
Dissolution kinetics of low grade complex copper ore in ammonia-ammonium chloride solution
LIU Wei(�� ά), TANG Mo-tang(������), TANG Chao-bo(�Ƴ���),
HE Jing(�� ��), YANG Sheng-hai(������),YANG Jian-guang(���)
School of Metallurgical Science and Engineering, Central South University, Changsha 410083, China
Received 5 June 2009; accepted 13 October 2009
Abstract: The leaching kinetics of Tang-dan refractory low grade complex copper ore was investigated in ammonia-ammonium chloride solution. The concentration of ammonia and ammonium chloride, the ore particle size, the solid-to-liquid ratio and the temperature were chosen as parameters in the experiments. The results show that temperature, concentration of ammonia and ammonium chloride have favorable influence on the leaching rate of copper oxide ores. But, leaching rate decreases with increasing particle size and solid-to-liquid ratio. The leaching process is controlled by the diffusion of the lixiviant and the activation energy is determined to be 23.279 kJ/mol. An equation was also proposed to describe the leaching kinetics.
Key words: leaching kinetics; ammonia; ammonium chloride; low grade copper ore
1 Introduction
In recent years, more and more attentions have been focused on the extraction of oxidized copper ores due to the depletion of higher grade copper sulfides[1-2]. Sulfuric acid is the most common lixiviant used. The preponderance of carbonates in some ore deposits often induces tremendous increase in acid consumption, thus a more selective reagent is needed for such deposits. Ammonia has attracted a lot of researchers�� interests because it does not react with carbonates, which greatly reduces the cost eventually[3-4].
BRYDEN[5] studied the leaching kinetics of a copper ore containing malachite and chrysocolla in ammonium sulfate solution. It was found that the leaching rate was controlled by the surface reaction; and the activation energy was calculated to be 48.5 kJ/mol. A Langmuir adsorption type model was also applied to describe the kinetics. MENA and OLSON[6] investigated the leaching kinetics of chrysocolla in ammonia-ammonium carbonate solutions in view of following possible kinetic controlling stages: 1) diffusion through microcracks and some pores, 2) adsorption of ion pairs at surface-active sites, and 3) chemical reaction at the active surface sites. The activation energy obtained was variable with both total ammonia concentration and temperature. The leaching behavior had first-order dependence on the concentration of ammonium carbonate. PAUL and FERRON[7] studied the leaching kinetics of malachite in ammonium carbonate solution. The whole dissolution procession was divided into two stages. At the first stage, the initial dissolution of malachite proceeded rapidly. Then, the rate was reduced due to the surface blockage of a needle-structured immediate product, presumably Cu(OH)2. The activation energies of the two stages were 64 kJ/mol and 75 kJ/mol, respectively. And these two stages were both controlled by heterogeneous reaction. The dissolution kinetics of malachite in ammonium chloride solutions was studied by YARTASI and COPUR[8]. The results demonstrated that the dissolution rate was controlled by lixiviant diffusion through the product film, and the activation energy was determined to be 81.3 kJ/mol. EKMEKYAPAR and OYA[9] also chose ammonium chloride solution as the lixiviant and the activation energy for the dissolution reaction was calculated to be 71 kJ/mol. The dissolution rate is controlled by mixture kinetics and a mathematical model of 1-2(1-x)1/3+ (1-x)2/3=10-5c2.10��dp-1.96����S/L-0.64��n1.78��exp(-8 500/T)��t was proposed to represent the reaction kinetics, where x, c, dp, ��S/L, n, T and t were the fractional conversion of malachite, ammonium chloride concentration, particle diameter, solid to liquid ratio, stirring speed, temperature, and reaction time, respectively. K?NK?L et al[10] examined the dissolution of malachite in ammonia solutions. They declared that the activation energy was 22.338 kJ/mol and the whole process was controlled by diffusion through the ash film. ARZUTUG and KOCAKERIM[11] investigated the leaching kinetics of malachite in ammonia gas saturated water. It was found that the leaching rate followed a pseudo-second-order kinetic model with an activation energy of 85.16 kJ/mol. BING?L et al[12] found that interface transfer and diffusion across the product layer was the controlling step during leaching of the oxide copper ore containing malachite in NH3-(NH4)2CO3 solution and the activation energy of dissolution was 15 kJ/mol.
Tang Dan refractory copper oxide ore is by now the largest copper mine in China for its about 115��104 t reserves. It is famous for its high grade calcium magnesium carbonate gangues and poor floatable characteristics. Its average content of copper is 0.75% (mass fraction) and the phase composition of copper is very complex[13]. Leaching the ores in the ammoniacal systems is always performed as the only feasible and economical choice. Ammonia-ammonium chloride solution was chosen as the lixiviant in the present work and the leaching kinetics was studied in detail. Though many researchers have done a lot of studies on the leaching kinetics of copper oxide ores, the materials used in those researches always contained a high content of copper (��10%) and the copper usually presented in one main single phase such as malachite and chrysocolla. The characteristics of those materials are quite different from Tang Dan refractory copper oxide ore and it is very necessary to investigate the kinetics of leaching Tang-dan refractory copper oxide ore with ammoniacal solution.
2 Experimental
2.1 Materials
The copper ore used in this study was obtained from Tang Dan, Yunnan Province, China. The chemical composition and X-ray diffraction pattern are given in Table 1 and Fig.1, respectively. The material is a typical lean copper ore, which is composed by a majority of oxide minerals and a small quantity of sulfides, just as shown in Table 2. The content of copper in the ore is too low to be detected by the X-ray diffraction method. And the phases of copper were identified by the polarizing microscope method to be mainly malachite, chrysocolla and bornite, corresponding to free copper oxide, copper silicates and secondary copper sulfide, respectively. Tenorite, chalcocite, chalcopyrite, etc, also exit in a very low content. Gangue usually contains high grade calcium magnesium carbonate and silica. These minerals always consume a lot of acidic lixiviant and the basic ammoniacal solution was chosen to leach the copper.
Table 1 Chemical composition of copper ore used in this study (mass fraction, %)

Table 2 Phase composition of the copper ore

Free copper oxide corresponds to copper compound which can be leached out in 2 h in the solution containing 30 mL/L ethylenediamine + 50 g/L NH4Cl + 50 g/L Na2SO3 solution, including sulphates, chlorides, carbonates and oxides of copper.
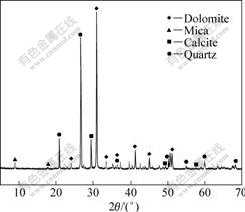
Fig.1 X-ray diffraction pattern of copper ore
2.2 Methods
The leaching experiments were carried out in a 500 mL spherical glass reactor equipped with a mechanical stirrer, a temperature control unit and a cooler to avoid evaporation loss of solution and ammonia. 250 mL solution containing specific concentration of ammonia and ammonium chloride was placed into glass reactor. After the desired stirring speed and reaction temperature were attained, the solid sample was added to the solution in reactor. 5 mL solution was withdrawn and filtered after specific time for analyzing the concentration of copper in the solution by atomic absorption spectrophotometer (AAS) and 5 mL fresh lixiviant was added into the reactor immediately to maintain the volume of the solution constant.
According to the former experimental results, it was found that the effect of stirring speed could be neglected when it was larger than 450 r/min. All leaching experiments were carried out at the stirring speed of 500 r/min. Ammonia and ammonium chloride concentration, particle diameter, solid-to-liquid ratio, reaction temperature and stirring speed, which may affect the dissolution rate, were employed as experimental variables.
3 Results and discussion
3.1 Dissolution reaction
The dissolutions of malachite and chrysocolla, which are the main phases of copper in the ore, can be described as Eq.(1) and Eq.(2), respectively:

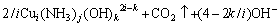 ,
,
i=1, 2; j=0, 1, 2, 3, 4, 5; k=0, 1, 2, 3, 4 (1)

 ,
,
i=1, 2; j=0, 1, 2, 3, 4, 5; k=0, 1, 2, 3, 4 (2)
The solubility of malachite and chrysocolla in ammonia-ammonium chloride solution was investigated in another work and the results are shown in Fig.2. The solubility of bornite in ammonia-ammonium chloride solution is neglectable when no extra oxidant is added. And it is considered difficult to be leached out.
3.2 Effect of operation parameters on copper leaching rate
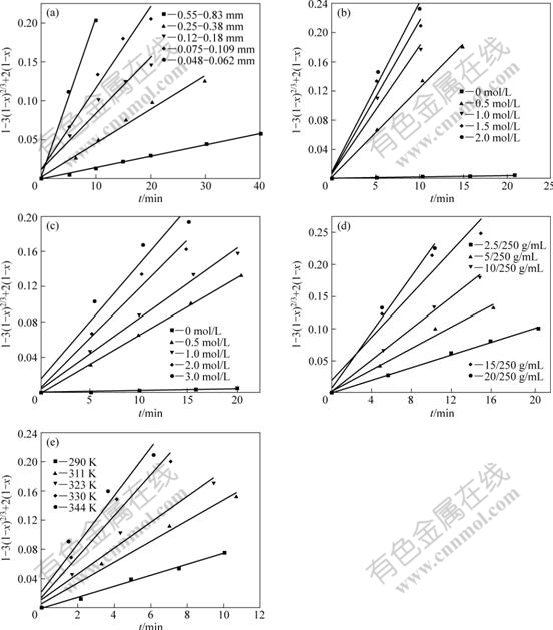
The effect of particle size, concentration of lixiviant, solid-to-liquid ratio and temperature on the leaching rate of copper is shown in Fig.3. The fixed conditions were chosen as the particle size of 0.074-0.104 mm, ammonia concentration of 0.5 mol/L, ammonium chloride concentration of 2 mol/L, solid-to-liquid ratio of 10/250 g/mL and operation temperature of 293 K. As shown in Fig.3, the leaching rate of copper increases with the particle diameter decreasing. This could be attributed to the increase in the particle number per amount of solid when the particle size decreases, and an corresponding increase in the surface area, which causes better exposure of copper ores to the solution. Larger ammonia concentration leads to the increase of the leaching rate of copper. The increase in the ammonia concentration means more reactant to attack the solid, thus increasing the leaching rate. Without ammonia, the extremely low leaching rate could be attributed to the extremely low concentration of free ammonia in the solution. The leaching rate increases as the concentration of ammonium chloride increases. This is attributed to two reasons, one of which is that ammonium chloride can form a buffer solution with ammonia and pH value of the solution is kept constant at a certain value; and the other is that NH4+ could react with OH- to form NH3, which subsequently increases the concentration of reactant and increases the leaching rate:
NH4++OH-=NH3+H2O (3)
The leaching rate increases as the solid-to-liquid ratio decreases. This can be explained by the decrease in the amount of lixiviant per unit amount of the solid with the increase of solid-to-liquid ratio. The temperature elevation also has favorable influence on the leaching rate of copper oxide ore.
3.3. Phase and morphology transformation
The SEM images of the copper oxide ore particles and the leached residue with the diameter of 0.55-0.83 mm are presented in Fig.4 and Fig.5, respectively. The copper containing minerals existing as single particles on the surface and edge of the particles could easily be found in the image of the particles before leaching, while scarcely found in the images of the leached residue. Copper minerals which still have not been leached out mostly exist in the inner of the ore particles and these minerals as well as the gangue are found to contain high

Fig.2 Solubility of malachite (a) and chrysocolla (b) in ammonia-ammonium chloride solution at 298 K
Fig.3 Effects of operation parameters on copper leaching rate: (a) Particle size; (b) Ammonia concentration; (c) Ammonium chloride concentration; (d) Solid-to-liquid ratio; (e) Temperature

content of silicon, which means copper containing minerals which have not been leached out are likely to be silicates. The phase compositions of the particles with different diameters before and after leaching are shown in Table 3 and Table 4, respectively. As shown in Table 3 and Table 4, the free copper oxide is easy to leach out; and almost all of the free copper oxides are leached out when the particle size is as small as 0.048-0.0623 mm. Copper silicates are very difficult to leach out though the solubility in the solution is high enough according to the thermodynamic studies. Copper atoms in this kind of phase always disperse in the crystal lattice of the gangue
Table 3 Phase composition of particles with different diameters before leaching

Table 4 Phase composition of particles with different diameters after leaching


Fig.4 SEM images of copper ore particles with diameter of 0.55-0.83 mm (White parts: copper containing minerals): (a) Copper minerals emerging on surface of ore particle; (b) Copper minerals emerging on edge of ore particle; (c) Copper minerals emerging on inside of ore particle

Fig.5 SEM image of leached residue with diameter of 0.55-0.83 mm
ores. And the diffusion of lixiviant through the lattice is very difficult and consequently affects the leaching rate. The phase of copper sulfides is also leached out partly, especially when the particle size is small. The possible reason for this phenomenon may be as follows. The copper sulfides are oxidized and eventually leached out to solution with the oxygen dissolved in the solution as an oxidant and Cu2+ in the solution as a catalyzer[14].
4 Kinetic analysis
Fluid-solid reactions are numerous and of great industrial importance. In the leaching processes of ores with lixiviant solutions, the change in solid particle size can be neglected during the heterogeneous reaction if the ore contains large amounts of unreactive impurities in presence of a nonflaking ash. In this kind of reaction, there are some physical steps that can affect the reaction rate, in addition to the chemical reaction, such as diffusion through the fluid film and/or inert solid layer of the reactant or product.
The most important models suggested for the derivation of the rate expression of a noncatalytic fluid-solid reaction are the shrinking core model and the progressive conversion model. According to the shrinking core model, it is thought that the reaction takes place on the outer surface of the solid and this surface shrinks toward the center of the solid as the reaction proceeds, leaving behind an inert solid layer, called ��ash layer��, around the unreacted shrinking core[15].
Considering a solid particle M immersed in a fluid N and reacts with it as the following equation:
N(fluid)+bM(solid)��Products (4)
If the reaction rate of the particle is controlled by diffusion of the fluid N through ash layer, the time t required for a spherical solid to react can be calculated by the following equation:
 (5)
(5)
where x is the fractional conversion of malachite; t is the leaching time, min; D is the diffusivity of ions through the ash layer, m2/s; c0 is the concentration of fluid outside the particle, mol/L3; cB is the apparent concentration of the solid reactant, mol/L3; r0 is the initial outside radius of the particle, m.
If the reaction rate is controlled by chemical reaction, the integrated rate equation is expressed by the following equation:
 (6)
(6)
where kd is the chemical reaction rate constant.
Dissolution kinetics of Tang Dan copper oxide ores in NH3?H2O-NH4Cl solution can be described by shrinking core model. It is determined that the leaching process is controlled by the diffusion through the ash layer around the shrinking unreacted core, as shown in Fig.6.
In order to determine the effect of temperature, particle size, solid-to-liquid ratio (S/L), ammonia concentration and ammonium chloride concentration on the reaction kinetics, the following semi-empirical model is established:

 (7)
(7)
Fig.6 Plots of t versus 1-3(1-x)2/3+2(1-x) at different operation parameters: (a) Particle size; (b) Ammonia concentration; (c) Ammonium chloride concentration; (d) Solid-to-liquid ratio; (e) Temperature
where T is the temperature, K; C(NH3?H2O) is the ammonium chloride concentration, mol/L; C(NH4Cl) is the ammonium chloride concentration, mol/L; k0 is the apparent reaction rate coefficient, s�C1; E is the activation energy, kJ/mol; S/L is the solid-to-liquid ratio, g/mL; and dp is the particle diameter, mm.
To different ammonia concentrations, when other parameters maintain constant, Eq.(7) could be written as
 (8)
(8)
 (9)
(9)
d(1-3(1-x)2/3+2(1-x))/dt is the slope of the straight lines corresponding to different ammonia concentrations in Fig.6(b). The values of ln[d(1-3(1-x)2/3+2(1-x))/dt] versus lnC(NH3?H2O) are plotted to form a straight line, and from the slope, it is calculated that a=0.471 05. Plotting the value of ln[d(1-3(1-x)2/3+2(1-x))/dt] versus lnC(NH3?Cl), ln(S/L) and lndp, b, c and d could be calculated, respectively, and the values of them are 0.392 74, 0.692 58 and -0.976 6, respectively, as shown in Fig.7.
When other parameters are fixed while temperature is chosen as variable factor, the semi-empirical equation could be written as
 (10)
(10)
 (11)
(11)
Plotting the value of ln[d(1-3(1-x)2/3+2(1-x))/dt] versus 1/T, the value of activation energy is calculated to be 23.279 kJ/mol from the slope of the straight line, as shown in Fig.8. The increase in leaching rate with temperature for the systems controlled by diffusion is relatively small and results in a low activation energy value of about 20 kJ/mol[16]. The low activation energy value found in this study confirms that the process is controlled by the diffusion through the ash layer around the unreacted shrinking core.
Substituting the value of a, b, c, d and E into Eq.(7) and the value of k0 is calculated to be about 181.38 when using the equation to fit different straight lines in Fig.6.

Fig.7 Plots of ln[d(1-3(1-x)2/3+2(1-x))/dt] versus lndp (a), lnC(NH3?H2O) (b), lnC(NH4Cl) and ln(S/L) (d)

Fig.8 Plot of ln[d(1-3(1-x)2/3+2(1-x))/dt] versus T-1
5 Conclusions
1) The leaching rate is controlled by the diffusion through the ash layer around the shrinking unreacted core and the activation energy is calculated to be 23.279 kJ/mol.
2) The leaching kinetics could be described by the following equation:



3) Free copper oxide could be leached out easily and copper silicates are difficult to leach out. Part of the phase of copper containing sulfide can also be leached out.
References
[1] PEACEY J, GUO Xian-jian, ROBLES E. Copper hydrometallurgy��current status, preliminary economics, future direction and positioning versus smelting [J]. Transactions of Nonferrous Metals Society of China, 2004, 14(3): 560-568.
[2] JU Shao-hua, TANG Mo-tang, YANG Sheng-hai, TANG Chao-bo. Thermodynamics of Cu(II)-NH3-NH4Cl-H2O system [J]. Transactions of Nonferrous Metals Society of China, 2005, 15(6): 1414-1419.
[3] FANG Jian-jun, LI Yi-fen, LU Xiang-lin, ZHANG Wen-bin. Effects and industry application of ammonia leaching of low copper oxide ore under normal temperature and pressure[J]. Mining and Metallurgical Engineering, 2008, 28(3): 27-38. (in Chinese)
[4] WANG Cheng-yan. Exploitation of rebellious low-grade copper oxide ore with high alkality gangues [J]. Mining and Metallurgy, 2001, 10(4): 49-52. (in Chinese)
[5] BRYDEN K O. Ammonium sulphate leaching of malachite and chrysocolla [D]. Salt Lake City: Department of Metallurgical Engineering, University of Utah, 1975: 25-48.
[6] MENA M, OLSON F A. Leaching of chrysocolla with ammonia- ammonium carbonates solutions [J]. Metallurgical Transaction B, 1985, 16(3): 441-448.
[7] PAUL D O, FERRON A O. Leaching kinetics of malachite in ammonium carbonate solutions [J]. Metallurgical Transaction B, 1983, 14(1): 33-40.
[8] YARTASI A, COPUR M. Dissolution kinetics of copper (��) oxide in ammonium chloride solutions [J]. Minerals Engineering, 1996, 9(6): 639-698.
[9] EKMEKYAPAR A, OYA R. Dissolution kinetics of an oxidized copper ore in ammonium chloride solution [J]. Chemical and Biochemical Engineering Quarterly, 2003, 17(4): 261-266.
[10] K?NK?L M M, KOCAKERIM S, DEMIRBA? Y A. Leaching kinetics of malachite in ammonia solutions [J]. International Journal of Mineral Processing, 1994, 41(3): 167-182.
[11] ARZUTUG M E, KOCAKERIM M M. Leaching of malachite ore in NH3-saturated water [J]. Industrial & Engineering Chemistry Research, 2004, 43(15): 4118-4123.
[12] BING?LA D, CANBAZO?LUB M, AYDO?AN S. Dissolution kinetics of malachite in ammonia/ammonium carbonate leaching [J]. Hydrometallurgy, 2005, 76(1/2): 55-62.
[13] CHENG Qiong, ZHANG Wen-bin. Technical process in ammonia leaching of Tang-dan oxidized copper ore containing alkaline gangues [J]. Yunnan Metallurgy, 2005, 34(6): 80-92. (in Chinese)
[14] JU Shao-hua. Study in hydrometallurgy thermodynamics of metal (Cu, Ni, Au) in the system of Me-NH4Cl-NH3-H2O and heap leaching process of their low-grade ores [D]. Changsha: School of Metallurgical Science and Engineering, Central South University, 2006: 50-51. (in Chinese)
[15] LEVENSPIEL O. Chemical reaction engineering [M]. New York: Wiley, 1972: 361-371.
[16] JACKSON E. Hydrometallurgical extraction and reclamation [M]. Chichester: Ellis Horwood Ltd., 1982: 46-47.
(Edited by YANG Bing)
Foundation item: Projects(2007CB613604) supported by the National Basic Research Program of China; Project(50674104) supported by the National Natural Science Foundation of China
Corresponding author: LIU Wei; Tel/Fax: +86-731-88830470; E-mail: ase.6520@163.com
DOI: 10.1016/S1003-6326(09)60235-1