
Electro-generative mechanism for simultaneous leaching of pyrite and MnO2 in presence of A. ferrooxidans
XIAO Li(肖 利)1, 3, FANG Zheng(方 正)1, QIU Guan-zhou(邱冠周)2, LIU Jian-she(柳建设)2
1. School of Chemistry and Chemical Engineering, Central South University, Changsha 410083, China;
2. School of Resources Processing and Bioengineering, Central South University, Changsha 410083,China;
3. Hunan Metallurgic Professional Technical College, Zhuzhou 412000, China
Received 15 July 2007; accepted 10 September 2007
Abstract: A dual cell system was used to study the output power, output voltage, galvanic polarization of anode and cathode, and the relationship between the electric quantity(Q) and some factors, such as the dissolved Fe2+ magnitude, the time in the electro- generative simultaneous leaching with bacteria(BEGSL) and without bacteria(EGSL). A three-electrode system was adopted to study their individual self-corrosion current, which was smaller compared with the galvanic current. The results show that the output power and voltage in BEGSL are higher than those in EGSL. The accumulated sulfur on the surface of sulfides produced in BEGSL can be oxidized by A. ferrooxidans, and the ratio of biologic electric quantity reaches 51.50% in 72 h. The first stage both in EGSL and in BEGSL is the dissolution of pyrite on the surface to ferrous ion and sulfur element, which was oxidized by A. ferrooxidans in the further procedure.
Key words: pyrite; MnO2; bio-oxidation; simultaneous leaching; electro-generation
1 Introduction
Many sulfides of metal such as galena, sphalerite, chalcopyrite, and pyrite, are semiconductors. When two kinds of such minerals contact each other in an electrolyte, a galvanic couple, where the mineral of lower rest potential as anode, and that of higher rest potential as cathode forms. Manganese dioxide is also a semiconductor with much higher rest potential than all sulfides mentioned above, so that a galvanic couple in which both the minerals would dissolve simultaneously can form, when it contacts with any of the sulfides.
The simultaneous leaching of metal oxides and sulfides, such as ZnS-MnO2[1-2], CuFeS2-MnO2[3-5], and FeS2-MnO2[6-10] has been investigated. RATH et al[2] have examined the galvanic interaction between sphalerite and MnO2 and observed that the rates of corrosion of the two minerals are much higher than their self-corrosion rates in the individual anodic and cathodic insides. Similarly, the galvanic current between CuFeS2-MnO2[5] and FeS2-MnO2[10] in dilute sulfuric acid solutions has also been reported as high as about 2.40 and 1.7 mA/cm2, respectively. But little work has been reported on simultaneous leaching with electro- generation. Therefore, the study has been done on the electro-generatively simultaneous leaching(EGSL) of MnO2 and sulfide minerals[11-13].
It is well known that chemical energy releases as heat in the oxidation leaching of sulfide. If the process is carried out by electro-generation, the heat released can transform to useful electrical energy. XIAO et al[12] have pointed out that the surface of leached sulfides covered by the accumulated sulfur, thwarting the anodic reaction to continue and greatly reducing the output of electric energy in EGSL. It is also shown by a leaching experiment for chalcopyrite[5] that the sulfur film forms on the mineral surface during anodic dissolution, presenting the passivity regions. TAKAMI et al[14] have proposed that the removal of elemental sulfur may improve the galvanic reaction rate, and dissolution rates are enhanced by the Acidithiobacillus ferrooxidans (A. ferrooxidans) in the simultaneous leaching system of zinc sulfide and manganese dioxide.
A dual cell set and A. ferrooxidans were used to the simultaneous leaching with electro-generation in this work, which was called as bio-electro-generative simultaneous leaching(BEGSL). The electro-generative mechanism of FeS2-MnO2 in the presence of A. ferrooxidans was studied, by the galvanic electric quantity and BEGSL was compared with the electro- generative simultaneous leaching(EGSL).
2 Experimental
2.1 Principle
In the simultaneous leaching of FeS2-MnO2 in dilute HCl, it has been confirmed that there are two corrosion couples, MnO2/Fe2+ and FeS2/Fe3+, which depend on each other[9]. The corrosion reactions of the couples are as follows:
To MnO2/Fe2+,
Cathode:
MnO2(s)+4H+(aq)+2e→Mn2+(aq)+2H2O (1)
Anode:
Fe2+→Fe2++e (2)
To FeS2/Fe3+,
Cathode:
Fe3++e→Fe2+ (3)
Anode:
FeS2→Fe2++2S0+2e (4)
and/or
 (5)
(5)
2.2 Minerals
The selected pyrite was a natural hand-sorted ores from a domestic mine. The XRD analysis showed that FeS2 was predominant in the ores, and the size was (17.8±5.3) μm. The element analysis is listed in Table 1. The manganese oxide was commercial reagents (Shanghai Chemical Regents Industries, Ltd.).
Table 1 Chemical composition of pyrite (mass fraction, %)

2.3 Set and electrodes for electro-generative leaching
The set used was an electrolysis cell made of PVC in water bath, which was divided into two compartments, anolyte and cathlyte, each of 200 mL, connected by anion membrane, across which anions migrated freely, and the ferrous ions dissolved from FeS2 in anode room did not act MnO2 directly in cathode room. The dissolution was electrochemical in nature and followed the principles of corrosion, i.e. reactions (4) or (5) for anode and reaction (1) for cathode.
The anode and cathode were made of the powders of pyrite and MnO2, respectively. Two air-blowing tubes were inserted into anolyte and catholyte, respectively, to supply the oxygen for bacteria and to agitate. The pH value, the electrode potentials (vs SCE) and the output voltage of the leaching cell were measured with a PHS-3C digital acidimeter, the output current with an ampere meter and the concentration of oxygen with a Degussa oxygen meter. All of the measured instruments were calibrated before each run.
2.4 Solutions and bacteria
The bacteria culture medium consisted of (NH4)2SO4 3.0 g/L, KCl 0.1 g/L, K2HPO4 0.5 g/L, MgSO4?7H2SO4 0.5 g/L and Ca(NO3)2 0.01 g/L. To compare BEGSL with EGSL, the medium was acidified first to pH 1.8 by H2SO4, and then taken as anolyte of EGSL, and the same nutrient medium with exponential growth phase bacteria was used as anolyte of BEGSL.
The bacterial culture used was a pure strain Yunnan 3 (PQ321745) of A. ferrooxidans that was suitable for growth on pyrite ores and was obtained from Yunnan Province of China.
The solutions used for the present experiments were prepared using AR reagents and distilled water. Oxygen concentration in the solution was measured to be 5.9 mg/L.
2.5 Self-corrosion measurements
A three-electrode system with a working electrode of MnO2 or pyrite-graphite paste, a platinum counter and a saturated calomel(SCE) reference was used for self-corrosion measurements. Polarization was measured with the scanning rate of 0.1 mV/s by the CHI660B electrochemical workstation with a computer. All potentials presented in this work were against SCE.
3 Results and discussion
3.1 Galvanic current-power-potential
The galvanic current, power, and potential in BEGSL and EGSL were measured under the condition of the various exterior loads. Each run was first set up under the open-circuit, then the voltage and current outputs were measured under the circuit-closed conditions of changing the exterior resistance from 1 000 to 0 Ω. When the exterior load approached zero, the current was maximum and the galvanic potential was minimum due to the interior resistance of solution, ion membrane and electrode.
The results are shown in Fig.1, which indicates that the useful power and galvanic potential in the presence of A. ferrooxidans are higher than those in the absence of the bacteria. The load of 8 Ω corresponding to the maximum powder was selected for the next electro-generative experiments to ensure the maximum power of the galvanic cell.

Fig.1 Output current―power―potential of EGSL and BEGSL for pyrite-MnO2
3.2 Anodic and cathodic polarizability in EGSL and BEGSL
The Evans diagram, anodic and cathodic polarization, and interior resistance of the cell were used to study the control factor in EGSL and BEGSL processes. Generally, the bigger the value of resistance, the greater the effect on the leaching reaction.
As shown from Fig.2, the currents increase with the over-potential of cathode ?εc and the over-potentials anode ?εa. We define polarization as
 (6)
(6)
where ?ε is the over-potentials, and I is the galvanic current.

Fig.2 Evans diagram of EGSL and BEGSL for pyrite-MnO2
The polarizations of anode and cathode are Pa and Pc:
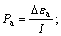
 (7)
(7)
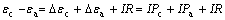 (8)
(8)
 (9)
(9)
where R is the interior resistance and Pa, Pc, R and I are listed in Table 2. The over-potentials of anode and cathode increase with the decrease of galvanic voltage for 36 h in SLEG and BEGSL processes.
Table 2 Pc, Pa, R and J of EGSL and BEGSL for pyrite-MnO2
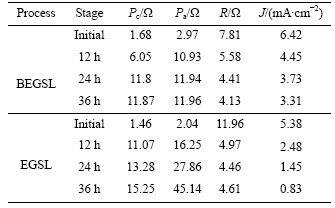
It can be seen from Table 2 that the interior resistance R is larger than Pa and Pc when the leaching is initiated, and follows by drop to a steady value. Perhaps, the reason is that the interface resistance between electrolyte and pyrite decreases as the reaction comes to steady state.
Another important feature of polarization reflects to the data of different stages (see the columns 3, and 4 in Table 2). Pa and Pc are smaller at first due to the electric catalytic activity of acetylene black [15-16]. Compared with EGSL, the polarization Pa comes to a steady-value about 10-11 Ω with the decrease of the galvanic current in BEGSL process as the reactions are continued. This could be attributed to the fact that elemental sulfur accumulated in the anodic electrode stops reaction in EGSL, while be oxidized by A. ferrooxidans in BEGSL. The increase of polarization Pc in both processes might be the reason for the decrease in acidity of the solution.
3.3 Self-corrosion for pyrite and MnO2 electrodes
In addition to the galvanic current in EGSL and BEGSL, there also exists the self-corrosion in two electrodes, respectively. Comparison of the galvanic current flow with the self-corrosion current could estimate which one is predominant. A convenient way to determine the self-corrosion is to plot Tafel curves of the individual half-cells. The Tafel curves of pyrite anode with or without A. ferrooxidans and MnO2 cathode in 1 mol/L H2SO4 are shown in Fig.3. Ecorr, Jcorr, and Rp obtained from the Tafel curve s are listed in Table 3.
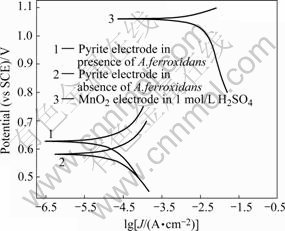
Fig.3 Tafel curves
Table 3 Ecorr, Jcorr and Rp calculated from Tafer curves
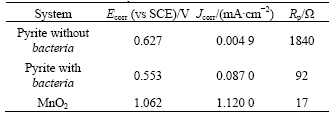
For pyrite, the self-corrosion current Jcorr as seen from Table 3 is negligible compared the galvanic current I shown in Table 2, and for MnO2, the self-corrosion current almost same magnitude as the galvanic current. Accordingly, the galvanic interaction for pyrite predominates over their individual self-corrosion whether in EGSL or BEGSL processes.
3.4 Electric quantity and leaching ratio in EGSL and BEGSL processes
Assuming that the all-transferred charge is due to 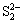 to S0 and is considered the theoretic electric quantity(TEQ), which can be calculated by the Faraday’s law. However, the measured electric quantity (MEQ) in BEGSL process is larger than the theoretic one. This means that the transferred charge is not only
to S0 and is considered the theoretic electric quantity(TEQ), which can be calculated by the Faraday’s law. However, the measured electric quantity (MEQ) in BEGSL process is larger than the theoretic one. This means that the transferred charge is not only 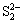 to S0 but also part of S0 to sulfate group that is called as biologic electric quantity(BEQ). BEQ is defined as the difference between the measured and the theoretic one. The ratio of BEQ to the MEQ can be used to predict the progress of BEGSL process. Fig.4 shows the relationships between the MEQ and reaction time for 12 h in EGSL and BEGSL processes.
to S0 but also part of S0 to sulfate group that is called as biologic electric quantity(BEQ). BEQ is defined as the difference between the measured and the theoretic one. The ratio of BEQ to the MEQ can be used to predict the progress of BEGSL process. Fig.4 shows the relationships between the MEQ and reaction time for 12 h in EGSL and BEGSL processes.
It can be seen from Fig.4, the MEQ in EGSL and BEGSL processes is linear with respect to time respectively, and the equations are given as follows:
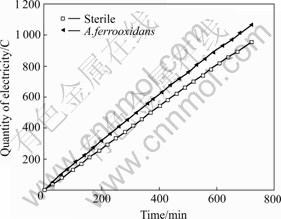
Fig.4 Relationship between measured electric quantity and time in 12 h for EGSL and BEGSL
Q(EGSL)=0.966 51+1.333 36 t, R=0.999 93 (10)
and
Q(BEGSL)=17.760 77+1.478 49 t, R=0.999 65 (11)
From the slopes of the straight lines it can be seen that A. ferrooxidans accelerate the oxidative reaction resulting in the increase of the MEQ. But, the advantages in BEGSL are unremarkable in 12 h. The leaching time in both processes further lasted until the output current was below 5 mA. Table 4 shows the relationship between time and such indexes as the dissolved ferrous and manganese ion ratio, TEQ and MEQ in EGSL after 12 h. Table 5 lists the BEQ and rate of BEQ to the measured one in BEGSL process.
Table 4 Relationship between time and such indexes as dissolved ferrous and manganese ion rate, TEQ and MEQ in EGSL after 12 h

As observed from Table 4, the difference between TEQ and MEQ is very small, indicating that the proceed reaction in EGSL accords with reaction (4). The overall reaction in EGSL is as follows:
MnO2(s)+4H+(aq)+FeS2→
Mn2+(aq)+Fe2+(aq)+2S(s)+2H2O (6)
Comparing Table 4 with Table 5, it can be seen that the amount of ferrous and manganese ions and the output
Table 5 Relationship between time and such indexes as dissolved ferrous and manganese ion rate, BEQ and rate of BEQ in BEGSL after 12 h
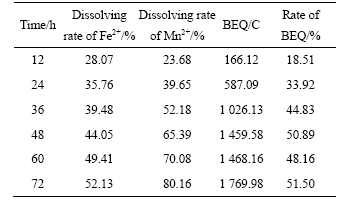
electric quantity in BEGSL is larger than that in EGSL. After 12 h in BEGSL, the ratio is 18.51%, which shows that the bacterial oxidation on the surface of pyrite has initiated. Subsequently, the increase in the ratio of BEQ with the increase of time, is up to 51.50% for 72 h, which indicates that the transferred charge due to the bacterial oxidation is more than that of EGSL process.
3.5 Mechanism of electro-generation for pyrite in presence of A. ferrooxidans
Fig.5 shows the SEM images of the oxidation debris after 24 h in BEGSL and EGSL to study the changes occurring on the surface of pyrite.

Fig.5 SEM images of oxidation debris for pyrite: (a) EGSL; (b) BEGSL
It can be seen from Fig.5 that there is an obvious difference of the oxidation debris of pyrite in EGSL and BEGSL. For the EGSL, a large quantity of floccules accumulate on the surface of the ores, while for BEGSL with few floccules. The mole fraction of Fe and S atoms measured by AAS is listed in Table 6. The increase of element S and the decrease of Fe compared with that before leaching, and the quantity of element S in the sample of EGSL for 24 h is larger than that in BEGSL, which indicates that the produced sulfur is oxidized by A. ferrooxidans in BEGSL process.
Table 6 Mole fraction of main element for pyrite before and after leaching

The data of SEM leached residues indicate that the reaction mechanism in BEGSL could be supposed as a successive reaction of two independent sub-processes. First is that the pyrite dissolves and produces the ferrous ion and elemental sulfur as shown in reaction (4), and second is that elemental sulfur is consecutively oxidized by A. ferrooxidans to sulfate group according to the following reaction:
 (13)
(13)
As mentioned above, the rate of BEQ in the second stage is low, and the sulfur accumulates on anode in BEGSL, which appears to be the controlling step in BEGSL process.
4 Conclusions
1) The electric power and galvanic potential in the presence of A. ferrooxidans are higher than those in the absence of the bacteria.
2) The galvanic current for the pyrite electrode in EGSL and BEGSL processes predominates over its self-corrosion process.
3) The leaching rate and the electric quantity output in BEGSL are larger than those in EGSL, and the rate of BEQ is as high as 51.50% for 72 h BEGSL, which indicates that the charge transferred by the bacterial oxidation is more than that of pure EGSL process.
4) The mechanism in BEGSL process is determined by two independent sub-processes: pyrite is oxidized to sulfur in the initial stage of oxidation, elemental sulfur is oxidized to sulfate group by A. ferrooxidans in the following step, and the latter one is the controlling step.
References
[1] MADHUCHHANDA M, DEVI N B, RATH P C, RAO K S, PARAMGURU R K. Leaching of manganese nodule in hydrochloric acid in presence of sphalerite [J]. Canadian Metallurgical Quarterly, 2003, 42(1): 49-60.
[2] MADHUCHHANDA M, DEVI N B, RATH P C, RAO K S, PARAMGURU R K. Dissolution of metal values from deep-sea manganese nodule in HC1 medium in presence of sphalerite (Part MM): Metallurgy and material science division [J]. Journal of the Institution of Engineers, 2000, 81(2): 48-51. (in India)
[3] DEVI N B, MADHUCHHANDA M, RAO K S, RATH P C, PARAMGURU R K. Oxidation of chalcopyrite in the presence of manganese dioxide in hydrochloric acid medium [J]. Hydrometallurgy, 2000, 57(1): 57-76.
[4] DEVI N B, MADHUCHHANDA M, RATH P C, RAO K S, PARAMGURU R K. Simultaneous leaching of a deep-sea manganese nodule and chalcopyrite in hydrochloric acid [J]. Metallurgical and Materials Transactions B: Process Metallurgy and Materials Processing Science, 2001, 32(5): 777-784.
[5] GANTAYAT B P, RATH P C, PARAMGURU R K, RAO S B. Galvanic interaction between chalcopyrite and manganese dioxide in sulfuric acid medium [J]. Metallurgical and Materials Transactions B: Process Metallurgy and Materials Processing Science, 2000, 31(1): 55-61.
[6] NAYAK B B, MISHRA K G., PARAMGURU R K. Kinetics and mechanism of MnO2 dissolution in H2SO4 in the presence of pyrite [J]. Journal of Applied Electrochemistry, 1999, 29(2): 191-200.
[7] PARAMGURU R K, KANUNGO S B. Electrochemical phenomena in MnO2-FeS2 leaching in dilute HCl (Part 1): Theoretical derivation of rate equations under various limiting conditions [J]. Canadian Metallurgical Quarterly, 1998, 37(5): 389-393.
[8] PARAMGURU R K, MISHRA K G., KANUNGO S B. Electrochemical phenomena in MnO2-FeS2 leaching in dilute HCl (Part 2): Studies on polarization measurements [J]. Canadian Metallurgical Quarterly, 1998, 37(5): 395-403.
[9] PARAMGURU R K, KANUNGO S B. Electrochemical phenomena in MnO2-FeS2 leaching in dilute HCl (Part 3): Manganese dissolution from Indian Ocean nodules [J]. Canadian Metallurgical Quarterly, 1998, 37(5): 405-417.
[10] PARAMGURU R K, NAYAK B B. Galvanic interaction between manganese dioxide and pyrite [J]. Journal of the Electrochemical Society, 1996, 143(12): 3987-3991.
[11] WANG S F, FANG Z. Electrogenerative leaching of galena with ferric chloride [J]. Minerals Engineering, 2003,16(2): 869-872.
[12] XIAO Li, LIU Jian-she, FANG Zheng, QIU Guan-zhou. Factors affecting output powen in electro-generative leaching system of chalcopyrite [J]. The Chinese Journal of Process Engineering, 2006, 6(4): 576-579. (in Chinese)
[13] WANG S F, FANG Z. Simultaneous electrogenerative leaching of chalcopyrite concentrate and MnO2 [J]. J Cent South Univ Technol, 2006, 13 (1): 49-52.
[14] TAKAMI K, SUENAGA Y I, MIGITA A, TAKAHASHI T. Kinetic model for simultaneous leaching of zinc sulfide and manganese dioxide in the presence of iron-oxidizing bacteria [J]. Chemical Engineering Science, 2000, 55(17): 3429-3436.
[15] WANG S F, FANG Z, WANG Y Y. Application of carbon paste electrode on the electro-generative leaching process of sulfide minerals [J]. Electrochemistry, 2005, 11(1): 77-82.
[16] AHLBERG E, ASBJORNSSON J. Carbon paste electrodes in mineral processing: An electrochemical study of galena [J]. Hydrometallurgy, 1993, 34(2): 171-185.
(Edited by LI Xiang-qun)
Foundation item: Project(2004CB619204) supported by the Major State Basic Research Development Program; Project(50374077) supported by the National Natural Science Foundation of China; Project(07D069) supported by the Education Department Foundation of Hunan Province, China
Corresponding author: QIU Guan-zhou; Tel: +86-731-8660356; E-mail: xiaoli_csu@163.com