
Effects of C6H18O24P6 on properties of micro arc coatings formed on AZ91HP magnesium alloy in Na2SiO3 electrolyte
ZHANG Rong-fa(张荣发)1, XIANG Jun-huai(向军淮)1, CHAO Qiang-hua(巢强花)2, DUO Shu-wang(多树旺)1,
LI Ming-sheng(李明升)1, LI Wen-kui(李文魁)1, HE Xiang-ming(何向明)1, HU Chang-yuan(胡长员)1
1. Jiangxi Key Laboratory of Surface Engineering, Jiangxi Science and Technology Normal University,Nanchang 330013, China;
2. Jinhua College of Profession and Technology, Jinhua 321007, China
Received 15 July 2007; accepted 10 September 2007
Abstract: In a basic solution containing 18 g/L Na2SiO3, the anodizing of AZ91HP magnesium alloy was investigated with and without addition of phytic acid (C6H18O24P6). The thickness, composition and morphologies of anodic coatings formed at different concentrations of C6H18O24P6 were determined. The corrosion resistance of anodized samples was evaluated by salt spray test in 5% (mass fraction) NaCl solution. The results show that C6H18O24P6, which is harmless and environmentally friendly, greatly affects the properties of anodic coatings. Under the same electric parameters, the final voltage increases with the concentrations of C6H18O24P6. The coating thickness slightly increases from 8 mm formed in the base electrolyte without C6H18O24P6 to 9-10 mm in C6H18O24P6 solution. The P content and color of anodic coatings separately increases and darkens with the addition of C6H18O24P6. After the anodizing solutions were changed from 0 to 16 g/L addition of C6H18O24P6, the largest size of micropores decreases from 4 mm to 3 mm, while the number of micropores per area on coating surface decreases from 0.076 to 0.047 mm-2. The salt spray test shows that C6H18O24P6 can improve the corrosion resistance of the anodic coatings and the coating formed in the electrolyte containing 12 g/L C6H18O24P6 exhibits the highest corrosion resistance.
Key words: AZ91HP magnesium alloy; anodic coatings; electrolyte
1 Introduction
Because of the abundant reserves, low density and high specific strength, magnesium and its alloy offer various potentials as regards applications in the automotive, electronic and aeronautical industries[1-2]. However, the surface treatment methods need to be applied to improve their corrosion and wear resistance prior to service. Micro arc oxidation is widely used[3] and the properties of anodic coatings are mainly decided by the compositions and concentrations of the electrolytes[4]. Because magnesium is highly active and susceptible to corrosion, its anodizing solutions usually contain harmful materials such as dichromate, phosphate and fluoride[5-6]. At present, developing environmentally friendly anodizing processes has received much interest.
Phytic acid (C6H18O24P6), as an innoxious organic big molecule compound, was first used on aluminum[7] and then on magnesium[8-9] as reactant to form conversion coating. The result shows that the corrosion resistance of phytic acid conversion coating is comparable with the chromate containing[9]. Up to date, the effects of phytic acid on coating properties on magnesium alloy have not been reported although it has been used as the anodizing electrolyte for magnesium[10].
In this paper, in a basic solution containing 18 g/L Na2SiO3, the effects of C6H18O24P6 on coating properties formed on AZ91HP magnesium alloy were investigated by an eddy current instrument, scanning electron microscopy (SEM) and salt spray test.
2 Experimental
An ingot of AZ91HP magnesium alloy was employed and its chemical compositions are as follows (mass fraction, %): Al 8.93, Zn 0.47, Mn 0.22, Si 0.03,Cu 0.002, Ni 0.001, Fe 0.001, Mg balance.
Samples were anodized after they were polished successively on SiC paper up to 1 000 grit finish, degreased by acetone, washed with distilled water and dried in a cool air stream. In our experiment, unipolar current was used under constant current mode. The same electric parameters, namely, current density 40 mA/cm2, frequency 2 000 Hz, duty cycle 20% and anodizing time 3 min, were used for different processes. The base solution is 10 g/L NaOH and 18 g/L Na2SiO3. Different concentrations of phytic acid were separately added into the base solution to investigate its effects on coating properties. The compositions and concentrations of different anodizing processes are listed in Table 1.
Table 1 Compositions and concentrations of anodizing electrolytes

The morphologies of anodized samples obtained were observed after they were rinsed with distilled water and then dried in a cool air stream before coating with gold. Coating thickness was measured by a 6000-FN1 eddy current instrument (Made in Hong Kong). The corrosion resistance of the anodized samples was evaluated after 120 h salt spray test in 5% NaCl solution.
3 Results and discussion
3.1 Voltage-time responses of the five processes
Fig.1 shows the voltage―time curves of the five anodizing processes.
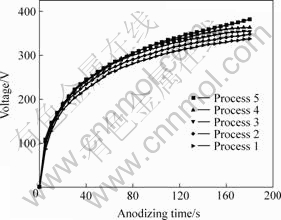
Fig.1 Curves of voltage―time by different anodizing processes
According to Fig.1, in the original anodizing stage, the working voltage increases fast for all processes. 20 s later, the slopes of curves become smaller. After anodizing for 3 min, the final voltage for process 1, 2, 3, 4, 5 are separately 338, 349, 354, 364 and 382 V. Therefore, with increasing phytic acid, the final voltage increases, too. Final voltage is an important electric parameter and it is mainly decided by the compositions and concentrations of anodizing electrolytes[11-12]. Under the same electric parameters, the final voltage increases with the concentrations of phytic acid, which may result from the decrease of the solution conductivity because the solution probably consists of large ions such as  and C6H6O6(PO3)6-
and C6H6O6(PO3)6-  [8].
[8].
3.2 Coating thickness
The thickness of anodic coatings obtained with and without addition of different concentrations of phytic acid is shown in Fig.2.
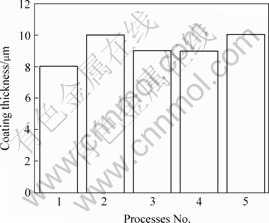
Fig.2 Coating thickness formed by different concentrations of phytic acid
Anodization in base solution (Process 1) produced an 8 mm coating. After addition of phytic acid, the coating reaches 9-10 mm but there is not an evident relation between the coating thickness and concentrations of phytic acid, which may result from the precision errors. The result indicates that phytic acid only slightly contributes to coating thickness.
3.3 Appearance and surface morphologies of anodized samples
The appearance and roughness of anodized samples by different processes were first observed with bare eyes after they were rinsed with water and dried in a cool air stream. The sample obtained by Process 1 is gray. After addition of phytic acid, the samples are smooth and their color becomes dark with the increase of C6H18O24P6. The anodized sample becomes dark when 16 g/L C6H18O24P6 was added into the solution (Process 5). Therefore, C6H18O24P6 contributes to the film color.
EDX analysis shows that the coating contains Mg, Al, Si and O (Fig.3(a)) for the base solution and Mg, Al, Si, O and P (Fig.3(b)) for the processes with addition of phytic acid. The P appearance in EDX spectrum indicates that the phytic acid takes part in the coating formation and enters the anodic coatings.
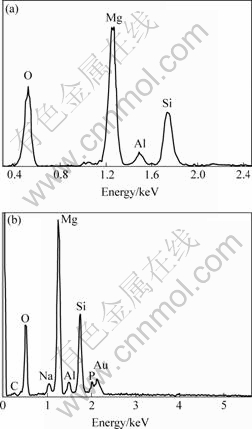
Fig.3 EDX spectra of AZ91HP anodized in solution without(a) and with(b) addition of phytic acid
The chemical compositions of anodic coatings obtained by different processes are listed in Table 2.
Table 2 Compositions of anodic coatings anodized on AZ91HP with and without addition of phytic acid (mole fraction, %)
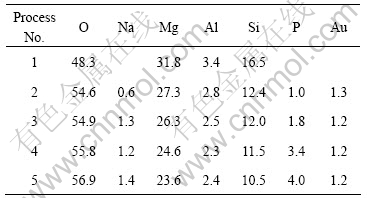
The P content of anodic coatings increases with increasing phytic acid added into the solutions. For example, the anodic coatings separately contain 1.0% and 4.0% P in solutions with 4% and 16% phytic acid (Table 2).
The surface morphologies of anodic coatings by different processes are shown in Fig.4. The coating obtained in the base electrolyte is porous and the largest size of micropores is 4 mm. Some defects shown by circles can be seen on the coating surface (Fig.4(a)). After addition of phytic acid, the coating becomes smooth (Figs.4(b)-(e)) and the largest diameter of the micropores decreases, for example, to 3 mm for coating formed in 16 g/L phytic acid. In addition, with increasing phytic acid, the number of micropores per area decreases and the data are separately 0.076, 0.074, 0.06, 0.057 and 0.047 mm-2 in solutions with additions of 0, 4, 8, 12 and 16 g/L phytic acid.
3.4 Corrosion resistance of anodized samples by different processes
After 120 h salt spray test, the corrosion resistance of anodized samples were evaluated and the sequence is as follows: Process 4>Process 5>Process 3>Process 1>Process 2. About 10% and 80% of sample surfaces by Process 4 and Process 2 are separately eroded.
Properties of anodic coatings on magnesium alloy depend on many factors, such as the electrolyte[13], electric parameters[14] and the substrate[15]. Among the affecting factors as above, the electrolyte plays a main role. As an important objective for anodizing, the high corrosion resistance of anodized samples is concerned and it is related with the coating structure, surface morphology and thickness[16]. After addition of phytic acid, the coating morphology and structure were the main factors to control its corrosion resistance because the coatings were only slightly thickened. First, in the base solution with Na2SiO3, Mg2SiO4 may be formed in anodic coatings[17-18]. After phytic acid addition, it took part in the coating formation and may exist in the coating as magnesium phytate[8]. In addition, the content of magnesium phytate in the coating increases with the concentration of phytic acid (Table 2). Second, the surface morphology of anodic coatings obtained in phytic acid is different from that in base solution. In the base solution, the number of pores per area is 0.076 mm-2. After 4 g/L phytic acid was added into the base solution, the pore number slightly decreased to 0.074 mm-2. Because the content of magnesium phytate is too low to improve the corrosion resistance, the coating obtained by Process 2 behaves the worst corrosion resistance among the five processes. When the concentration of phytic acid becomes larger (8 g/L), the number of pores per area decreases considerably and the corrosion resistance becomes better. However, when the concentration is very large (16 g/L), the corrosion resistance of anodic coatings worsens although the number of pores is small. Therefore, there exists an optimal value for the concentration of phytic acid, in which the obtained anodic coating reaches the best corrosion resistance.
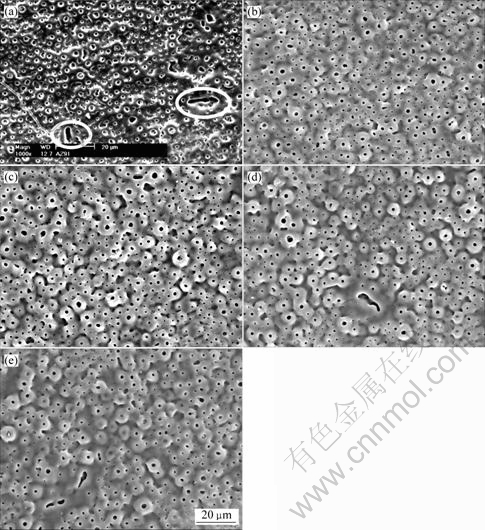 Fig.4 Surface morphologies of AZ91HP anodized at different concentrations of phytic acid: (a) 0; (b) 4 g/L; (c) 8 g/L; (d) 12 g/L; (e) 16 g/L
Fig.4 Surface morphologies of AZ91HP anodized at different concentrations of phytic acid: (a) 0; (b) 4 g/L; (c) 8 g/L; (d) 12 g/L; (e) 16 g/L
4 Conclusions
1) Under the same electric parameters, the final voltage increases with the concentrations of C6H18O24P6.
2) C6H18O24P6 can slightly thicken anodic coatings from 8 mm formed in the basic solution containing 18 g/L Na2SiO3 to 9-10 mm after its addition.
3) With increasing C6H18O24P6, the P content and the color of anodic coatings separately increases and darkens, while the largest diameter and the number of micropores per area decreases. In the anodizing solutions, after the addition of C6H18O24P6 changing from 0 to 16 g/L, the largest size of micropores decreases from 4 mm to 3 mm, while the number of micropores per area on the coating surface decreases from 0.076 to 0.047 mm-2.
4) C6H18O24P6 can improve the corrosion resistance of anodic coatings and the optimal value is 12 g/L.
References
[1] MORDIKE B L, EBERT T. Magnesium properties-applications- potential[J]. Mater Sci Eng A, 2001, 302: 37-45.
[2] MEHTA D S, MASOOD S H, SONG W Q. Investigation of wear properties of magnesium and aluminum alloys for automotive applications[J]. Journal of Materials Processing Technology, 2004, 155/156:1526-1531.
[3] GRAY J E, LUAN B. Protective coatings on magnesium and its alloys-a critical review [J]. Journal of Alloys and Compounds, 2002, 336: 88-113.
[4] CAI Q Z, WANG L S, WEI B K, LIU Q X. Electrochemical performance of microarc oxidation films formed on AZ91D magnesium alloy in silicate and phosphate electrolytes[J]. Surface and Coatings Technology, 2006, 200: 3727-3733.
[5] Dow Chemical Co. Bath for and method of producing a corrosion resistant coating upon light metals. GB 762195[P]. 1956.
[6] EVANGELIDES H A. Method of electrolytically coating magnesium and electrolyte therefor. US 2723952[P]. 1955.
[7] YSAHIRO, KUNI J, MORIYA, YOSHIO. Metal surface coating agent[P]. US: 4341558, 1982.
[8] ZHENG R F, LIANG C H, SHAO L. Study of composition and corrosion protection afforded by phytic acid conversion coatings applied to AZ91D magnesium alloy[J]. Journal of Dalian University of Technology, 2006, 46(1): 16-19. (in Chinese)
[9] LIU J R, GUO Y N, HUANG W D. Study on the corrosion resistance of phytic acid conversion coating for magnesium alloys[J]. Surface and Coatings Technology, 2006, 201: 1536-1541.
[10] HUI H Y, YU G, YE L Y. A research on an environmentally-friendly electrolyte for magnesium alloy anodizing[J]. Electroplating and Pollution Control, 2005, 25(6): 34-37. (in Chinese)
[11] YAHALOM J, HOAR T P. Galvanostatic anodizing of aluminium[J]. Electrochimica Acta, 1970, 15: 877-884.
[12] IKONOPISOV S. Theory of electrical breakdown during formation of barrier anodic films[J]. Electrochimica Acta, 1977, 22: 1077-1082.
[13] MA Y, NIE X, NORTHWOOD D O, HU H. Systematic study of the electrolytic plasma oxidation process on a Mg alloy for corrosion protection[J]. Thin Solid Films, 2006, 494: 296-301.
[14] SHI Z M, SONG G L, ATRENS A. Influence of anodising current on the corrosion resistance of anodised AZ91D magnesium alloy[J]. Corrosion Science, 2006, 48(8): 1939-1959.
[15] SHI Z M, SONG G L, ATRENS A. Influence of the β phase on the corrosion performance of anodised coatings on magnesium- aluminium alloys[J]. Corrosion Science, 2005, 47: 2760-2777.
[16] LIANG J, HU L T, HAO J C. Characteristization of microarc oxidation coatings formed on AM60B magnesium alloy in silicate and phosphate electrolytes[J]. Applied Surface Science, 2007, 253: 4490-4496.
[17] ZHANG R F, SHAN D Y, HAN E H, DUO S W. Effects of current mode on properties of anodic coatings of magnesium alloys[J]. Rare Metal Materials and Engineering, 2006, 35(9): 1392-1395. (in Chinese)
[18] FUKUDA H, MATSUMOTO Y. Effects of Na2SiO3 on anodization of Mg-Al-Zn alloy in 3 mol/L KOH solution[J]. Corrosion Science, 2004, 46: 2135-2142.
(Edited by LONG Huai-zhong)
Foundation item: Project(0650034) supported by the Natural Science Foundation of Jiangxi Province, China
Corresponding author: ZHANG Rong-fa; Tel: +86-791-3831266; E-mail: rfzhang-10@163.com