
Protection behavior of SO2-containing cover gases to molten magnesium alloy
WANG Xian-fei, XIONG Shou-mei
State Key Laboratory of Automotive Safety and Energy,
Department of Mechanical Engineering, Tsinghua University, Beijing 100084, China
Received 25 September 2010; accepted 26 December 2010
Abstract: Sulphur dioxide (SO2) mixed with carrier gases was used as an alternative to SF6 to protect molten magnesium alloys. The protection behavior of AZ91D alloy in a sealed melting furnace was investigated under the atmosphere containing SO2 with different mixed gases. The morphology and composition of the surface film were studied. The melt was well protected in an atmosphere of SO2 and a proper amount of air, and was not protected properly in the other atmospheres. Based on the understanding of the protective effects of SO2 in the sealed furnace, the protection mechanism of SO2-containing cover gases on molten AZ91D alloy was studied in an open melting furnace. The cover gas protected the melt by reacting with the melt to form a coherent protective film with a network structure on the melt surface. The film contains MgO and MgS. MgS increases the Pilling and Bedworth ratio of the surface film and enhances the protective capability of the films.
Key words: magnesium alloy; SO2; melt protection; surface film
1 Introduction
Magnesium and its alloys are being widely used in many areas due to their excellent properties such as high specific strength and low density[1]. However, molten magnesium and its alloys oxidize rapidly in air, and magnesium oxide film does not protect the molten metal from further oxidation[2]. The cover gases containing SF6 mixed with N2 or dry air are commonly used to protect molten magnesium, but the SF6 gas has become recognized as a very potent greenhouse gas with a high global warming potential value of 23 900 compared with CO2 and a long atmospheric lifetime of 3 200 years[3]. Therefore, the magnesium industry has been looking for new substitutes to SF6[4-5]. Studying the protective mechanism is very important for finding replacements.
FRUEHLING and HANAWALT[6] firstly studied the protection effect of molten magnesium under protective atmosphere of SF6 containing cover gases, and found that SF6 was consumed in the protection process, and attributed this to the absorption of SF6 to MgO surface. CASHION[7] reported that the surface film formed in protective atmosphere containing SF6 was a mixture of MgO and MgF2 phases and the film thickness varied between 1 and 2 um. PETTERSEN et al[8] analyzed the microstructure characteristic of the surface film formed in F-containing cover gas and found that long-term exposure of magnesium melt increased the film thickness with an increased concentration of MgF2. AARSTAD[9] indicated that particles containing MgF2 formed at the interface of MgO film and bulk magnesium, which was confirmed by XIONG and LIU[10], and the particles grew up until they covered 25%-50% of the total surface area.
The oxidation rate of pure iron and 304L steel can be slowed down by traces of SO2 in O2 and O2+40%H2O[11-12]. Sulfur dioxide mixed with carrier gases can also be used to protect molten magnesium alloy[13]. But the protection mechanism of SO2-containing cover gases to molten magnesium alloy has not been clearly understood. In this work, the protection behavior of AZ91D alloy in a sealed melting furnace was firstly investigated under the atmospheres containing SO2 with different mixed gases. Then, the protection mechanism of SO2-containing cover gases to molten AZ91D alloy was studied in an open melting furnace.
2 Experimental
2.1 Experimental method in sealed furnace
Pressure in the furnace can be pumped to below 6 Pa and the pressure rising rate is below 0.67 Pa/h. Firstly, the pressure in the furnace was pumped to a vacuum level of 6 Pa, and then, the gas mixture of SO2 and N2 or air controlled by flow meters was filled into the furnace. Finally, the gas inlet valve and the flow meter were closed when the pressure in the furnace increased to 105 Pa. The protection effects on molten magnesium alloy and the morphology and composition of the surface films were studied.
2.2 Experimental method in open furnace
Figure 1 shows a schematic diagram of the apparatus. A silicon temperature controller was used to adjust the temperature of the melt (error band ≤3 °C). Several mass flow meters were used to control the composition and flow rate of the protective gas. A stainless steel scraper was used to remove the old surface film.

Fig.1 Schematic diagram of apparatus used in this experiment
First, commercial AZ91D alloy was added to the stainless steel bowl in the crucible, producing mixed gas, which was controlled by mass flow meters. Twenty minutes later, heat was applied until a constant temperature was reached, and then the scraper was used to remove the old surface film so that a fresh melt surface could be obtained. The melt was left in the furnace for a certain period. The samples with the stainless steel bowl were then lifted up to the cooling area of the furnace and cooled rapidly (10 °C /min).
3 Results and discussion
3.1 Results in sealed furnace
3.1.1 Protection effect in SO2+N2
Figure 2 shows the macro morphology of the film formed in N2 atmosphere containing 4.0% SO2 (volume fraction) at 680 °C for 60 min. It indicates that the sample is not fully protected with some black cauliflower-like oxides formed on the surface. In this experiment, the ratio of the protected area to the whole area of the sample was used to estimate the protection effect (η) in different conditions. The larger the ratio becomes, the better the protection effect is. Table 1 lists the protection effect of gases with the change of the SO2 content and the melting temperature.
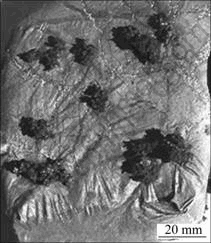
Fig.2 Macro morphology of sample held at 680 °C for 60 min in 4.0%SO2+96.0%N2
Table 1 Protection effect of gases to AZ91D with change of SO2 content and melting temperature

It can be seen from Table 1 that the protection effect decreases when the melting temperature is increased and the cauliflower-like oxides first appears at 680 °C. When the melting temperature reaches 700 °C, the sample is fully not protected and the surface of the sample is entirely covered by the black oxides. Meanwhile, there are abundant white volatiles on the inside surface of the sealed furnace. This could be due to the fact that the vapor pressure of magnesium increases largely and magnesium is more prone to vaporizing when the melting temperature increases, therefore, the protection of the mixed cover gas on the samples becomes more difficult. Table 1 also indicates that all the samples could not been fully protected at 680 °C and increasing the SO2 content could partly enhance the protection effect.
3.1.2 Surface appearance and composition of films in SO2+N2
Figure 3 shows the surface morphology, EDS spectrum and element concentration. EDS spectrum shows that the cauliflower-like oxide mainly contains magnesium and oxygen and their mole ratio is almost 1, which could be regarded as MgO. EDS spectrum also indicates that the protected surface film contains a small amount of sulfur except magnesium and oxygen. The detected aluminum might be the signal coming from the bulk magnesium alloy.
Figure 4 shows the morphologies of protected surface of the films formed at 680 °C for 60 min under atmospheres of N2 containing different SO2 contents. The average composition of 80 μm×80 μm area of the films is shown in Table 2. It can be seen that the film becomes smoother when the SO2 content is increased, and the film formed in 0.5% SO2 only contains magnesium, aluminum and oxygen, whereas the films formed in 2.0% and 4.0% SO2 also contain a small amount of sulfur element. Therefore, the increased protection effect by the increase of the SO2 content could attribute to the sulfur element in the film, which enhances the protective capability of the film.

Fig.3 SEM micrograph of surface film (a) (AZ91D, 680 °C, 4.0% SO2+96.0% N2, 60 min), EDS results of cauliflower-like oxides (b) and protected surface (c) in (a)

Fig.4 SEM images of surface film formed in N2 containing different SO2 contents (AZ91D, 680 °C, 1 h): (a) 0.5%; (b) 2.0%; (c) 4.0%
Table 2 Composition of surface film in Fig.4

3.1.3 Effect of adding air on protection behavior of AZ91D
The impact of adding air into the cover gas on the protection effect to the molten magnesium alloy was also investigated. Figure 5 shows the macro morphologies of the film formed at 680 °C for 10 min under atmospheres of 0.5% SO2 and N2 mixture with 5%, 20%, 40%, 60%, 80% and 90% air. It indicates that the samples are not protected when the air content are 5% and 20%, whereas the samples could be mostly protected only with a small amount of cauliflower-like oxides formed on the surface when 40%, 60% and 80% air are added into the cover gas. When the air content in the cover gas is increased to 90%, the sample could be fully protected without cauliflower-like oxides formed on the surface. Therefore, it could conclude that the protection of SO2-containing cover gases on the molten magnesium alloy needs a proper amount of air in the cover gases.
3.2 Protection mechanism of SO2-containing cover gas
3.2.1 Film appearance and composition
Figure 6 shows a view of the cracked surface film formed on the surface of sample held in 0.5% SO2 at 680 °C for 60 min in the open melting furnace. It indicates that there is a regular film on the surface of the alloy, and the thickness of the film is between 0.5 and 3.0 μm.

Fig.5 Macro morphologies of samples formed in N2+0.5%SO2 containing different air content: (a) 90%; (b) 80%; (c) 60%; (d) 40%; (e) 20%; (f) 5%

Fig.6 SEM images of surface morphology of oxide film/metal substrate of sample held at 680 °C for 60 min in 0.5% SO2+99.5% N2
Figure 7 shows the surface appearance of the sample held at 680 °C for 90 min in the atmosphere containing 0.5%SO2. Table 3 lists the EDS results of points 1-4 corresponding to the associated position in Figure 7. The results indicate that the surface film is a network structure and the boundary area and interior area have almost the same element content. XRD pattern not shown here indicates that the surface film is composed of MgO and MgS.
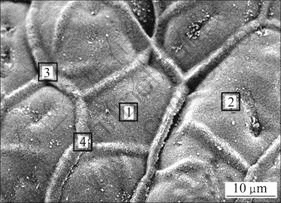
Fig.7 Microstructure of surface of sample held at 680 °C for 90 min in 0.5% SO2+99.5% N2
Table 3 EDS results of positions 1-4 in Fig.3(mole fraction, %)

3.2.2 Film appearance and composition with different holding time
Figure 8 shows the surface appearance of samples held at 680 °C for different time under 0.5% SO2. Figure 9 shows the element composition of the sample in Fig.8. It can be seen that, the protective film first forms for 10 min exposure and all the surface films are in a network structure, the boundary area becomes more extrusive than the interior area when holding time increases. It also can be seen that the film with 3 min exposure only contains magnesium and oxygen, and the oxygen and sulfur contents increase with the increase of the holding time, whereas the magnesium content decreases. Therefore, a thin layer of MgO first forms on the sample surface and then MgS forms as the holding time increases. When the holding time reaches 90 min, oxygen content exceeds magnesium content and magnesium content could not balance oxygen and sulfur content with MgO and MgS. It can be attributed that MgS is not stable in thermodynamics and will be partly oxidized to MgSO4.
The forming process of protective film can be concluded as follows: in the early stage, the film only composed of MgO forms on the surface of samples by the prior reaction between magnesium and oxygen in the atmosphere. As the MgO film is porous and not compact, SO2 and O2 molecules could pass through the macro and micro holes in the film and react with magnesium to form MgS. When the composite layers of MgO and MgS form, the outside diffusion of magnesium ion controlls the growth of the film. Because the diffusion rate of magnesium ion in MgS is lager than that in MgO, the growth rates of different areas with different MgS contents in the film are different, finally, the film with a network structure forms.
3.2.3 Protection mechanism
It is known that magnesium oxide film does not protect the molten magnesium alloy from further oxidation due to the mismatch of the molar volume of magnesium oxide and the molar volume of the equivalent amount of magnesium (the Pilling-Bedworth ratio)[14-15]. Since the Pilling-Bedworth ratio of the magnesium oxide film is 0.73[16], magnesium ion can still react with oxygen on the surface of samples by diffusing through the porous oxide film. When some MgS with a Pilling-Bedworth ratio of 1.4[16] forms in the film, the film becomes dense and can protect magnesium alloy effectively.

Fig.8 SEM micrographs of samples held at 680 °C for different time in 0.5% SO2+99.5% N2: (a) 3 min; (b) 10 min; (c) 30 min; (d) 60 min; (e) 90 min; (f) 150 min
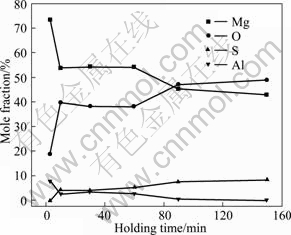
Fig.9 Composition of films produced with different holding time (0.5% SO2+99.5% N2, 680 °C)
4 Conclusions
1) Molten AZ91D magnesium alloy is not protected properly in a sealed furnace under atmosphere containing SO2 and N2, with cauliflower-like MgO formed on the sample surface. Protection effect on the melt obviously decreases with the increase of the melting temperature, whereas increasing the SO2 content can partly enhance the protection effect and it can be attributed to the increased sulfur content in the film. The melt is well protected when a proper amount of air is added into the atmosphere of SO2 and N2.
2) The cover gas containing SO2 and air protects molten magnesium alloy by reacting with the melt to form a coherent protective film with a network structure. The protective film is first composed of MgO, and with the increase of the holding time, MgS forms in the film by SO2 molecule diffusing through the porous magnesium oxide film and reacting with magnesium ion. MgS increases the Pilling and Bedworth ratio of the surface film and enhances the protective capability of the film.
References
[1] THARUMARAJAH A, KOLTUN P. Is there an environmental advantage of using magnesium components for light-weighting cars [J]. Journal of Cleaner Production, 2007, 15: 1007-1013.
[2] MEBARKI N, RAVI KUMAR N V, BLANDIN J J, SUERY M, PELLOUX F, KHELIFATI G. Correlation between ignition and oxidation behaviors of AZ91 magnesium alloy [J]. Materials Science and Technology, 2005, 21(10): 1145-1151.
[3] ZENG Y W, MAO M X, HUANG Z Q. Research on magnesium melting protected by cover gas mixtures containing HFC-32 [J]. Foundry, 2006, 55(8): 776-779. (in Chinese)
[4] HILLIS J E. The international program to identify alternatives to SF6 for magnesium melt protection [C]// International Conference on SF6 and the Environment, San Diego, America, 2002.
[5] BARTOS S, CURTIS L, SCHARFENBERG J, KANTAMANENI R. Is there an environmental advantage of using magnesium components for light-weighting cars [J]. Journal of Cleaner Production, 2007, 15: 979-987.
[6] FRUEHLING J W, HANAWALT J D. Protective atmospheres for melting magnesium alloys [J]. American Foundryman’s Society Transactions, 1969, 77: 159-164.
[7] CASHION S P. The use of sulphur hexafluoride(SF6) for protecting molten magnesium [D]. Australia: Department of Mining, Minerals and Materials Engineering, University of Queensland, 1998.
[8] PETTERSEN G, ?VRELID E, RNELL G. Characterization of the surface films formed on molten magnesium in different protective atmospheres [J]. Materials Science and Engineering A, 2002, 332: 285-294.
[9] AARSTAD K. Various techniques to study the surface of magnesium protected by SF6 [C]// Magnesium Technology 2003, San Diego, America: TMS (The Minerals, Metals & Materials Society), 2003: 35-38.
[10] XIONG S M, LIU X L. Microstructure, composition, and depth analysis of surface films formed on molten AZ91D alloy under protection of SF6 mixtures [J]. Metallurgical and Materials Transactions A, 2007, 38: 428-434.
[11] JONSSON T, J?RDN?S A, SVENSSON J E, JOHANSSON L G, HALVARSSON M. The effect of traces of SO2 on iron oxidation: A microstructural study [J]. Oxidation of Metals, 2007, 67: 193-213.
[12] J?RDN?S A, SVENSSON J E, JOHANSSON L G. Influence of SO2 on the oxidation of 304L steel in O2+40%H2O at 600 °C [J]. Oxidation of Metals, 2008, 69: 249-263.
[13] FRUEHLING J W. Protective atmospheres for molten magnesium [D]. Michigan, USA: The University of Michigan, 1970.
[14] PILLING N B, BEDWORTH R E. The oxidation of metals at high temperatures [J]. Journal of the Institute of Metals, 1923(28): 534-591.
[15] ZHOU H, WANG M X, LI W, WANG L, ZHAO Y. Effect of Ce addition on ignition point of AM50 alloy powders [J]. Materials Letters, 2006, 60: 3238-3240.
[16] AARSTAD K. Protective films on molten magnesium [D]. Norway: Norwegian University of Science and Technology, 2004.
含SO2气氛对熔态镁合金的保护行为
王先飞, 熊守美
清华大学 机械工程系,汽车安全与节能国家重点实验室,北京 100084
摘 要:在密封和开放条件下研究含SO2的不同气氛对AZ91D合金的保护行为,分析表面膜的形貌与成分。结果表明:在密封条件下,含SO2气体对熔体的保护需要一定量的O2;含SO2和O2的气氛通过与熔体反应,在熔体表面形成一层致密的网状结构表面膜,表面膜主要含有MgO和MgS;MgS提高了表面膜的致密度,从而提高了表面膜的保护作用。
关键词:镁合金;SO2;熔体保护;表面膜
(Edited by LI Xiang-qun)
Foundation item: Project (2009AA03Z114) supported by the National High-tech Research and Development Program of China; Project (20090002110029) supported by the Specialized Research Fund for the Doctoral Program of Higher Education, China
Corresponding author: XIONG Shou-mei; Tel/Fax: +86-10-62773793; E-mail:smxiong@tsinghua.edu.cn
DOI: 10.1016/S1003-6326(11)60785-1