High efficient removal and mineralization of Cr(VI) from water by functionalized magnetic fungus nanocomposites
��Դ�ڿ������ϴ�ѧѧ��(Ӣ�İ�)2020���5��
�������ߣ������� ���� ����Ө ��ƽ ��־�� �����
����ҳ�룺1503 - 1514
Key words��wastewater; Cr(VI); fungus nanocomposites; biomineralization
Abstract: A hydroxyl-functionalized magnetic fungus nanocomposite (MFH@GO) was prepared by a simple one-pot method for the removal of Cr(VI) from wastewater. The adsorption behavior of MFH@GO to Cr(VI) in wastewater was discussed in detail. At pH of 5.0 and temperature of 323.15 K, MFH@GO had higher adsorption capacity to Cr(VI) (58.4 mg/g) than the unmodified fungus and GO. Fourier transform infrared spectroscopy (FTIR), X-ray diffraction (XRD), thermogravimetry and differential thermal analysis (TG-DTA), scanning electron microscopy and energy dispersive X-Ray spectroscopy (SEM-EDX) were employed to determine the characteristics of MFH@GO. Results showed that magnetic graphene oxide nanoparticles significantly enhanced the physiochemical properties of the fungi. In addition, the adsorption mechanisms analyses show that Cr(VI) could be reduced and mineralized into ferric chromate in residues. These results suggested that MFH@GO could be used as an promising and alternative biosorbent for removal of Cr(VI) from industrial wastewater.
Cite this article as: CHEN Run-hua, CHENG Yu-ying, WANG Ping, LIU Zhi-ming, WANG Yu-guang, WANG Yang-yang. High efficient removal and mineralization of Cr(VI) from water by functionalized magnetic fungus nanocomposites [J]. Journal of Central South University, 2020, 27(5): 1503-1514. DOI: https://doi.org/10.1007/s11771- 020-4386-y.

J. Cent. South Univ. (2020) 27: 1503-1514
DOI: https://doi.org/10.1007/s11771-020-4386-y
CHEN Run-hua(����)1, CHENG Yu-ying(����Ө)1, WANG Ping(��ƽ)1,
LIU Zhi-ming(��־��)3, WANG Yu-guang(�����)4, WANG Yang-yang(������)2
1. College of Environmental Science and Engineering, Central South University of Forestry and Technology,Changsha 410007, China;
2. National Demonstration Center for Environmental and Planning, College of Environment & Planning,Henan University, Kaifeng 475004, China;
3. Department of Biology, Eastern New Mexico University, Portales, NM88130, USA;
4. School of Minerals Processing and Bioengineering, Central South University, Changsha 410083, China
 Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2020
Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2020
Abstract: A hydroxyl-functionalized magnetic fungus nanocomposite (MFH@GO) was prepared by a simple one-pot method for the removal of Cr(VI) from wastewater. The adsorption behavior of MFH@GO to Cr(VI) in wastewater was discussed in detail. At pH of 5.0 and temperature of 323.15 K, MFH@GO had higher adsorption capacity to Cr(VI) (58.4 mg/g) than the unmodified fungus and GO. Fourier transform infrared spectroscopy (FTIR), X-ray diffraction (XRD), thermogravimetry and differential thermal analysis (TG-DTA), scanning electron microscopy and energy dispersive X-Ray spectroscopy (SEM-EDX) were employed to determine the characteristics of MFH@GO. Results showed that magnetic graphene oxide nanoparticles significantly enhanced the physiochemical properties of the fungi. In addition, the adsorption mechanisms analyses show that Cr(VI) could be reduced and mineralized into ferric chromate in residues. These results suggested that MFH@GO could be used as an promising and alternative biosorbent for removal of Cr(VI) from industrial wastewater.
Key words: wastewater; Cr(VI); fungus nanocomposites; biomineralization
Cite this article as: CHEN Run-hua, CHENG Yu-ying, WANG Ping, LIU Zhi-ming, WANG Yu-guang, WANG Yang-yang. High efficient removal and mineralization of Cr(VI) from water by functionalized magnetic fungus nanocomposites [J]. Journal of Central South University, 2020, 27(5): 1503-1514. DOI: https://doi.org/10.1007/s11771- 020-4386-y.
1 Introduction
Heavy metals pollution has been considered one of the main environmental problems in the world nowadays [1-3]. Hexavalent chromium (Cr(VI)), as a major toxic compound, has attracted the attention of researchers in recent years [4-6]. Cr(VI) is released to environment mainly by anthropogenic activities, including smelting, mining, electroplating, and textile manufacturing, etc [7, 8]. The credible evidence found that Cr(VI) is carcinogenic and neurotoxic, which is considered one of the three internationally recognized carcinogenic metals [9]. It can be accumulated in organisms and then causes more serious harm, such as skin ulcer, liver and kidney damages [10, 11]. Therefore, remediation of Cr(VI) in industrial water is urgently required.
Various treatment technologies have been applied to removing Cr(VI) from wastewater and contaminated soil. In recent years, the application of adsorbent to remove Cr(VI) has become a hotspot [10, 12]. An adsorption experiment was conducted for removal of Cd(II) and Cr(VI) using Aspergillus sp. and Rhizopus sp. The results showed that the maximum adsorption capacity for Cd(II) and Cr(VI) was 2.72 mg/g and 1.20 mg/g by Aspergillus sp., respectively, and the maximum adsorption capacity of Rhizopus sp. for Cd(II) and Cr(VI) was 2.72 mg/g and 4.33 mg/g, respectively [13]. The magnetotactic bacteria have also been used to adsorb Cr(VI) from water with the adsorption efficiency of 77%. However, compared with other adsorbents, such as biochar [14], activated carbon[15], clinoptilolite [16], the adsorption capacity of the microorganisms to heavy metals is still lower.
On the other hand, the widely application of nanoscale materials is crucial for wastewater treatment [17-19], such as graphene oxide (GO). The functionalized graphene removed Cr(VI) (500 mg/L) efficiently from water within 3 h [20]. The graphene oxide/NiO nanocomposite was used to adsorb Cr(VI) from water, and its maximum adsorption capacity to Cr(VI) at pH of 4.0 and temperature of 25 ��C reached 198 mg/g [21]. The adsorption capacity of magnetic Fe3O4@poly(m- phenylenediamine) to Cr(VI) reached 246.09 mg/g [22]. However, the removal of these suspended adsorbents from continuous flow water is a challenge [23]. Besides, the graphene oxide is prone to agglomeration due to adsorption, resulting in a significant decrease in its adsorption performance, and it is not easy to recycle [24]. It is expensive to make nanomaterials, and it is difficult to realize their stability without biological matrix immobilization, which limits their application [25].
In this study, a novel multi-functional magnetic nanocomposite MFH@GO with micro- nano structure and macro-size was prepared through combination of fungal hyphae with GO and Fe, and the Aspergillus niger hyphae was used as a template. Batch adsorption experiments were used to evaluate the change adsorption capacity of MFH@GO towards Cr(VI) with the change of environmental factors, such as initial concentration of Cr(VI), pH value and temperatures. The possible adsorption mechanisms of Cr(VI) on MFH@GO were also explored.
2 Experimental designs
2.1 Materials
The freeze-dried strain tube of Aspergillus niger (ATCC 16404) was purchased from the General Microorganisms Collection and Management Center (Wuhan, China). Glucose, ethanol, magnesium sulfate heptahydrate (MgSO4��7H2O), dipotassium hydrogen phosphate (K2HPO4), ferrous sulfate (FeSO4), sodium borohydride (NaBH4), hydrogen peroxide (H2O2) and potassium dichromate (K2Cr2O7) were purchased from Sinopharm Chemical Reagent Co., Ltd. The stock solution of Cr(VI) was prepared by dissolving K2Cr2O7 in ultrapure water. All the reagents were of analytical grade and used as received.
2.2 Culture of Aspergillus niger
The medium used for culture of Aspergillus niger was according to the description of ZHANG et al [26]. The culture conditions were 175 r/min and 30 ��C for 24-48 h in a rotary shaker. The growth and the globulation of hyphae were observed every half day to prevent the growth of spores, and finally Aspergillus niger pellet with uniform size was obtained.
2.3 Composite of hyphae (FH) and iron
The Aspergillus niger pellet was washed repeatedly with ultrapure water until the filter liquor was clarified. After inactivation by hot water bath, the hyphae were crushed with a stirrer, and the bottles of hyphae slag were cryopreserved. 5 mL of the Aspergillus niger hyphae solution was taken and then filtered to calculate the concentration of the hyphae solution. The ferrous sulfate particles were weighed in proportion and completely dissolved in distilled water, and the hyphae solution was added and stirred uniformly. Sodium borohydride was added dropwise to the mixture by means of a syringe pump, and in the process, the mixer was continuously agitated so that the iron could be uniformly covered on the surface of the hyphae. It was observed that the color of the solution changed from light green to black, basically no bubbles at the end point, and the magnetism could be clearly felt by moving the magnet on the wall of the beaker.
2.4 Composite of hyphae (FH)/Fe and graphene oxide (GO)
The filtrate was repeatedly rinsed with distilled water until it was as clear as distilled water to rinse off the reductant on the surface. The filtered residue was mixed with distilled water, and then the graphene oxide was added and treated by ultrasound. In this process, the mixture was stirred continuously, while hydrogen peroxide was also added at the same time. The obtained mixture was then sub-packed in 150 mL Erlenmeyer flask and placed in a shaker for 1 h to uniformly coat the graphene oxide on the surface of the hyphae material. MFH@GO composites were made by repeatedly rinsing with distilled water and filtering until the filtrate clarified. The filtrate was dried completely in an oven until completely being dried, yielding MFH@GO nanoparticles for further use.
2.5 Batch adsorption experiment
Batch adsorption experiment was carried out in a 150 mL Erlenmeyer flask including 0.1 g MFH@GO composite or graphene oxide, 100 mL Cr(VI) solution with known concentration. The initial concentration of Cr(VI) solution was adjusted by quantitative dilution of standard solution until the target concentration was achieved. Herein, effects of contact time, initial Cr(VI) concentration, initial pH value of solution and temperature of adsorption on adsorption reaction of Cr (VI) by MFH@GO were determined by single factor analysis. The adsorption reaction time was adjusted from 1 min to 480 min (1, 5, 10, 20, 40, 60, 120, 240, 480 min). The initial Cr(VI) concentrations were adjusted from 30 mg/L to 90 mg/L (30, 40, 50, 70, 90 mg/L). The initial pH value of solution was adjusted from 1.0 to 10.0 by adding HCl or NaOH solution. The temperature of adsorption reaction was adjusted to three different temperatures (273.15, 298.15, 323.15 K). Then, the conical flasks containing reaction solution were shaken in a constant temperature shaker at 150 r/min. The concentration of Cr(VI) was analyzed with a UV spectrophotometer by the method of diphenylcarbazide spectrophotometry (UV-2350, Shimadzu). The absorbance of samples was measured at 540 nm. The adsorption capacity (qt, mg/g) and rate (��, %) of Cr(VI) were reckoned by the following formulas:
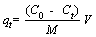 (1)
(1)
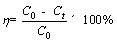 (2)
(2)
where C0 (mg/mL) and Ct (mg/mL) refer to the concentration of Cr(VI) solution in initial states and adsorbed for t time respectively; V (mL) is the solution volume; M (g) is the adsorbent amount used. Three parallels were set up in the experiment.
2.6 Characterization
SEM imaging was performed with Sachi S4800 at 5 kV. XRD was conducted on D8 ADVANCE type (Bruker Company, Germany). TGA was carried out using STA449F3 type (NETZSCH Company, Germany) under nitrogen with heating rate of 10 ��C/min. FT-IR spectra of films were taken by a spectrum one Perkin-Elmer spectrometer at 4000-400 cm-1 wavelength.
3 Results and discussion
3.1 Characterization of MFH@GO
The morphology, microstructure and elemental constitution of MFH@GO were shown by SEM and EDAX (Figure 1). The formation process of MFH@GO composite can be clearly seen in Figure 1(a). The films with great tensile resistance were made (Figure 1(b)). Winding rope-like structure of MFH@GO with uniform sizes could be seen in Figure 1(c), which conforms to fungal morphology and is beneficial to adsorption. Elemental composition of solid samples is usually analyzed by EDAX. Figure 1(d) shows that the main elements of MFH@GO were C, O, Fe and S by EDAX, indicating that the graphene oxide and iron were successfully grafted on the surface of mycelial materials.
The chemical structures of the material and the functional groups on the surface of the material were determined by FTIR (Figure 2). As seen in Figure 2 (before being treated), a wide tensile strength peak was found near 3390 cm-1, which is caused by the ��OH groups on the surface of mycelium. The band at 2900 cm-1 belongs to the asymmetric stretch of ��CH2 groups and there is corresponding symmetric stretch at 2852 cm-1. Protein-related C=O bonds can be observed at 1650 cm-1. The peak at 1394 cm-1 represents functional groups containing oxygen such as a highly conjugated C��O stretching in carboxyl groups. The C��O stretching in ether or alcohol and methoxyl groups was observed at around 1035 cm-1 [27]. This suggests the presence of ��OH, C��O, C=O compounds in the mycelial surface which acts as a capping or cheating base. As shown in Figure 2 (MFH@GO), the weak stretch is observed at 3390 cm-1. It is generally believed that the process of Fe and graphene oxide cheated onto mycelial surface is mainly through ligand exchange with��OH group. However, the significant changes can be seen the peak at 809 and 631 cm-1 for Fe��O stretching vibrations [28]. These functional groups on the MFH@GO surface can effectively complex with heavy metals, which greatly enhances the adsorption of heavy metals and expands magnetic collection and regeneration properties of the material.
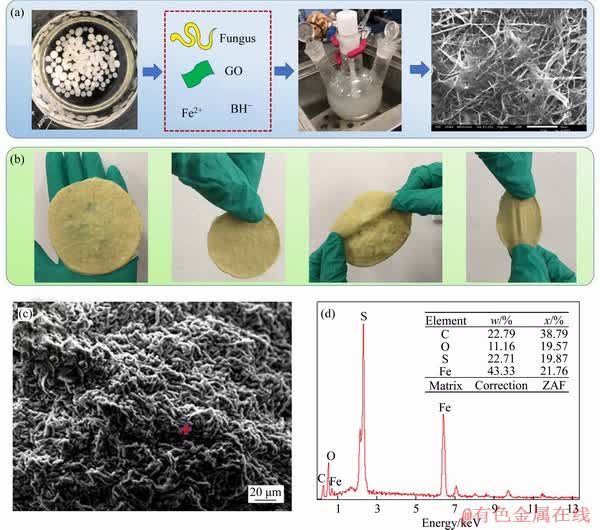
Figure 1 Synthetic process of MFH@GO composites (a), macro-ductility of synthesized FH materials(b), surface structure of MFH@GO characterized by SEM (c) and EDS analysis results of MFH@GO (d)

Figure 2 FTIR spectra of raw fungus before treatment and MFH@GO
The thermogravimetric-differential thermal analysis (TG-DTA) was conducted for MFH@GO composite material (Figure 3). The weight loss occurred in the range from 0 ��C to 100 ��C, corresponding to a weight loss of 2.61%, which is ascribed to the disappearance of the interstitial water. The second weight loss was from 100 ��C to 430 ��C, arising from the loss of the hyphae of MFH@GO. During the stage, biomass was converted to water and carbon dioxide, and the rate of weight loss was the fastest, which was 18.91%. The third stage ranged from 430 ��C to 540 ��C. At this stage, the oxidation of iron led to the production of iron oxide, which increased the weight by 6.16%. Finally, the residue was 91.78%. The TG-DTA further proves the existence of iron and mycelium in composite materials, which is consistent well with the design ideas and the data analysis of EDS and FTIR.
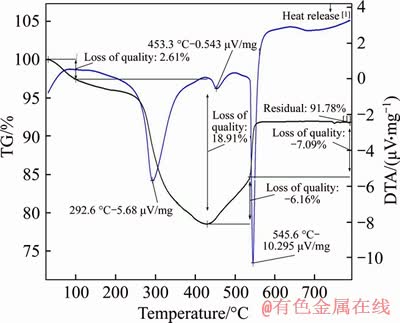
Figure 3 TG-DTA thermo analytical response curves of MFH@GO in airflow at a heating rate 10 ��C/min
3.2 Adsorption experiment on Cr(VI)
3.2.1 Effects of various factors
Figure 4(a) reveals the effect of contact time on the adsorption capacity, which was studied in 50 mg/L Cr(VI) solution, 298 K, pH 5.0(��0.1). Within 10 min of the reaction beginning, the adsorption capacity rose sharply. When the adsorption time reached 60 min, the adsorption reaction gradually slowed down and the adsorption rate decreased significantly. At 120 min, the adsorption reaction on MFH@GO composites reached equilibrium. It is speculated that MFH@GO may have many mechanisms for Cr(VI) adsorption reaction. In initial stage of adsorption reaction, physical adsorption is dominant, and Cr(VI) ions rapidly diffuse near the adsorbent and contact with the solid surface of the adsorbent to occupy the active sites. In the second stage, chemical reaction is dominant, because iron is an active metal element, and its electrode potential is E��(Fe2+/Fe)=-0.4402 V, and its reduction ability is strong. The Cr(VI) has a strong oxidizing ability, and it can be reduced by chemical reaction, so that Cr(VI) can be transformed into more stable Cr(III) [29-31], and then the purpose of removing Cr(VI) is achieved. After 8 h reaction, the adsorption capacities of MFH@GO and GO are 48.8 and 4.4 mg/g, respectively. Results show that the removal efficiency of Cr(VI) by MFH@GO composite is better than that of GO, because the specific surface area and reactivity of MFH@GO composite are higher than those of GO.
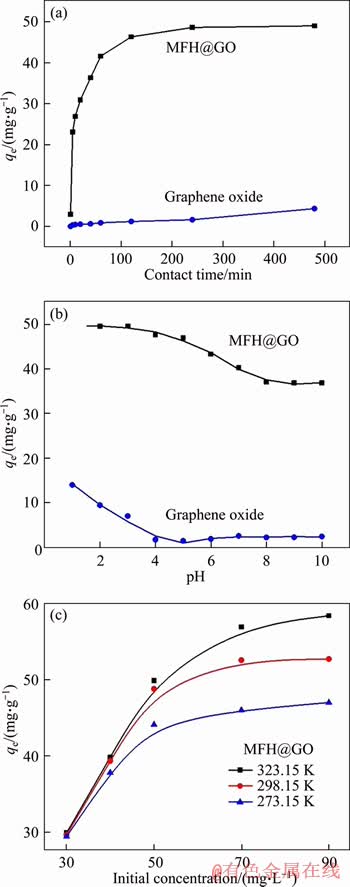
Figure 4 Effects of contact time (a), pH (b) and initial concentration at various temperature (c) on adsorption of GO and MFH@GO to Cr(VI) (qe: equilibrium adsorption capacity)
The effect of initial pH on the adsorption reaction of Cr(VI) on MFH@GO and GO was discussed in 50 mg/L Cr(VI) solution at 298 K. In Figure 4(b), the adsorption reaction of Cr(VI) was affected obviously by the pH of Cr(VI) solution. When the pH value increased from 1.0 to 5.0, the removal effect of Cr(VI) by GO decreased with the increase of the initial pH value of the solution, and then reached its equilibrium value. With the increase of pH from 1.0 to 8.0, the removal effect of Cr(VI) by MFH@GO decreased with the increase of the initial pH value of the solution, and when pH value reached 8, the adsorption reaction reached equilibrium. Compared to GO (14.8 mg/g), MFH@GO (49.5 mg/g) had a great removal effect on Cr(VI) ions. The better adsorption efficiency of MFH@GO under acidic conditions may be due to the following two reasons. On one hand, in lower pH environment, amino groups on the surface of the Aspergillus niger were positively charged due to protonation, and the negatively charged Cr(VI) in the chromium-containing wastewater was easily attracted to the surface of MFH@GO, and then reacted with positively charged functional groups. On the other hand, when the pH was too high, the proportion of hydroxide ions in the solution was large, and iron reacted with it to produce precipitations or complexes, which affected the treatment effect of the composite material. Therefore, the adsorption rate of Cr(VI) by MFH@GO decreased with the increase of pH.
The effect of initial concentration at various temperature (273.15, 298.15, 323.15 K) for Cr(VI) adsorption is shown in Figure 4(c). The curve shows that the adsorption capacity of Cr(VI) on the composite all increased with the increase of initial concentration of Cr(VI) at various temperatures (273.15, 298.15, 323.15 K). When the concentration of Cr(VI) was 90 mg/L, the adsorption capacity of the composite reached the maximum. At higher concentrations (the initial concentration of Cr(VI)��50 mg/L), the possibility of collision with the adsorbent is increased due to the large number of adsorbable Cr(VI) ions in the solution, and the degree of influence of the equilibrium adsorption capacity also increases. When the concentration of Cr(VI) in the wastewater was 90 mg/L, the maximum adsorption capacity of the composite reached 58.4 mg/g at 323.15 K, while at 273.15 K, which was only 47.0 mg/g. This change indicates that the rising temperature promotes the adsorption reaction. Because the increase of temperature is beneficial to improve diffusion rate of the adsorbate in the solution, which contributes to the probability of collision between Cr(VI) and MFH@GO. When the probability of collision between Cr(VI) and MFH@GO is higher the Cr(VI) removal in solution. In addition, the temperature change may also cause the expansion of the pores inside the adsorbent, which is beneficial to increase equilibrium adsorption capacity of adsorbent.
3.2.2 Adsorption isotherms
Figure 5 shows the typical adsorption isotherms of Cr(VI) on MFH@GO. The Cr(VI) adsorption capacity of MFH@GO increased rapidly with the increase of equilibrium concentration. The adsorption behavior of MFH@GO on Cr(VI) fitted well with Langmuir isotherm [32] adsorption model (Figure 5(a)), and the correlation coefficients R2 were 0.968, 0.962 and 0.981 at three different temperatures (273.15, 298.15, 323.15 K), respectively. This indicated that the functional groups adsorbed on the surface of MFH@GO composite were uniformly distributed, and all adsorption sites had the same affinity for Cr(VI). It can be inferred that the adsorption reaction belongs to the surface chemical adsorption of single molecular layer. The maximum theoretical adsorption capacity calculated by the formula was similar to the actual measured adsorption capacity. The maximum theoretical adsorption capacity increased with the increase of temperature. According to Freundlich isotherm [33] adsorption formula (Figure 5(a)), the n value calculated was more than 1, indicating that MFH@GO composite had a good adsorption performance and strong adsorption reaction intensity. Clearly, its react to Cr(VI) was preferential adsorption. When the Kf value increased, the adsorption capacity of MFH@GO composite also increased with the increase of temperature. The value of adsorption energy E can be calculated by D-R model (Figure 5(b)). As shown in Table 1, the E values at three temperatures are 26.45, 23.84 and 20.99 kJ/mol, respectively. It is proved that the adsorption of Cr(VI) on MFH@GO belongs to chemical adsorption.
3.2.3 Adsorption kinetics
The effect of contact time was investigated in 50 mg/L Cr(VI) solution at pH 5(��0.1), 298 K. Adsorption capacity was measured at three different temperatures (273.15, 298.15, 323.15 K) at different times (1, 5, 10, 20, 40, 60, 120, 240, 480 min). The experimental data were fitted by pseudo-first-order [34] (Figure 6(a)) and pseudo- second-order [35] (Figure 6(b)) kinetics models, respectively. The fitting results of the experimental data showed that the adsorption kinetics of Cr(VI) on MFH@GO followed the pseudo-second-order kinetic model, and correlation coefficients were 0.999, 0.999 and 0.998 at three different temperatures (273.15, 298.15, 323.15 K), respectively (Table 2). Thus the adsorption rate of Cr(VI) was limited by the step of chemisorption. This is due to the unique structure of MFH@GO, which has many chelating sites for Cr(VI) and the MFH@GO composite could adsorb Cr(VI) rapidly. It is as same as the result of adsorption isotherm analysis.
3.2.4 Adsorption thermodynamics
In order to further elucidate the adsorption mechanism, Gibbs-Helm Holtz equation and Van��t Hoff equation were used for thermodynamic analysis. As shown in Figure 6(c), linear correlation equation can be obtained by linear fitting with 1/T as abscissa and lnK as ordinate. According to the formula, the values of free energy ��G, standard enthalpy ��H, and standard entropy ��S can be calculated (Table 3). Because the standard enthalpy change ��H at three different temperatures (273.15, 298.15, 323.15 K) is positive, the adsorption reaction of MFH@GO to Cr(VI) is an endothermic process with the degree of confusion increasing. Gibbs free energy ��G was negative at different temperatures, and the absolute value of ��G increased with the increase of temperature, indicating that the adsorption reaction is spontaneous and increasing temperature is conducive to the reaction.
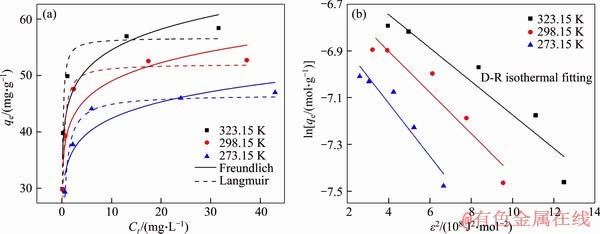
Figure 5 Non-linear fitting of equilibrium data to Langmuir and Freundlich isotherms (a) and D-R isotherm (b) for adsorption of Cr(VI) onto MFH@GO at various temperatures
Table 1 Fitting comparison of isothermal adsorption models at different temperatures
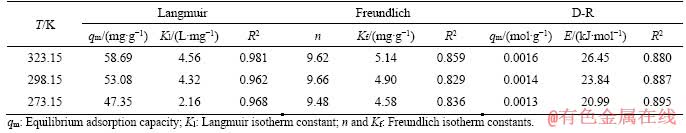
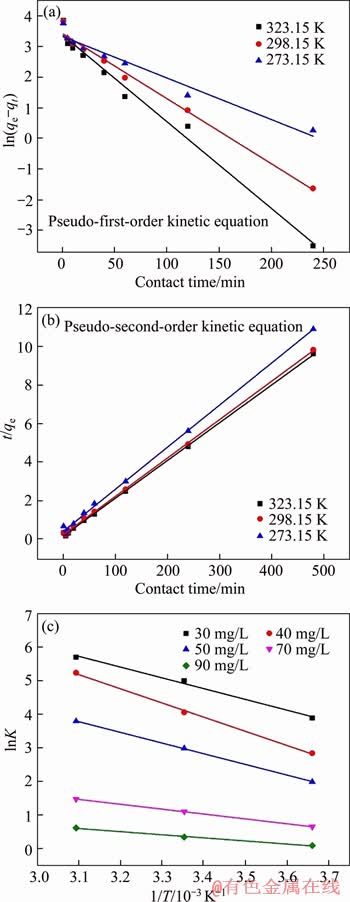
Figure 6 Pseudo-first-order kinetic (a), pseudo-second order kinetic (b) and thermodynamic analysis diagrams (c) of uptake of Cr(VI) by MFH@GO
3.3 Adsorption mechanisms
The morphology of MFH@GO composite changed into porous helical chain structure after adsorption, and the surface of the MFH@GO composite was covered by Cr(VI), which reduced the porosity of the material and the roughness of surface. Elemental mapping images of the MFH@GO adsorption samples after adsorption are shown in Figure 7(a), showing the even distribution of C, N, O, S on the surface of MFH@GO after adsorption. It can be observed that after the adsorption reaction, a large number of bright color spots appeared on the hyphae, indicating that the chromium can be well adsorbed on the composite material.
Compared with FTIR spectra of MFH@GO before adsorption, all the characteristic peaks of MFH@GO composites were retained in the infrared spectra after adsorption reaction (Figure 7(b)). However, because of the adsorption reaction, the corresponding bands were slightly reduced and the adsorption bands were offset. The response peaks of O��Fe��O and Fe��O on the curve were weakened in comparison with those before the adsorption [36]. It is presumed that the adsorption of Cr(VI) by MFH@GO composites is mainly related to the iron-oxygen bond. The absorption peaks of ��OH stretching vibration can be observed at 3320 cm-1 before the adsorption [37], but the absorption peaks became blunt after adsorption reaction, presumably because ��OH participates in the adsorption reaction.
The thermogravimetric-differential thermal analysis of MFH@GO before and after adsorption was compared in Figure 7(c). The weight change of MFH@GO in three stages after adsorption is a little more obvious than that before adsorption. However, the weight increase of MFH@GO after the adsorption (1.09%) is not obvious compared with that before the adsorption (7.09%) in the range from 590 ��C to 789.5 ��C, and the residue of MFH@GO after adsorption is 77.95%, which is less than that before the adsorption (91.78%). It may be due to the loss of iron in the adsorption process. Zero-valent iron reduced hexavalent chromium to trivalent chromium, while the valence state of zero-valent iron increased, and the structure of formed ferric chromate cannot be oxidized again, so the weight increase is not significant.
Table 2 Fitting comparison between pseudo-first-order kinetic model and pseudo-second-order kinetic model at different temperatures
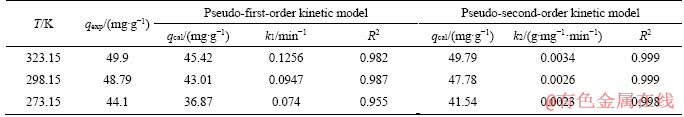
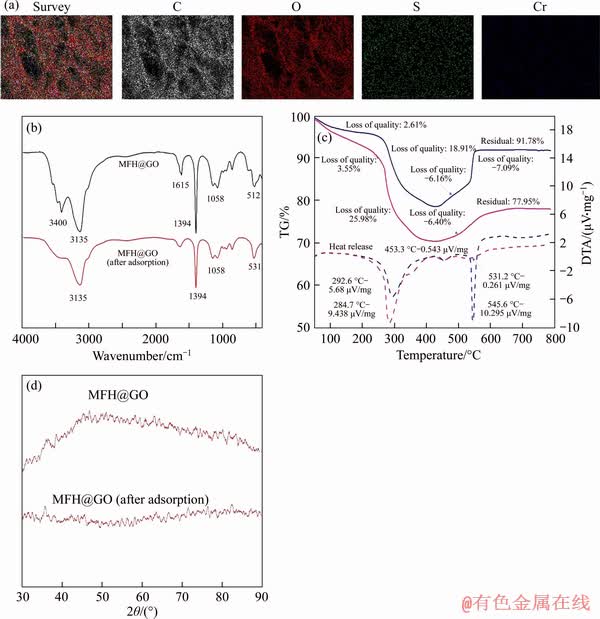
Figure 7 Survey, C, O, S and Cr corresponding elemental mapping images of MFH@GO-Cr(VI) species (a), FTIR spectra of samples (b), TG-DTA spectra of samples (c) and XRD pattern of samples (d)
XRD spectra of MFH@GO and MFH@GO composites after adsorption are shown in Figure 7(d). The characteristic diffraction peaks of zero-valent iron appeared near 2��=44.6��, which may be due to the low diffraction intensity of the composite coated by graphene oxide. After the adsorption, the curves of the composites show diffraction peaks of iron oxides and iron hydroxides near 2��=35.5�� and 2��=39.5��, respectively. The peak value of zero-valent iron decreases, which indicates that zero-valent iron may deposit on the surface of MFH@GO after oxidation reaction to form iron oxides or iron hydroxides. In addition, new diffraction peak appeared near 2��=53.5��, which belongs to FeCr2O4 [38]. It is presumed that Cr (VI) deposited in the form of Fe-Cr oxides after the reduction of Cr (VI) by MFH@GO [39].
4 Conclusions
MFH@GO with the porous plume-like structure was prepared by loading magnetic nanomaterials on mycelium carriers. It is an environmentally friendly and low-cost method for designing a green and recyclable nanocomposite, which provides another way of thinking for the preparation of nanocomposites. The MFH@GO showed a great adsorption capacity of Cr(VI), which is mainly owing to the carboxyl and hydroxyl groups on its surface. Furthermore, MFH@GO is magnetic and easy to separate, and has the advantages of simple synthesis process and excellent regeneration performance. This study confirmed that the prepared bio-nanocomposite MFH@GO is a promising and valuable biosorbent for removing Cr(VI) from contaminated water.
References
[1] WANG Yang-yang, LIU Yi-dan, ZHAN Wen-hao, NIU Liu-min, ZOU Xue-yan, ZHANG Chao-sheng, RUAN Xin-ling. A field experiment on stabilization of Cd in contaminated soils by surface-modified nano-silica (SMNS) and its phyto-availability to corn and wheat [J]. Journal of Soils and Sediments, 2020, 20: 91-98. DOI: 10.1007/ s11368-019-02416-1.
[2] WANG Yang-yang, LIU Yi-dan, ZHAN Wen-hao, ZHENG Kai-xuan, LIAN Ming-ming, ZHANG Chao-sheng, RUAN Xin-ling, LI Tao. Long-term stabilization of Cd in agricultural soil using mercapto-functionalized nano-silica (MPTS/nano-silica): A three-year field study [J]. Ecotoxicology and Environmental Safety, 2020, 197: 110600. DOI: 10.1016/j.ecoenv.2020.110600.
[3] SINGH P K, WANG W, SHRIVASTAVA A K. Cadmium- mediated morphological, biochemical and physiological tuning in three different Anabaena species [J]. Aquatic Toxicology, 2018, 202: 36-45. DOI: 10.1016/j.aquatox.2018. 06.011.
[4] CHAI Li-yuan, DING Chun-lian, TANG Chong-jian, YANG Wei-chun, YANG Zhi-hui, WANG Yang-yang, LIAO Qi, LI Jia-wei. Discerning three novel chromate reduce and transport genes of highly efficient Pannonibacter phragmitetus BB: From genome to gene and protein [J]. Ecotoxicology and Environmental Safety, 2018, 162: 139-146. DOI: 10.1016/j.ecoenv.2018.06.090.
[5] MA Ya-meng, LI Fang-fang, YANG Wei-hua, LV Lv, XUE Hao-tian, WANG Yang-yang. Remediation of Cr(VI)- contaminated soil using the acidified hydrazine hydrate [J]. Bulletin of Environmental Contamination and Toxicology, 2016, 97(3): 392-394. DOI: 10.1007/s00128-016-1862-z.
[6] WANG Yang-yang, PENG Bing, YANG Zhi-hui, CHAI Li-yuan, LIAO Qi, ZHANG Zhi, LI Chuang. Bacterial community dynamics during bioremediation of Cr(VI)- contaminated soil [J]. Applied Soil Ecology, 2015, 85: 50-55. DOI: 10.1016/j.apsoil.2014.09.002.
[7] MDLALOSE L, BALOGUN M, SETLEGO K, WUKULULA M, CHIMUKA L, CHETTY A. Synthesis, characterization and optimization of poly (p-phenylenediamine)-based organoclay composite for Cr(VI) remediation [J]. Applied Clay Science, 2017, 139: 72-80. DOI: 10.1016/j.clay.2017.01.015.
[8] WANG Yang-yang, CHAI Li-yuan, LIAO Qi, TANG Chong-jian, LIAO Ying-ping, PENG Bing, YANG Zhi-hui. Structural and genetic diversity of hexavalent chromium- resistant bacteria in contaminated soil [J]. Geomicrobiology Journal, 2016, 33(3, 4): 222-229. DOI: 10.1080/01490451. 2015.1054006.
[9] OWLAD M, AROUA M K, DAUD W A W, BAROUTIAN S. Removal of hexavalent chromium-contaminated water and wastewater: A review [J]. Water Air and Soil Pollution, 2009, 200(1-4): 59-77. DOI: 10.1007/s11270-008-9893-7.
[10] SINGH R, MISRA V, SINGH R P. Removal of Cr(VI) by Nanoscale zero-valent iron (nZVI) from soil contaminated with tannery wastes [J]. Bulletin of Environmental Contamination and Toxicology, 2012, 88(2): 210-214. DOI: 10.1007/s00128-011-0425-6.
[11] MIN Xiao-bo, WANG Yang-yang, CHAI Li-yuan, YANG Zhi-hui, LIAO Qi. High-resolution analyses reveal structural diversity patterns of microbial communities in chromite ore processing residue (COPR) contaminated soils [J]. Chemosphere, 2017, 183: 266-276. DOI: 10.1016/ j.chemosphere.2017.05.105.
[12] LI Yao, CUI Wen-quan, LIU Li, ZONG Rui-long, YAO Wen-qing, LIANG Ying-hua, ZHU Yong-fa. Removal of Cr (VI) by 3D TiO2-graphene hydrogel via adsorption enriched with photocatalytic reduction [J]. Applied Catalysis B: Environmental, 2016, 199: 412-423. DOI: 10.1016/j.apcatb. 2016.06.053.
[13] ZAFAR S, AQIL F, AHMAD I. Metal tolerance and biosorption potential of filamentous fungi isolated from metal contaminated agricultural soil [J]. Bioresource Technology, 2007, 98(13): 2557-2561. DOI: 10.1016/ j.biortech.2006.09.051.
[14] WANG Yang-yang, LIU Yi-dan, ZHAN Wen-hao, ZHENG Kai-xuan, WANG Jun-nan, ZHANG Chao-sheng, CHEN Run-hua. Stabilization of heavy metal-contaminated soils by biochar: challenges and recommendations [J]. Science of the Total Environment, 2020, 30: 139060. DOI: 10.1016/ j.scitotenv.2020.139060.
[15] LIU Wei-fang, ZHANG Jian, ZHANG Cheng-lu, WANG Yi-fu, LI Ye. Adsorptive removal of Cr(VI) by Fe-modified activated carbon prepared from Trapa natans husk [J]. Chemical Engineering Journal, 2010, 162(2): 677-684. DOI: 10.1016/j.cej.2010.06.020.
[16] ZENG Yue-bin, WOO H, LEE G, PARK J. Removal of chromate from water using surfactant modified Pohang clinoptilolite and Haruna chabazite [J]. Desalination, 2010, 257(1-3): 102-109. DOI: 10.1016/j.desal.2010.02.039.
[17] KIM Y J, CHOI J, KIM E S. Removal of heavy metal ion in wastewater by dendritic nano-structured complex material [J]. Journal of Nanoscience and Nanotechnology, 2019, 19(2): 1010-1013. DOI: 10.1166/jnn.2019.15960.
[18] WANG Yang-yang, ZHAN Wen-hao, ZHENG Kai-xuan, LIU Yi-dan, ZOU Xue-yan, ZHANG Chao-sheng, RUAN Xin-ling. Effect of surface-modified nano-silica on the mobility and fraction of Cd in contaminated agricultural soils [J]. Soil and Sediment Contamination: An International Journal, 2020, 29(1): 96-106. DOI: 10.1080/15320383. 2019.1679086.
[19] LIU Xue-ming, SONG Kai-nan, LIU Wei-zhen, XIONG Yue-cheng, XU Yun-yun, SHI Zheng-qing, ZHAO Dong-ye, LIN Zhang. Removal and recovery of Pb from wastewater through a reversible phase transformation process between nano-flower-like Mg(OH)2 and soluble Mg(HCO3)2 [J]. Environmental Science: Nano, 2019, 6(2): 467-477. DOI: 10.1039/C8EN01173G.
[20] DINDA D, GUPTA A, SAHA S K. Removal of toxic Cr(VI) by UV-active functionalized graphene oxide for water purification [J]. Journal of Materials Chemistry A, 2013, 1(37): 11221-11228. DOI: 10.1039/C3TA12504A.
[21] ZHANG Ke-xin, LI Hai-yan, XU Xing-jian, YU Hong-wen. Synthesis of reduced graphene oxide/NiO nanocomposites for the removal of Cr(VI) from aqueous water by adsorption [J]. Microporous and Mesoporous Materials, 2018, 255: 7-14. DOI: 10.1016/j.micromeso.2017.07.037.
[22] WANG Ting, ZHANG Li-yuan, LI Chao-fang, YANG Wei-chun, SONG Ting-ting, TANG Chong-jian, MENG Yun, DAI Shuo, WANG Hai-ying, CHAI Li-yuan, LUO Jian. Synthesis of core�Cshell magnetic Fe3O4@ poly (m-phenylenediamine) particles for chromium reduction and adsorption [J]. Environmental Science & Technology, 2015, 49(9): 5654-5662. DOI: 10.1021/es5061275.
[23] MEHTA D, MAZUMDAR S, SINGH S K. Magnetic adsorbents for the treatment of water/wastewater��A review [J]. Journal of Water Process Engineering, 2015, 7: 244-265. DOI: 10.1016/j.jwpe.2015.07.001.
[24] LI WEI, MU Bing-nan, YANG Yi-qi. Feasibility of industrial-scale treatment of dye wastewater via bio-adsorption technology [J]. Bioresource Technology, 2019, 227: 157-170. DOI: 10.1016/j.biortech.2019.01.002.
[25] BAHRAMI H R T, AZIZI A, SAFFARI H. Dropwise condensation heat transfer enhancement on surfaces micro/nano structured by a two-step electrodeposition process [J]. Journal of Central South University, 2019, 26(5): 1065-1076. DOI: 10.1007/s11771-019-4071-1.
[26] ZHANG Li-yuan, WANG Yang-yang, PENG Bing, YU Wan-ting, WANG Hai-ying, WANG Ting, DENG Bai-wan, CHAI Li-yuan, ZHANG Kai, WANG Jie-xi. Preparation of a macroscopic, robust carbon-fiber monolith from filamentous fungi and its application in Li�CS batteries [J]. Green Chemistry, 2014, 16(8): 3926-3934. DOI: 10.1039/ c4gc00761a.
[27] LI Qing-zhu, CHAI Li-yuan, YANG Zhi-hui, WANG Qing-wei. Kinetics and thermodynamics of Pb(II) adsorption onto modified spent grain from aqueous solutions [J]. Applied Surface Science, 2009, 255(7): 4298-4303. DOI: 10.1016/j.apsusc.2008.11.024.
[28] KONG Miao-miao, SONG Hui, LI Feng-hua, DAI Dong-mei, GAO Hong-tao. Facile synthesis of Bi2Fe4O9 nanoplate and its application as a novel adsorbent for Cu(II) removal [J]. Journal of Environmental Chemical Engineering, 2017, 5(1): 69-78. DOI: 10.1016/j.jece.2016.11.020.
[29] DONG Hao-ran, DENG Jun-min, XIE Yan-kai, ZHANG Cong, JIANG Zhao, CHENG Yu-jun, HOU Kun-jie, ZENG Guang-ming. Stabilization of nanoscale zero-valent iron (nZVI) with modified biochar for Cr(VI) removal from aqueous solution [J]. Journal of Hazardous Materials, 2017, 332(15): 79-86. DOI: 10.1016/j.jhazmat.2017.03.002.
[30] WANG Lin, SONG Huan, YUAN Li-yong, LI Zi-jie, ZHANG Peng, GIBSON J K, ZHENG Li-rong, WANG Hong-qing, CHAI Zhi-fang, SHI Wei-qun. Effective removal of anionic Re (VII) by surface-modified Ti2CTx MXene nanocomposites: Implications for Tc (VII) sequestration [J]. Environmental Science & Technology, 2019, 53(7): 3739-3747. DOI: 10.1021/acs.est.8b07083.
[31] SHI Li-na, LIN Yu-man, ZHANG Xin, CHEN Zu-liang. Synthesis, characterization and kinetics of bentonite supported nZVI for the removal of Cr(VI) from aqueous solution [J]. Chemical Engineering Journal, 2011, 171(2): 612-617. DOI: 10.1016/j.cej.2011.04.038.
[32] WANG Yan, TANG Xiao-wu, WANG Heng. Characteristics and mechanisms of Ni (II) removal from aqueous solution by Chinese loess [J]. Journal of Central South University, 2015, 22(11): 4184-4192. DOI: 10.1007/s11771-015-2966-z.
[33] HUANG Guan-xing, ZHANG Ying, SUN Ji-chao, JING Ji-hong, LIU Jing-tao, WANG Ying. Effects of different conditions on Pb2+ adsorption from soil by irrigation of sewage in South China [J]. Journal of Central South University, 2012, 19(1): 213-221. DOI: 10.1007/s11771- 012-0994-5.
[34] JIANG Zhu-wu, WANG Xue-jiang, NING Xue. Removal of lead (II) from aqueous solutions by activated carbon developed from surplus sludge [J]. Journal of Central South University, 2014, 21(9): 3568-3574. DOI: 10.1007/s11771- 014-2337-1.
[35] HOJATI S, KHADEMI H. Cadmium sorption from aqueous solutions onto Iranian sepiolite: Kinetics and isotherms [J]. Journal of Central South University, 2013, 20(12): 3627- 3632. DOI: 10.1007/s11771-013-1889-9.
[36] RAO M P, MUSTHAFA S, WU J J, ANANDAN S. Facile synthesis of perovskite LaFeO3 ferroelectric nanostructures for heavy metal ion removal applications [J]. Materials Chemistry and Physics, 2019, 232(15): 200-204. DOI: 10.1016/j.matchemphys.2019.04.086.
[37] LIU Chong, WU Tong, HSU Po-chun, XIE Jin, ZHAO Jie, LIU Kai, SUN Jie, XU Jin-wei, TANG Jing, YE Zi-wen, LIN Ding-chang, CUI Yi. Direct/alternating current electrochemical method for removing and recovering heavy metal from water using graphene oxide electrode [J]. ACS Nano, 2019, 13(6): 6431-6437. DOI: 10.1021/ acsnano.8b09301.
[38] LIU Jing, LI Ruo-fan, YAO Yi-hao, LIU Ai-rong. Fate and mechanistic insights into the transformation of aged nanoscale zero-valent iron (nZVIA) reacted with Cr(VI): Impact of aging time in oxic water [J]. ACS Earth and Space Chemistry, 2019, 3(7): 1288-1295. DOI: 10.1021/ acsearthspacechem.9b00095.
[39] ZHANG Bao, WANG Yin-hui, WANG Shao-peng, WEI Chao-shuai, WANG Rui, ZHANG Wei. Oxidation of high iron content electroplating sludge in supercritical water: stabilization of zinc and chromium [J]. Environmental Science and Pollution Research, 2019, 26(15): 15001-15010. DOI: 10.1007/s11356-019-04897-6.
(Edited by YANG Hua)
���ĵ���
���Թ��ܻ���˿���ײ��ϵ��Ʊ������Чȥ����ˮ�е�Cr(VI)
ժҪ�����IJ���һ�ּ��º͵ķ����Ʊ��˸������ܻ��ŵĴ��Ծ�˿���ϲ���MFH@GO�������ڴ�����ˮ�е�Cr(VI)����ϸ���о���MFH@GO��Cr(VI)���������ܡ������������pHֵΪ5.0���¶�Ϊ323.15 K�������£�MFH@GO��Cr(VI)������������ﵽ58.4 mg/g��Զ����δ���Եľ�˿�ۺ�����ʯīϩ��ͨ��FTIR��XRD��TG-DTA��SEM��EDX�ȷ���MFH@GO�����Լ���Cr(VI)�����������������������������ʯīϩ���������������˾�˿���������ԣ�MFH@GO�ܹ���Cr(VI)��ԭ�����ĸ���������������ڲ������С����о��������MFH@GO�ڴ���Cr(VI)��ˮ�о��нϴ��DZ����Ӧ�ü�ֵ��
�ؼ��ʣ���ˮ�����۸�����˿�����ϲ��ϣ������
Foundation item: Project(18B195) supported by Excellent Youth Project of Hunan Education Department, China; Projects(51804353, 51704093) supported by the National Natural Science Foundation of China; Project(kq1801074) supported by Key Projects of Changsha Science and Technology Plan, China; Project(2018JJ4010) supported by Hunan Provincial Natural Science Foundation of China (Joint Funds of Provincial and Zhuzhou Municipal); Project(2018JJ3885) supported by Natural Science Foundation of Hunan Province of China (Science Foundation for Youths)
Received date: 2019-06-28; Accepted date: 2020-04-01
Corresponding author: WANG Yang-yang, PhD, Associated Professor; Tel/Fax: +86-15194602157; E-mail: wangyangyangxyz@ 163.com; ORCID: 0000-0003-1519-8796

