
Bacteria adherence property of molybdenum nitride modified layer on Ti6Al4V alloy
FAN Ai-lan(������), TIAN Lin-hai(���ֺ�), QIN Lin(�� ��), TANG Bin(�� ��)
Research Institute of Surface Engineering, Taiyuan University of Technology, Taiyuan 030024, China
Received 15 July 2007; accepted 10 September 2007
Abstract: Molybdenum nitride (Mo-N) modified layer on Ti6Al4V alloy was obtained by the plasma surface alloying technique. The bacteria adherence property of the Mo-N modified layer on Ti6Al4V alloy on the oral bacteria Streptococcus Mutans was investigated and compared with that of Ti6Al4V alloy by fluorescence microscopy. The mechanism of the bacteria adherence was discussed. The sample was characterized by X-ray diffractometry (XRD), X-ray photoelectron spectroscopy (XPS) and rough-meter. The results show that the Mo-N modified layer is composed of phase Mo2N (fcc) and Mo2N (tetr). There are Mo 3d, N 1s, C 1s and O 1s in the Mo-N modified layer Ti 2p, O 1s, C 1s, in the Ti6Al4V alloy. The surface roughness (Ra) of Ti6Al4V alloy and the Mo-N modified layer is (0.06��0.01) ��m and (0.16��0.01) ��m, respectively. The Mo-N modified layer inhibits the bacteria adherence. Mo and N on surface of modified layer play a vital role in inhibiting the bacteria adherence.
Key words: Ti6Al4V; Mo-N modified layer; bacteria adherence property
1 Introduction
Titanium and titanium alloys, especially Ti6Al4V, are extensively used in such in vivo applications as dental orthopaedic and cardiovascular surgery, which is due to their remarkable biocompatibility, low density, excellent mechanical and anti-corrosion properties [1-4]. However, the applications of titanium and titanium alloys are limited due to low surface hardness and low wear resistance. Until now, many surface treatment methods have been applied to improve the tribological property of titanium alloys [5, 6]. The Mo-N surface modified layer on Ti6Al4V substrate with plasma surface alloying technique can effectively improve the antifriction and antiwear properties of titanium alloy [7]. But there is less work concerning the bacteria adherence of the Mo-N surface modified layer. It was reported that the invalidation of biomaterial was caused mainly by biomaterial centered infection (BCI), and bacterial adhesion on biomaterial surface was the initiating factor leading to this infection[8-10].
It is necessary to study the bacteria adherence of the Mo-N surface modified layer. In the work, the Mo-N surface modified layer on Ti6Al4V substrate was obtained by the plasma surface alloying technique. The bacteria adherence properties of the Mo-N surface modified layer on Ti6Al4V substrate on the oral bacteria Streptococcus mutans was investigated and compared with those of Ti6Al4V alloy.
2 Experimental
The substrate material was annealed Ti6Al4V alloy with 16 mm in diameter and 2 mm in thickness. The samples were abraded and polished to mirror finish using alumina paste and then cleaned in acetone. The device for experiment was the LS-750 plasma surface alloying device that was described in Ref. [7]. The process parameters were as follows: the Ar�CN2 mixture gas pressure was 35 Pa, the source voltage for supplying Mo elements was from -1 200 to 1 220 V, the cathode (specimen) voltage was from -600 to 65 V, the distance from the source target to the substrate sample was 15mm, the process temperature was 850 �� and the process duration was 3 h.
The bacteria adherence properties of the Mo-N surface modified layer on Ti6Al4V substrate and Ti6Al4V alloy were tested using Streptococcus Mutans strain ATCC#25175. Bacteria were precultured in nutrient broth at 37 �� for 24 h, then the Tris-HCl suspension was diluted to approximately 1.5��107/mL with sterilized distilled water. Ti6Al4V alloy and surface modified specimens were put into sterile glass bottles with 5 mL of bacterial suspension and were incubated at 37 �� for 1 h. After incubation, the samples were rinsed with distilled water for six times, fixed in 3% glutaraldehyde at 4 �� for 30 min and stained with 1% acridine orange solution for 30 min. Bacteria number and bacteria morphology on each sample was analyzed using a fluorescence microscopy (OLYMPUS-BX51T- 32000).
The phase structure of the modified layer was analyzed by D/max 2500 X-ray diffraction (XRD) with Cu K��. The surface chemical compositions of the Mo-N modified layer on Ti6Al4V substrate and Ti6Al4V alloy were obtained by X-ray photoelectron spectroscopy (XPS) using ESCALAB 250 spectrometer with Al K�� excitation. The binding energies obtained in XPS analysis were corrected by referencing the C 1s line to 284.6 eV. The surface roughness (Ra) of the Mo-N modified layer and Ti6Al4V alloy were investigated by TR 240 rough-meter.
3 Results and discussion
3.1 Bacteria adherence property
Fluorescence microscope images of bacteria adherence on the Ti6Al4V alloy and the Mo-N modified layer surface are shown in Figs.1 (a) and (b), respectively. The rounded region is the effective observing field of vision of the fluorescence microscope, and the small circles are the individual bacteria adherence on the specimen surface. Fig.2 shows the number of bacteria adherence on the Ti6Al4V alloy and the Mo-N modified layer. Figs.1 and 2 show that the number of bacteria adhered on the Mo-N modified layer is lesser than that on the Ti6Al4V alloy.
3.2 Surface characteristics
In order to review bacteria adherence property, surface structure and composition tests were performed. Fig.3 shows X-Ray diffraction spectrum of the Mo-N modified layer. It can be observed that the Mo-N modified layer is composed of phase Mo2N (fcc) and Mo2N (tetr). Fig.4 shows the XPS spectra of the Ti6Al4V alloy and Mo-N modified layer. For the Ti6Al4V alloy, the main peaks in the spectra include Ti 2p, C 1s and O 1s. For the Mo-N modified layer, the
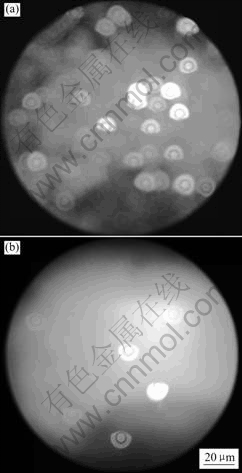
Fig.1 Fluorescence microscope images of bacteria adherence on different substrates: (a) Ti6Al4V alloy; (b) Mo-N modified layer surface

Fig.2 Number of bacteria adhered on Ti6Al4V alloy and Mo-N modified layer
main peaks in the spectra include Mo 3d, N 1s, C 1s and O 1s. JEYACHANDRAN et al [11] confirmed that the chemical composition of the materials might determine the hydrophobicity and electrostatic interactions, resulting in the bacteria adherence through unspecific binding, and it was the principle factor influencing
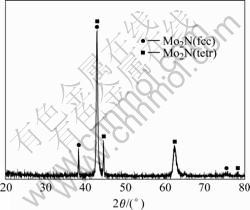
Fig.3 X-ray diffraction pattern of Mo-N modified layer
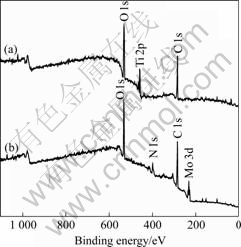
Fig.4 XPS survey spectra for surfaces of (a) Ti6Al4V alloy and (b) Mo-N modified layer
bacteria adherence. Accordingly, based on the result of the analysis, Mo and N on surface of modified layer play a vital role in inhibiting the bacteria adherence.
In order to review another principle factor to bacteria adherence property, surface roughness tests were performed. The results show that the surface roughness (Ra) of polished Ti6Al4V alloy and the Mo-N modified layer is (0.06��0.01) ��m and (0.16��0.01) ��m respectively. The surface roughness of the Mo-N modified layer is greater than that of Ti6Al4V alloy. In general, rough surfaces are believed to support more bacteria adherence. But QUIRYNEN et al [12] confirmed when the surface roughness value (Ra) of the implant base was less than 0.2 ��m, the surface roughness of samples would have little influence on bacteria adherence. So in the present study, the surface roughness of samples would have little influence on bacteria adherence.
In addition to surface chemical composition and surface roughness of the materials, other factors have also significant influence on bacteria adherence. Therefore, the mechanism of bacteria adherence is complex and more research work needs to be performed.
4 Conclusions
1) By employing the plasma surface alloying technique, Mo-N modified layer on Ti6Al4V substrate is obtained.
2) Mo-N modified layer inhibits the bacteria adherence on Streptococcus Mutans.
3) Mo and N on surface of modified layer play a vital role in inhibiting the bacteria adherence.
References
[1] Giglio E D, Guascito M R, Sabbatini L, Zambonin G. Electropolymerization of pyrrole on titanium substrates for the future development of new biocompatible surfaces[J]. Biomaterials, 2001, 22(19): 2609-2616.
[2] Park J Y, Gemmell C H, Davies J E. Platelet interactions with titanium: Modulation of platelet activity by surface topography[J]. Biomaterials, 2001, 22(19): 2671-2682.
[3] Long M, Rack H J. Titanium alloys in total joint replacement��a materials science perspective[J]. Biomarials, 1998, 19(8): 1621-1639.
[4] [4]Sarr�� M I, Moreno D A, Ranninger C, King E, Ruiz J. Fluence of gas nitriding of Ti6Al4V alloy at high temperature on the adhesion of Staphylococcus aureus[J]. Surface & Coatings Technology, 2006, 201: 2807-2812.
[5] Fridrici V, Fouvry S, Kapsa P. Fretting wear behavior of a Cu-Ni-In plasma coating[J]. Surface & Coatings Technology, 2003, 163/164: 429-434.
[6] Liu C, Bi Q. Matthews A. Tribological and electrochemical performance of PVD TiN coatings on the femoral head of Ti-6Al-4V artificial hip joints[J]. Surface & Coatings Technology, 2003, 163/164: 597-604.
[7] TANG Bin, WU Pei-qiang, LI Xiu-yan, FAN Ai-lan, XU Zhong, Celis J P. Tribological behavior of plasma Mo-N surface modified Ti-6Al-4V alloy[J]. Surface and Coatings Technology,2004, 179(2/3): 333-339.
[8] Higuchi K W, Folmer T, Kultje C. Implant survival rates in partially edentulous patients: A 3-year prospective multicenter study[J]. Oral Maxillofac Surg, 1995, 53(3): 264-268.
[9] Gottenbos B, Henny C, Mei V D, Klatter F, Nieuwenhuis P, Henk J. Busscher in vitro and in vivo antimicrobial activity of covalently coupled quaternary ammonium silane coatings on silicone rubber[J]. Biomaterials,2002, 23(6): 1417-1423.
[10] Costerton J W, Stewart P S, Greenberg E P. Bacterial biofilms: A common cause of persistent infections[J]. Science, 1999, 284(5418): 1318-1322.
[11] Jeyachandran Y L, Narayandass Sa K, Mangalaraj D, Bao C Y, LI W, Liao Y M, Zhang C L, Xiao L Y, Chen W C. A study on bacterial attachment on titanium and hydroxyapatite based films[J]. Surface & Coatings Technology, 2006, 201(6): 3462-3474.
[12] QUIRYNEN M, BOLLEN C M, PAPAIOANNOU W, et al. The influence of titanium abutment surface roughness on plaque accumulation and gingivitis: Short-term observations [J]. Int J Oral Maxillofac Implants, 1996, 11(2): 169-178.
(Edited by CHEN Can-hua)
Foundation item: Project(50501016) supported by the National Natural Science Foundation of China; project(2007AAO3Z521) supported by the National High-tech Research and Development program; Projects (20051049, 2006021023) supported by the Natural Science Foundation of Shanxi Province, China; Project(2006-27) supported by the Foundation for Young Subjects Leaders in University of Shanxi and Scientific Foundation for Returned Overseas Scholars of Shanxi Province, China
Corresponding author: TANG Bing, Professor, PhD; E-mail: tangbin@tyut.edu.cn