Article ID: 1003-6326(2005)03-0648-05
Chemical behaviors of different arsenic-bearing sulphides bio-oxidated by thermophilic bacteria
YANG Hong-ying(���Ӣ)1, GONG En-pu(������)2,
YANG Li-li(������)2, WANG Da-wen(������)1
(1. School of Materials and Metallurgy, Northeastern University, Shenyang 110004, China;
2. School of Resources and Civil Engineering, Northeastern University, Shenyang 110004, China)
Abstract: The study on arsenopyrite and realgar of bacterial oxidation shows that the chemical behaviors of different arsenic-bearing sulphides oxidated by thermophilic bacteria are quite distinct. Arsenopyrite is active and quickly eroded in bacteria-bearing solution. With a high leaching rate over 95%, the arsenopyrite phase cannot be detected by X-ray diffraction(XRD). Arsenopyrite is highly toxic to bacteria that at the initial stage of bio-oxidation, bacterial growth is inhibited and the number of bacterium cell drops from 2.26��108/mL to the lowest 2.01��105/mL. At the later stages of bio-oxidation, bacteria grow fast and reach 2.23��108/mL. Comparably, realgar is inertial and resistive to bacterial corrosion and oxidation. Arsenic in realgar crystal is hard to be leached and the residue is still realgar phase, as indicated by XRD. The cell number of bacteria varies a little, decreasing from 2.26��108/mL to 2.01��107/mL, during the bacterial oxidation. The results show that the crystal structure and arsenic valency of arsenic-bearing sulphides play a vital role during the leaching process of bacterial oxidation.
Key words: thermophilic bacteria; bacterial oxidation; chemical behavior; arsenopyrite; realgar; toxicity CLC
number: TF111.31 Document code: A
1 INTRODUCTION
Arsenic-bearing gold ore is widely distributed in provinces of Yunnan, Guizhou, Sichuan, Guangxi, Guangdong, Hunan, Jiangxi, Liaoning, Jilin, Hebei, Shanxi, Gansu, Qinghai, Xinjiang, etc. With the process development of bacterium metallurgy[1, 2], such gold ore resource may be reasonably utilized, however, arsenic in these ores is highly toxic to the leaching bacteria and becomes the research focus. Arsenic is a complex and vital element of arsenic-bearing minerals with the electron configuration of 4s34p3[3]. When As loses three electrons of the upper lever, 4p3 orbit, As(��) forms, then it can be further oxidated to lose two electrons of the lower lever 4s3 orbit and As(��) is formed. According to chemistry theory, As may have four valencies of 3-, 0, 3+, 5+, while mineralogy investigation shows that As in minerals may have 10 states[4, 5], which is much more complex than the theoretical prediction of its valency and phase formats. There are kinds of arsenic-bearing minerals of gold ores, among which arsenopyrite(FeAsS) and realgar(As4S4 ) are common types. The former consists of the mineral anion radical with [AsS]2-, and the latter constitutes the sulphides of ring molecule with As4S4. Considering that the mineral crystal structure, valency, phase will lead to the complexity of bacterial oxidation, and directly influence the process stability and econotechnical norms of arsenic-bearing gold ores[6, 7], studies on arsenic-bearing sulphides of arsenopyrite and realgar are theoretically meaningful and practical to the industrialization of bio-oxidation process.
2 EXPERIMENTAL SAMPLE
2.1 Crystal structure, morphology and composition of arsenopyrite
Arsenopyrite(FeAsS) with the nominal composition of Fe 34.30%, As 46.01%, S 19.69% is assigned to the subtype of arsenopyrite/island-base sulphides, cobaltine-arsenopyrite group and arsenopyrite subgroup. Arsenopyrite belongs to the monoclinic system, multi-bond type, where arsenic-sulphur pair [AsS] is composed of S��As covalent bond and linearly paralleled. Its morphology is a rhombic prism type with columnar crystals stretching alone c-axis, and its aggregate is granular and compacted massive. Component of As and S in arsenopyrite anion radical varies from FeAs0.9-S1.1 to FeAs1.1S0.9. Considering that Fe and Co can partly form isomorphous replacement, Ni can replace Fe, and Au and Ag often exist as mechanical mixture, arsenopyrite is one of the most important Au-bearing minerals. In our experiment, 95% arsenopyrite sample is ��74.5��m, and its analysis composition is Fe 33.65%, As 44.60%, S 19.63%.
2.2 Crystal structure, morphology and composition of realgar
Realgar(As4S4 or AsS) has a nominal composition of As 70.10%, S 29.90% and a little impurity. It��s monoclinic with adamantine luster crystal plane, and molecular structure. As and S are linked by covalent bonds, ring molecule is composed of As4S4 and different rings are connected by molecular bonds. Realgar morphology can be assigned to rhombic prism crystal class, or sometimes columnar, short-columnar, needle pattern can be observed along c-axis, and there are small longitudinal striations on the cylinder. It��s granular, compacted massive or occasionally as argillaceous bulk, powder and shell aggregate. In our experiment, 95% realgar sample is ��74.5��m, and its analysis composition is As 63.00%, S 27.94%, others are impurity.
3 BIO-OXIDATION EXPERIMENTAL
3.1 Method
The bacteria in the leaching solution are derived from sulphide mines, which are excellent engineering bacteria mixed mainly by Thiobacillus ferrooxidans and Leptospirillum ferrooxidans, and characterized by its thermotolerance and arsenic resistance. In our experiment, 200mL bacteria solution were taken, then N, P, K-bearing inorganic salts and other nutrition were added, and pH value was adjusted. After that, homogeneous intermixture was subpackaged into two 500mL cone flasks, and the initial pH=1.4, ��h=632mV. The solution contains no As element. Then 2.54g arsenopyrite powders and 1.80g realgar powders were added into the two flasks respectively. The following tests were carried out at 45�� in a shaker-HZQ-QX at 190r/min. The oxidation tests of thermophilic bacteria lasted 14d, and parameters were measured every 24h. Potential of solution was measured on Hana-PH301 laboratory-size acidimeter. As content was tested by hypophosphite titration and the principle was as follows: Taking CuSO4 as catalyst in 6mol/L HCl solution, deoxidizing As to monomer with hypophosphite then lixiviating. After As was solved by iodine standard solution, the excessive iodine was titrated by sodium arsenite standard solution in alkality environment, then As amount can be calculated. Solid phase was analyzed by XRD on D/max-RB XRD diffractometer. Bacterium cell number was measured by micro-observation count with hemocyte tally, and the equation was: Bacterium cell number per bacteria-bearing solution=average cell number per lattice��4��106��dilute multiple of bacteria-bearing solution.
3.2 Results
3.2.1 Potential of bacteria-bearing solution
The solution potential changes much with the addition of arsenic-bearing arsenopyrite and realgar. After arsenopyrite is added, the solution potential decreases rapidly and reaches the lowest 300mV in the next day, then gradually increases. In the seventh day, such potential increment is accelerated to 485mV; in the eighth day, the potential increases in a quick and stable way and reaches the maximum 603mV in the fourteenth day. Comparably, after realgar is added, the solution potential decreases slowly and falls to 530mV in the next day. During the experiment, the solution potential varies rather gently within 500-532mV. It can be seen that there is complete difference between the potential changes of two bio-oxidated arsenic-bearing sulphide solutions. The potential of arsenopyrite-bacterium solution decreases in a wide range then increases violently, while for the realgar-bacterium solution, the potential previously descends in a much smaller range than that of arsenopyrite, then decreases a little, and during the whole process, the potential curve is rather flat(see Fig.1).

Fig.1 Potential during bio-oxidation of arsenopyrite and realgar
3.2.2 Arsenic content and oxidation rate in solution
For the flask added with arsenopyrite, arsenic in the mineral solves and in the first day, its content in the solution is 1.40g/L with its oxidation rate of 2.4%. Then, arsenic in arsenopyrite is oxidated gradually into the solution and in the eighth day, arsenic content and oxidation rate reach 4.89g/L and 43.27% respectively; in the ninth day, the above two values arrive at 10.6g/L and 95.22%, respectively. For the flask added with realgar, the arsenic content almost keeps constant. In the first day, arsenic content is 0.42g/L and then changes little; in the twelfth day, arsenic content is 0.71g/L, which is the maximum due to the difficulty of arsenic to be leached; its oxidation rate is 6.28%, which keeps a small and almost constant value(see Figs.2 and 3).
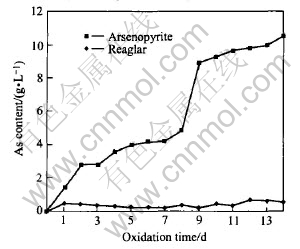
Fig.2 As content during bio-oxidation of arsenopyrite and realgar
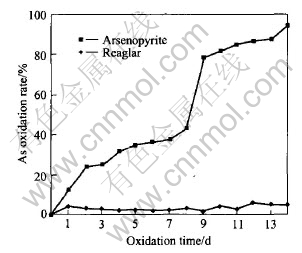
Fig.3 As oxidation rate of arsenopyrite and realgar during bio-oxidation
3.2.3 Bacterium growth
Bacterium growth during oxidation is investigated, and the variation of bacterium cell number is listed in Table 1. The initial bacterium amount is 2.26��108/mL. For the flask added with arsenopyrite, the amount drops to the minimum 2.01��105/mL in the first day, then the value increases and reaches 1.14��108/mL in the eighth day, and in the end of experiment, the bacterium amount arrives at the order of initial value 2.23��108/mL. For the flask added with realgar, the bacterium amount changes slowly to 2.13��108/mL in the first day, then decrease tardily and in the end of experiment, the value is 2.01��107/mL.
3.2.4 XRD analysis of bio-oxidation residue
The bio-oxidation residue of arsenopyrite and
Table 1 Bacterium growth during bio-oxidation of arsenic-bearing sulphides(/mL)
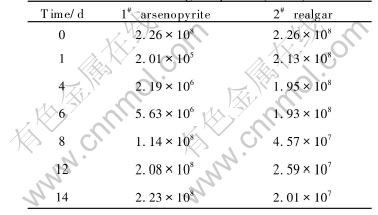
realgar is analyzed by XRD after the oxidation experiment. Fig.4 shows the XRD pattern of arsenopyrite bio-oxidation residue, which indicates that arsenopyrite crystal has been decomposed and new phases of arsenic oxide(Al2O3) and complex arsenate Pb(Fe2.54Al0.46)(As1.07O4)(S0.93O4)(OH)6 are crystallized. Fig.5 shows the XRD pattern of realgar bio-oxidation residue. The upper and lower ones describe the XRD results of oxidation residue and realgar respectively, and it can be seen that the two patterns consist with each other. Its chemical composition is As4S4 and no new phase is formed.

Fig.4 XRD pattern of arsenopyrite oxidation residue
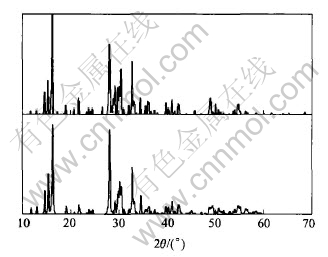
Fig.5 XRD pattern of realgar oxidation residue
4 DISCUSSION
Arsenic is a most harmful element to the growth and activity of bacteria(Thiobacillus ferrooxidans, etc) in leaching solution[8-10], therefore As content is a big problem and an important factor in bio-metallurgy process[11, 12]. The toxicity of As to leaching bacteria lies in the influence of arsenate on phosphate transformation in vivo bacteria to disturb the formation of phosphorylating intermediates. Then the bacteria will behave as "phosphate starvation" and result in enzymatic restrain and devitalization. Accordingly, DNA in-vitro recombination can be utilized to excise arsenoresistant enzyme, clone and construct Thiobacillus ferrooxidans, then arsenide cumulated in bacteria can be reduced and discharged particularly, and phosphate system of bacteria will work normally without As toxicity[13, 14]. The bio-oxidated experiments of arsenopyrite and realgar show that the chemical behaviors of disintegration and solution of different As-bearing minerals differ during bio-oxidation. Arsenopyrite will be solved rapidly in bacterium solution, while for realgar, the bio-oxidation is a second-order reaction. Here the first-order reaction is a catalytic oxidation reaction in the realgar crystal interface(see Eqn.(1)), where the products Fe2+ and As(��) largely enter the solution, then Fe2+ results in the drastical decrease of solution potential of 300mV, the toxicity of As(��) leads to the death of some bacteria and the bacterium amount drops greatly from 2.26��108/mL to the lowest value 2.01��108/mL. In the second-order reaction of realgar, the main reaction is between Fe3+ and As(��), and As(��) is converted to As(��) as described in Eqn.(2).
4FeAsS+11O2+6H2O��4FeSO4+4H3AsO3(1)
H3AsO3+2Fe3++H2O��
H3AsO4+2Fe2++2H+(2)
The thermophilic bacteria used in our experiments are well arsenoresistant and quickly oxidative. In the high arsenic solution, they will be rapidly oxidated; after the middle stage, the leaching bacteria will adapt to the high arsenic environment, the As(��) in the solution is little toxic to bacteria and results in the gradual increase of bacterium amount to 1.14��108/mL. The amount of Fe2+ solved from the realgar crystals to the solution exhibits a stable tendency, then Fe2+ is bio-oxidated to Fe3+. With the ratio increment of Fe3+/Fe2+ , the electric potential increases. At the later stage, the realgar crystals are completely oxidated, Fe3+/Fe2+ is largely enhanced and the potential greatly increases. The As(��) is oxidated to As(��) in the solution with lower toxicity, and bacteria begin their most bloom stage of growth, namely logarithm stage. In the twelfth day, the bacteria reaches 2.08��108/mL. At the final stage, the bacterium cell number reaches 2.23��108/mL and As oxidation rate is 95.22% calculated from liquid arsenic. Comparably, in the bio-oxidated experiment of realgar, the potential hardly changes, and the investigation on bacterium growth shows that the cell amount keeps almost constant. At the initial stage, the cell amount drops a little from 2.26��108/mL to 2.13��108/mL and such small decrease is caused by the adsorption of some bacteria on realgar crystals. During the whole process, bacterium cell amount in the flask added with realgar exhibits a stable tendency, which results from the insolubility of As of realgar. Then As cannot enter the solution and remains as solid phase, manifesting no As toxicity to bacteria. On the other hand, the bacteria show no growth tendency without the nutrition addition of Fe, S needed for the bacteria. In the end of bio-oxidation, bacterium cell number is 2.01��107/mL in the solution.
The crystal structure of Arsenopyrite [FeAsS] is island-base type, and the combination of transition metal Fe with arsenic-sulphur pair-[AsS] is characterized as a ligand. Realgar [As4S4] is argon-type sulphides with double covalent bonds and ring-base crystal structure. It��s composed of As4S4 rings with covalent bonds within rings and molecular bonds between rings[5]. During the bio-oxidation, the arsenopyrite [FeAsS] exhibits rather strong activity; while realgar [As4S4] is inertial. Their chemical behaviors are completely different. Realgar does not dissolve in the bacteria-bearing solution, and there is no toxic arsenic entering the solution. Then the activity of enzyme will not be restrained, Fe3+/Fe2+ is stable and the potential hardly decreases. Further details of the oxidation of arsenopyrite and realgar are analyzed by XRD. Fig.4 shows that the arsenopyrite oxidated by bacteria is completely oxidated, and after As entering the solution, new phases are formed as arsenic oxide(Al2O3) and complex arsenate Pb(Fe2.54Al0.46)-(As1.07O4)(S0.93O4)(OH)6. The XRD pattern of realgar bio-oxidation residue indicates that the residue is As4S4, identical to that of realgar, and the realgar crystal phase keeps the same before and after bio-oxidation.
The valency of As in arsenic-bearing minerals differs much as well, and their chemical behaviors are quite different during bio-oxidation. In the bacteria-bearing solution, arsenopyrite is quickly eroded and decomposed, then Fe2+ and As access the solution to change its electrochemical character. The oxidation of arsenopyrite can be divided into two stages: at the first stage, [AsS]2- of arsenopyrite is oxidated to As(��), which is toxic to bacteria. Due to their good arsenoresistant property, the thermophilic bacteria used in our experiment can grow and multiply rapidly at the middle and later stages, even in the solution of high As content. At the second stage, As(��) is oxidated to As(��) and largely decreases the toxicity of As to bacteria. The As in arsenopyrite is oxidated in the way of [AsS]2-��As(��) ��As(��) to accomplish de-arsenic by bio-oxidation. When As occurs in the form of realgar As4S4, the realgar is hard to be oxidated by bacteria and As can not access the solution, then the electrochemical property of bacteria-bearing solution will not be changed. It can be seen that the existing form of As in arsenic-bearing minerals is important to bacterium metallurgy. When As is formed as the arsenic-sulphur pair��[AsS], the mineral is suitable for the bio-oxidation process of de-arsenic and extraction aurum; however, when the realgar As4S4 exists abundantly, the mentioned process is unfit for realgar-bearing gold mines due to the uneasy deprivation of arsenic element.
5 CONCLUSIONS
1) The chemical behaviors of arsenic-bearing minerals oxidated by thermophilic bacteria are quite different. Arsenopyrite FeAsS and realgar As4S4 differ much during bio-oxidation: the former is active and can be rapidly eroded in bacteria-bearing solution, completely oxidated and the leaching content of As is high over 95%; while realgar is inertial and As in it is hard to be leached, resistant to bacterium corrosion and oxidation.
2) When arsenopyrite is bio-oxidated by leaching bacteria, most crystals dissolve and enter the solution; a little will redeposit and crystallize in the solution to form arsenic oxide(Al2O3) and complex arsenate Pb(Fe2.54Al0.46)(As1.07O4)(S0.93O4)-(OH)6. Comparably, XRD analysis shows that realgar is hardly oxidated.
3) Arsenopyrite is highly toxic to bacteria. At the initial stage the cell number drops to 2.01��105/mL, at the middle and later stages, bacteria grow fast to 2.23��108/mL. In the realgar-bearing solution, the bacterium cell amount decreases slowly within a small range.
4) In the bio-oxidation process, precondition of the process is to detect the As occurrence and valency of As in gold ores(the crystal structure and valency of As in gold mines should be found out first), the arsenopyrite-bearing gold ore is suitable for the bacterium oxidation-de-arsenic-extraction gold process. and if the arsenic exists as realgar As4S4, the above process is unfit for the ores.
REFERENCES
[1]Suzuki I. Microbial leaching of metals from sulfide minerals [J]. Biotechnology Advances, 2001, 19: 119-132.
[2]Blake �� R C, Sasaki K, Ohmura N. Does aporusticyanin mediate the adhesion of Thiobacillus ferrooxidans to pyrite? [J]. Hydrometallurgy, 2001, 59: 357-372.
[3]Breed A W, Glatz A, Hansford G B, et al. The effect of As(��) and As(��) on the batch bioleaching of a pyrite-arsenopyrite concentrate [J]. Minerals Engineering, 1996, 9(12): 1235-1252.
[4]LIU Ying-jun, CAO Li-ming, LI Zhao-lin, et al. Element Geochemistry [M]. Beijing: Science Press, 1984.(in Chinese)
[5]WANG Pu, PAN Zhao-lu, WENG Ling-yu. System Mineralogy [M]. Beijing: Beijing Geological Press,1982.(in Chinese)
[6]YANG Hong-ying, YANG Li, CHEN Gang, et al. Submicro-battery effect and selective bio-oxidation model of gold-bearing arsenopyrite by Thiobacillus ferrooxidans [J]. Trans Nonferrous Met Soc China, 2002, 12(6): 1199-1202.
[7]YANG Hong-ying, YANG Li, WEI Xu-jun. Mechanism on biooxidation of arsenopyrite with Thiobacillus ferrooxidans strain SH-T [J]. The Chinese Journal of Nonferrous Metals, 2001, 11(2): 323-327.(in Chinese)
[8]Jones R A, Koval S F, Nesbitt H W. Surface alteration of arsenopyrite(FeAsS) by Thiobacillus ferrooxidans [J]. Geochimica et Cosmochimica Acta, 2003, 67(5): 955-965.
[9]Ehrlich H L. Bacterial oxidation of arsenopyrite and enargite [J]. Econ Geol, 1964, 59: 1306-1312.
[10]Buckley A N, Walker G W. The surface composition of arsenopyrite exposed to oxidizing environments [J]. Appl Surf Sci, 1988, 35: 227-240.
[11]WANG Jin-xiang, HENG Dong-hui, WANG Li-qun. Primenton column leaching of gold from As-bearing refractory gold ore in Luerba by bacterial oxidation [J]. Hydrometallurgy of China, 2000, 19(3): 9-12.(in Chinese)
[12]MAO Zai-sha, LI Xi-ming, LUAN Mei-lang, et al. Experiments on bacterial dearsenication of refractory arsenical gold concentrate and gold leaching by cyanidation [J]. Engineering Chemistry & Metallurgy, 1996, 17(3): 200-204.(in Chinese)
[13]XU Hai-yan, YAN Wang-ming, LIU Zhen-ying, et al. Construction of antiarsenical vector and expression in Thiobacillus ferrooxidans [J]. Chinese Journal of Applied & Environmental Biology, 1995,1(3):238-243.(in Chinese)
[14]Willsky G P, Malamy M H. Effect of arsenate on inorganic phosphate transport in B.coli [J]. Journal Bacterial, 1981, 144: 366-374.
(Edited by PENG Chao-chun)
Foundation item: Projects(50274024; 50074015) supported by the National Natural Science Foundation of China; Project(20022022) supported by the Natural Science Foundation of Liaoning Province Received date: 2004-11-24; Accepted date: 2005-02-26
Correspondence: YANG Hong-ying, Professor, PhD; Tel: +86-24-83680373; E-mail: hyyang@mail.neu.edu.cn