Biomaterials Appl, 2011, 25(6): 595-617.
[6] WU T S, WANG K X, LI G D, SUN S Y, SUN J, CHEN J S. Montmorillonite-supported Ag/TiO2 nanoparticles: An efficient visible-light bacteria photodegradation material [J]. ACS Appl Mater Interfaces, 2010, 2(2): 544-550.
[7] HASANY S M, SAEED M M, AHMED M. Sorption of traces of silver ions onto polyurethane foam from acidic solution [J]. Talanta 2001, 54: 89-98.
[8] HANZLIK J, JEHLICKA J, SEBEK O, WEISHAUPTOVA O, MACHOVIC V. Multi-component adsorption of Ag(I), Cd(II) and Cu(II) by natural carbonaceous materials [J]. Water Res, 2004, 38: 2178-2184.
[9] GABR R M, HASSAN S H A, SHOREI A A M. Biosorption of lead and nickel by living and non-living cells of Pseudomonas aeruginosa ASU 6a [J]. International Biodeterioration and Biodegradation, 2008, 62: 195-203.
[10] AMINI M, YOUNESI H, BAHRAMIFAR N. Biosorption of nickel(II) from aqueous solution by aspergillus niger: Response surface methodology and isotherm study [J]. Chemosphere, 2009, 75: 1483-1491.
[11] TAY T, CANDAN M, ERDEM M,  H. Biosorption of cadmium ions from aqueous solution onto non-living lichen ramalina fraxinea biomass [J]. Clean, 2009, 37 (3): 249-255.
H. Biosorption of cadmium ions from aqueous solution onto non-living lichen ramalina fraxinea biomass [J]. Clean, 2009, 37 (3): 249-255.
[12] LIU Peng, LIU Guang-feng, CHEN Da-lin, CHENG Shao-yi, TANG Ning. Adsorption properties of Ag(I), Au(III), Pd(II) and Pt(IV) ions on commercial 717 anion-exchange resin [J]. Transactions of Nonferrous Metals Society of China, 2009, 19(6): 1509-1513.
[13] TORRES E, MATA Y N, BLAZQUEZ M L, MUNOZ J A, GONZALEZ F, BALLESTER A. Gold and silver uptake and nanoprecipitation on calcium alginate beads [J]. Langmuir, 2005, 21: 7951-7958.
[14] IGLESIAS M, ANTICO E, SALVADO V. The characterisation of silver sorption by chelating resins containing thiol and amine groups [J]. Solvent extraction and ion exchange, 2001, 19(2): 315-327.
[15] KONONOV O N, KHOLMOGOROV A G,  N V,
N V,  Z V. Sorption of gold and silver on carbon adsorbents from thiocyanate solutions [J]. Carbon, 2005, 43: 17-22.
Z V. Sorption of gold and silver on carbon adsorbents from thiocyanate solutions [J]. Carbon, 2005, 43: 17-22.
[16] YU Z, ZHOU A, QU J, ZHANG T. Study on behavior and kinetics of sorption of Ag+ by Shenfu3-1 coal [J]. Microporous and Mesoporous Mater, 2005, 85: 104-110.
[17] BUHACEANU R, SARGHIE I, BARSANESCU A, DULMAN V, BUNIA I. Silver sorption on acrylic copolymers functionalized with amines Equilibrium and kinetic studies [J]. Central Europ J Chem, 2009, 7(4): 827-835.
[18] LI X G, MA X I, SUN J, HUANG M R. Powerful reactive sorption of silver(I) and mercury(II) onto poly(o-phenylenediamine) microparticles [J]. Langmuir, 2009, 25: 1675-1684.
[19] EL-GHAFFAR M A A, MOHAMED M H, ELWAKEEL K Z. Adsorption of silver(I) on synthetic chelating polymer derived from 3-amino-1,2,4-triazole-5-thiol and glutaraldehyde [J]. Chem Eng J, 2009, 151: 30-38.
[20] ATIA A A, DONIA A M, YOUSIF A M. Comparative study of the recovery of silver(I) from aqueous solutions with different chelating resins derived from glycidyl methacrylate[J]. J Appl Polymer Sci, 2005, 97: 806-812.
[21] MONDAL B C, DAS D, DAS A K. Application of a new resin functionalised with 6-mercaptopurine for mercury and silver determination in environmental samples by atomic absorption spectrometry [J]. Analy Chimica Acta, 2001, 450: 223-230.
[22] EL-GHAFFAR M A A, ABDEL-WAHAB Z H, ELWAKEEL K Z. Extraction and separation studies of silver(I) and copper(II) from their aqueous solution using chemically modified melamine resins [J]. Hydrometallurgy, 2009, 96: 27-34.
[23] WANG L, XING R, LIU S, YU H, QIN Y, LI K, FENG J, LI R, LI P. Recovery of silver (I) using a thiourea-modified chitosan resin [J]. J Hazard Mater, 2010, 180: 577-582.
[24] ARASARETNAM A, KARUNANAYAKE L. Synthesis, characterization, and metal adsorption properties of tannin-phenol-formaldehyde resins produced using tannin from dried fruit of terminalia chebula (aralu) [J]. J Appl Polymer Sci, 2010, 115: 1081-1088.
[25] MA H W, LIAO X, LIU X, SHI B. Recovery of platinum(IV) and palladium(II) by bayberry tannin immobilized collagen fiber membrane from water solution [J]. J Membr Sci, 2006, 278: 373-380.
[26] WANG R,  B. Adsorption of bismuth(III) by bayberry tannin immobilized on collagen fiber [J]. J Chem Technol Biotechnol, 2006, 81: 1301-1306.
B. Adsorption of bismuth(III) by bayberry tannin immobilized on collagen fiber [J]. J Chem Technol Biotechnol, 2006, 81: 1301-1306.
[27] YURTSEVER M,  I A. Biosorption of Pb(II) ions by modified quebracho tannin resin [J]. J Hazard Mater, 2009, 163: 58-64.
I A. Biosorption of Pb(II) ions by modified quebracho tannin resin [J]. J Hazard Mater, 2009, 163: 58-64.
[28] MISHRA V, BALOMAJUMDER C, KUMAR AGARWAL V. Biosorption of Zn(II) ion onto eucalyptus leaves powder [J]. Clean-Soil, Air, Water, 2010, 38(11): 1062-1073.
[29] ONCEL M S, INCE M, BAYRAMOGLU M. Leaching of silver from solid waste using ultrasound assisted thiourea method [J]. Ultrasonics Sonochem, 2005, 12(3): 237-242.
[30] GHERROU A, KERDJOUDJ H, MOLINARI R, DRIOLI E. Removal of silver and copper ions from acidic thiourea solutions with a supported liquid membrane containing D2EHPA as carrier [J]. Sep Purif Technol, 2002, 28: 235-244.
[31]  A, OWCZAREK M, MARECKA A. Surface area of activated carbon determined by the iodine adsorption number [J]. Energy Sources Part A, 2007, 29: 839-850.
A, OWCZAREK M, MARECKA A. Surface area of activated carbon determined by the iodine adsorption number [J]. Energy Sources Part A, 2007, 29: 839-850.
[32] SEKHAR C P, KALIDHASAN S, RAJESH V, RAJESH N. Bio-polymer adsorbent for the removal of malachite green from aqueous solution [J]. Chemosphere, 2009, 77: 842-847.
[33] HORSFALL J R M, ABIA A A, SPIFF A I. Kinetic studies on the adsorption of Cd2+, Cu2+ and Zn2+ ions from aqueous solutions by cassava (Manihot sculenta Cranz) tuber bark waste [J]. Bioresour Technol, 2006, 97: 283-291.
[34] LANGMUIR I. The adsorption of gases on plane surface of glass, mica and platinum [J]. J Am Chem Soc, 1918, 40: 1361-1368.
[35] FREUNDLICH H M F. Over the adsorption in solution [J]. J Physical Chem, 1906, 57A: 385-470.
[36] TEMKIN M J, PYZHEV V. Kinetics of ammonia synthesis on promoted iron catalysts [J]. Acta Physicochim URSS, 1940, 12: 217-222.
[37] O��CONNOR T P, MUELLER J. Modeling competitive adsorption of chlorinated volatile organic compounds with the Dubinin-Radushkevich equation [J]. Micropor Mesopor Mater, 2001, 46: 341-349.
[38] WEBER W J, MORRIS J C. Advances in water pollution research: removal of biologically resistant pollutants from waste waters by adsorption[C]//International Conference on Water Pollution Symposium, Oxford: Pergamon, 1962(2): 231-266.
[39] LAGERGREN S. About the theory of so-called adsorption of soluble substances [J]. K Sven Ventenskapsakad Handl, 1898, 24: 1-39.
[40] HO Y S, MCKAY G. Pseudo-second order model for sorption processes [J]. Process Biochem, 1999, 34: 450-465.
[41] ELOVICH S Y, LARINOV O G. Theory of adsorption from solutions of non electrolytes on solid (I) equation adsorption from solutions and the analysis of its simplest form, (II) verification of the equation of adsorption isotherm from solutions [J]. Izv Akad Nauk SSSR Otd Khim Nauk, 1962, 2: 209-216.
Meral YURTSEVER,  . Ayhan
. Ayhan 
Department of Environmental Engineering, Engineering Faculty, Sakarya University, 54187 Sakarya, Turkey
ժ Ҫ�������ǵ��������ȩ�ۺ����γɵ�������Ϊ����������Ч����ȥˮ��Һ�е������ӡ��о��˲�ͬ�����ӳ�ʼŨ�ȡ���Һ�¶ȡ�pHֵ���������ǵ�������֬�ĸ��ͽ�����Ϊ���о����йؾ��鶯��ѧģ�͵���Ӧ�ԡ�α��������ѧģ�ͱ��������ӵ������Ǻܿ�ġ���FTIR��SEM�������ͽ��������Ӻ������ͮ��������ǵ�������֬���б�����������������������֬�ϵ��������������ӳ�ʼŨ�ȵ����Ӷ����ӣ�������Һ�¶ȵ����߶����١��ڵ�pHֵ��H2SO4��HNO3��HCl��Һ�н��н���ʵ�顣���ǵ�������֬��pH2.0~7.0��Һ�ж������ӱ��ֳ��ߵ������������������ӳ�ʼŨ��100.0 mg/L���¶�296 K��pH5.0�������´ﵽ����������97.08 mg/g����1 mol/L HCl+1%������Һ�������ӵ������ʴ�99.6%��
�ؼ��ʣ���������������������ѧ������Ag+���ӣ������������ǵ������֬
(Edited by YANG Hua)
Corresponding author: Meral YURTSEVER; Tel: +90-264-2955643; Fax: +90-264-2955601; E-mail: mevci@sakarya.edu.tr
DOI: 10.1016/S1003-6326(11)61541-0

