J. Cent. South Univ. Technol. (2007)03-0348-05
DOI: 10.1007/s11771-007-0069-1

Morphology and conductivit
y of in-situ PEO-LiClO4-TiO2
composite polymer electrolyte
PAN Chun-yue(潘春跃), FENG Qing(冯 庆), WANG Li-jun(王丽君),
ZHANG Qian(张 倩), CHAO Meng(巢 猛)
(School of Chemistry and Chemical Engineering, Central South University, Changsha 410083, China)
Abstract: PEO-LiClO4-TiO2 composite polymer electrolyte films were prepared. TiO2 was formed directly in matrix by hydrolysis and condensation reaction of tetrabutyl titanate. The crystallinity, morphology and ionic conductivity of composite polymer electrolyte films were examined by differential scanning calorimetry, scanning electron microscopy, atom force microscopy and alternating current impedance spectroscopy, respectively. The glass transition temperature and the crystallinity of composite polymer electrolytes are decreased compared with those of PEO-LiClO4 polymer electrolyte film. The results show that TiO2 particles are uniformly dispersed in PEO-LiClO4-5%TiO2 composite polymer electrolyte film. The maximal conductivity of 5.5×10-5 S/cm at 20 ℃ of PEO-LiClO4-TiO2 film is obtained at 5% mass fraction of TiO2.
Key words: polyethylene oxide (PEO); TiO2; composite polymer electrolyte; in-situ composite; conductivity
1 Introduction
Ionic conducting solid polymer electrolytes are increasingly used in the design of high energy batteries, electrochemical devices and chemical sensors because of the relative ease in fabricating compact and light mass devices with enhanced safety. An exemplary solid polymer electrolyte is a poly(ethylene oxide) (PEO) system containing lithium salts[1-4]. However, the polymers in such system tend to crystallize, resulting in low room-temperature ionic conductivity unsatisfied for practical application. Two approaches have been developed to address this problem, that is, incorporation of plasticizers[5-6] and ceramic fillers[7-8]. Especially the latter brings about improvement in the ionic conductivity, mechanical properties and interfacial stability with lithium electrode[9].
An extensive literature survey[10-12] reveals that almost all polymer-ceramic nanocomposites were prepared through mechanical blending of nanoscale ceramic particles, polymer, and salt in a compatible solvent. However, aggregation of these particles stemming from their high surface energy is a perennial problem that undermines the efficacy of the ceramic fillers. The particle size of the ceramic fillers is increased because of the aggregation of these particles, which leads to worsening of the electrolyte performance. The method of sol-gel process that overcomes such a problem has been widely used for preparing the polymer-ceramic composite materials. The nanosize ceramic fillers are precipitated in situ in the polymer matrix through a series of hydrolysis and condensation reactions of suitable precursors. Thus the ceramic fillers are uniformly distributed in the polymer and exhibit excellent properties. However very few studies have been published using this method for the preparation of composite polymer electrolytes[13]. In this study, PEO-LiClO4-TiO2 composite polymer electrolyte films were prepared. TiO2 was formed directly in situ in matrix by hydrolysis and condensation reaction of tetrabutyl titanate. The morphology and ionic conductivity of the composite polymer electrolyte films were examined by differential scanning calorimetry(DSC), scanning electron microscopy (SEM), atom force microscopy (AFM) and alternating current (AC) impedance spectroscopy, respectively.
2 Experimental
2.1 Materials
Polyethylene oxide (PEO, relative molecule mass 600 000) was obtained from Aldrich Chemicals and dried at 50 ℃ for 24 h under vacuum before use. Lithium perchlorate (LiClO4) was analytically pure and dried at 120 ℃ for 24 h under vacuum to remove crystal-water. Acetonitrile (99.5%, mass fraction) was used as the solvent in the film-casting process. Tetrabutyl titanate (Ti(OC4H9)4) was analytically pure.
2.2 Preparation of composite polymer electrolyte
films
PEO and LiClO4 were dissolved in acetonitrile and the mixture was stirred at room temperature for 3 h to obtain a homogeneous solution. The concentration of PEO in acetonitrile was maintained at 5%(mass fraction) for all solutions. The molar ratio of EO to Li was fixed at 8:1 for all the samples. A calculated amount of tetrabutyl titanate was added into the solution under continuous stirring. The TiO2 contents of the composite polymer electrolyte films were calculated by assuming complete conversion of tetrabutyl titanate into TiO2. The residual water in acetonitrile was adequate for hydrolysis, and no water was deliberately added to the mixture. The mixture was vigorously stirred for another 3 h before it was transferred to a Teflon dish to allow the solvents to evaporate. The composite polymer electrolyte films were further dried at 40℃ under vacuum for 2 d to completely remove the residual solvent.
2.3 Measurements of composite polymer electrolyte
films
DSC was performed by use of a Q-10 analyzer (TA, USA). Detailed surface images of the composite polymer electrolyte films were obtained by means of a KYKY-2800 scanning electron microscope (SEM) at 20 kV and Dimension-3000 atom force microscope (AFM) (DI, USA). AC impedance spectroscopy of the composite polymer electrolyte films was measured by use of CHI660 electrochemical workstation (Chenhua, Shanghai, China) between 1 Hz and 1×105 Hz with 30 mV amplitude. The films were sandwiched between two stainless steel electrodes and assembled into a tightly sealed test cell.
3 Results and discussion
3.1 Analysis of DSC for composite polymer electrolyte films
The DSC curves of the PEO-LiClO4 and the PEO-LiClO4-5%TiO2 composite polymer electrolyte films are shown in Fig.1. Firstly about 10 mg of samples were sealed in standard aluminum cans and cooled in a nitrogen atmosphere from room temperature to -60 ℃. The first heating curve was recorded when the samples were heated from -60 ℃ to 100 ℃ at the rate of 10 ℃/min.
The two DSC curves in Fig.1 show obvious transition at low temperature region assigned to the glass transition temperature (tg) of composite polymer electrolyte films. All the samples exhibit a relatively sharp endothermic peak near 45 ℃, which could be attributed to the melting of the crystalline region of PEO. The crystallinity (χc) of composite polymer electrolyte films can be obtained by comparing the heat of melting (ΔHm) with that (213.7 J/g)[14] of 100% crystalline PEO. The important data obtained from the first heating curves are summarized in Table 1.
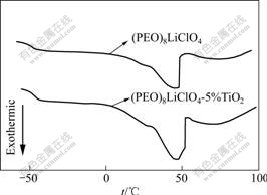
Fig.1 DSC curves of composite polymer electrolyte films
Table 1 Data of DSC curves for PEO-LiClO4-TiO2 composite polymer electrolyte films
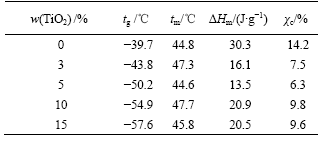
It is found that the addition of TiO2 causes obvious decrease in tg of composite polymer electrolyte films. This observation can be rationalized in terms of hydrogen bonding between the surface hydroxyl groups of TiO2 particles and the ether oxygen atoms in the polymer chains. This weakens the complexation between Li+ and these ether oxygen atoms, facilitates the migration of Li+, perturbs the PEO chain conformation and introduces additional free space among the polymer segments, thus yielding a lower tg. Furthermore, the crystallinity (χc) of composite polymer electrolyte films decreases after the addition of TiO2 and the value for PEO-LiClO4-5%TiO2 composite polymer electrolyte film is the least. They all indicate that the polymer segmental motion can be increased and it is beneficial to ion conduction.
After the first heating, the samples were cooled to -60 ℃ at 20 ℃/ min. The second heating curve was recorded when the samples were heated from -60 ℃ to 100 ℃ at the rate of 10 ℃/ min again. As seen from Fig.2, The value of tg for the second heating curve is increased from -50.2 ℃ to -33.9 ℃, indicating that there is a marked effect on tg for the thermal history of the samples. On the other hand, it perhaps should be noted that an exothermic peak is observed at 25.9 ℃ for the second heating curve, which is assigned to the cold crystalline phenomena of PEO. This phenomenon is attributed to the fact that the cooling rate at 20 ℃/ min is too rapid for PEO to form crystal and the crystalline region appears when the samples are secondly heated.
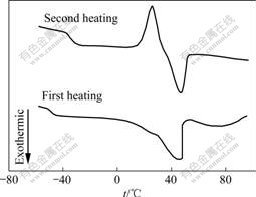
Fig.2 DSC curves for PEO-LiClO4-5%TiO2composite polymer electrolyte film
3.2 Morphology of composite polymer electrolyte films
The SEM images of PEO/LiClO4 polymer electrolyte films (Fig.3(a)) and PEO-LiClO4-5%TiO2 composite polymer electrolyte film (Figs.3(b) and (c)) are shown in Fig.3.
The surface is smooth and there are no holes detected in Fig.3(a). It can be seen that TiO2 particles are uniformly dispersed in the polymer matrix in Fig.3(b). Fig.3(c) is partially magnified image. We can see that the diameter of TiO2 particles is about 300 nm.
AFM was also used to characterize the film surface morphology of the solid polymer electrolytes (SPEs)[15]. Fig.4 presents 5 ?m×5 ?m surface plot of PEO-LiClO4- 5%TiO2 composite polymer electrolyte film (three- dimensional representation, Fig.4(b)) together with top view of the films (Fig.4(a)).
As seen from Fig.4, many nanoparticles TiO2 are homogeneously dispersed on the surface. The max perpendicular deviation is about 2 nm in Fig.4(b). The interface of the TiO2 particles and the polymer matrix is very illegible, that is to say, they are well consistent.
3.3 Conductivity of composite polymer electrolyte films
Fig.5 shows the AC impedance spectra of PEO-LiClO4 and PEO-LiClO4-5%TiO2 composite polymer electrolyte films. The point of intersection between the arc in the high frequency range and the

Fig.3 SEM images of composite polymer electrolyte films (a) PEO-LiClO4; (b), (c) PEO-LiClO4-5%TiO2
straight line in the low frequency range coincides with the bulk resistance of composite polymer electrolyte films. In our experiment, the semicircles in high frequency range are not observed, which may be concluded that the frequency is not high enough. The conductivity of the samples can be calculated by the following equation:
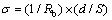 (1)
(1)
where σ is the conductivity, d and S represent the thickness and the area of composite polymer electrolyte films, and Rb is the bulk resistance. The conductivity of PEO-LiClO4-TiO2 composite polymer electrolyte films is shown in Fig.6 and Fig.7.

Fig.4 Top view (a) and three-dimensional surface plot (b) AFM images of PEO-LiClO4-5%TiO2 composite polymer electrolyte film

Fig.5 AC impedance spectra of composite polymer electrolyte films
(a) PEO-LiClO4; (b) PEO-LiClO4-5%TiO2
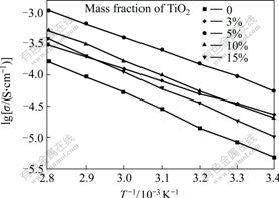
Fig.6 Dependence of conductivity on temperature for PEO-LiClO4-TiO2 composite polymer electrolyte
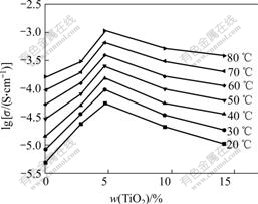
Fig.7 Conductivity of PEO-LiClO4-TiO2 composite polymer electrolyte films at different TiO2 contents
Fig.6 shows the temperature dependency of the conductivity for composite polymer electrolyte films. The plot of lg σ versus 1000/T is approximately linear, suggesting the Arrhenius equation could be a good fit for the experimental data:
σ=σ0exp[-Ea/(kT)] (2)
where σ0 is the conductivity, k is the conductivity pre-exponential factor and Ea is the activation energy for conduction. The activation energies for PEO-LiClO4- TiO2 composite polymer electrolyte are listed in Table 2.
Table 2 Activation energies of PEO-LiClO4-TiO2 composite polymer electrolyte films

The conductivity of an electrolyte is determined by the product of the concentration of ion charge carriers and their mobility:
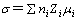 (3)
(3)
where Zi is the charge on the ions, ni is the number of charge carriers, and μi is the ionic mobility. Conductivity enhancement could be realized by increasing either the charge carrier number or their mobility. Table 2 shows clearly that the addition of TiO2 decreases the activation of the composite polymer electrolyte films, indicating the increased mobility of the ions μi. It is attributed to the fact that the addition of TiO2 decreases tg and χc of composite polymer electrolyte films from the DSC results, enhancing the movement of polymer chain and facilitating the migration of Li+. It is also generally believed that there are interactions among TiO2, Li salt and polymer chains[16-18]. Such interactions could increase the effective charge carrier number ni. Because both the ion mobility and the number of charge carrier are increased after the addition of TiO2, the conductivity is higher in PEO-LiClO4-TiO2 composite polymer electrolyte films.
As seen from Fig.7, the conductivity increases with increasing the TiO2 content and attains a maximum value when the mass fraction of TiO2 is 5%. Subsequently, the conductivity decreases with further increasing the TiO2 content. This could be understood in terms of badly dispersion of TiO2 particles, resulting in conduction paths being blocked. The maximum values of conductivity for PEO-LiClO4-5%TiO2 composite polymer electrolyte film are 5.5×10-5 S/cm and 1.1×10-3 S/cm, respectively, at 20 ℃ and 80 ℃, while the conductivity values of polymer electrolyte without TiO2 are about 4.7×10-6 S/cm and 1.5×10-4 S/cm at 20 ℃ and 80 ℃, respectively.
4 Conclusions
1) Nanocomposite polymer electrolytes based on PEO-LiClO4 system are synthesized, in which the ceramic filler, TiO2, is formed in-situ within the polymer matrix by a sol-gel process. Differential scanning calorimetry confirms the suppression of polymer crystallization in the presence of TiO2. The glass transition temperature (tg) and the crystallinity (χc) of composite polymer electrolyte films all decrease after the addition of TiO2.
2) SEM and AFM results show that polymer and the ceramic fillers are uniformly mixed at the nanometer level. The average size of TiO2 particles is about 300 nm.
3) Electrochemical measurements show higher ionic conductivity is obtained when the in-situ formed TiO2 is used. The conductivity shows a maximum value of 5.5×10-5 S/cm at 20 ℃ when the mass fraction of TiO2 is 5%, which is an order of magnitude higher than that in the absence of TiO2.
References
[1] AHN J H, WANG G X, LIU H K, et al. Nanoparticle-dispersed PEO polymer electrolytes for Li batteries[J]. J Power Sources, 2003, 119/121: 422-426.
[2] JI K S, MOON H S, KIM J W, et al. Role of functional nano- sized inorganic fillers in poly(ethylene) oxide-based polymer electrolytes[J]. J Power Sources, 2003, 117: 124-130.
[3] SUAREZ S, ABBRENT S, GREENBAUM S G, et al. Effect of nanosized SiO2 on the transport properties of solventless P(EO)20- LiBeTi polymer electrolytes: A solid-state NMR study[J]. Solid State Ionics, 2004, 166: 407-415.
[4] SHIN J H, PASSERINI S. Effect of fillers on the electrochemical and interfacial properties of PEO-LiN(SO2CF2CF3)2 polymer electrolytes[J]. Electrochimica Acta, 2004, 49: 1605-1612.
[5] KIM Y T, SMOTKIN E S. The effect of plasticizers on transport and electrochemical properties of PEO-based electrolytes for lithium rechargeable batteries[J]. Solid State Ionics, 2002, 149: 29-37.
[6] YANG X Q, LEE H S, et al. Development of a new plasticizer for poly(ethylene oxide)-based polymer electrolyte and the investigation of their ion-pair dissociation effect[J]. J Power Sources, 1995, 54: 198-204.
[7] CROCE F, APPETECCHI G B, PERSI L, et al. Nanocomposite polymer electrolytes for lithium batteries[J]. Nature, 1998, 394: 456-458.
[8] CROCE F, PERSI L, SCROSATI B, et al. Role of the ceramic fillers in enhancing the transport properties of composite polymer electrolytes[J]. Electrochimica Acta, 2001, 46: 2457-2461.
[9] LI Zhao-hui, SU Guang-yao, WANG Xia-yu, et al. Ionic conductivity study of PVF-HFP composite electrolyte filled with Al2O3 nanoparticles[J]. Chem J Chinese Universities, 2003, 24: 2065-2068. (in Chinese).
[10] CAPIGLIA C, MUSTARELLI P, QUARTARONE E, et al. Effects of nanoscale SiO2 on the thermal and transport properties of solvent-free, poly(ethylene oxide) (PEO)-based polymer electrolytes[J]. Solid State Ionics, 1999, 118: 73-79.
[11] CHU P P, REDDY M J. Sm2O3 composite PEO solid polymer electrolyte[J]. J Power Sources, 2003, 115: 288-294.
[12] WALLS H J, ZHOU Jian, YERIAN J A, et al. Fumed silica-based composite polymer electrolytes: Synthesis, rheology, and electrochemistry[J]. J Power Sources, 2000, 89: 156-162.
[13] LIU Y, LEE J Y, HONG L. In situ preparation of poly(ethylene oxide)-SiO2 composite polymer electrolytes[J]. J Power Sources, 2004, 129: 303-311.
[14] QIAN X M, GU N Y, CHENG Z L, et al. Impedance study of (PEO)10LiClO4-Al2O3 composite polymer electrolyte with blocking electrodes[J]. Electrochimica Acta, 2001, 46: 1829-1836.
[15] WANG Ming-kui, ZHAO Feng, DONG Shao-jun. A single ionic conductor based on Nafion and its electrochemical properties used as lithium polymer electrolyte[J]. J Phys Chem, 2004, 108: 1365-1370.
[16] CHUNG S H, WANG Y, PERSI L, et al. Enhancement of ion transport in polymer electrolytes by addition of nanoscale inorganic oxides[J]. J Power Sources, 2001, 97/98: 644-648.
[17] JOHANSSON P, JACOBSSON P. TiO2 nano-particles in polymer electrolytes: surface interactions[J]. Solid State Ionics, 2004, 170: 73-78.
[18] BLAOISE A C, DONOSO J P, MAGON C J, et al. NMR and conductivity study of PEO-based composite polymer electrolytes[J]. Electrochimica Acta, 2003, 48: 2239-2246.
(Edited by YANG Bing)
Received date: 2006-08-24; Accepted date: 2006-09-27
Corresponding author: PAN Chun-yue, Professor; Tel: +86-731-8836961; E-mail: Panchunyue@sina.com