J. Cent. South Univ. Technol. (2010) 17: 750-754
DOI: 10.1007/s11771-010-0551-z
Two-step flotation recovery of iron concentrate from Donganshan carbonaceous iron ore
YIN Wan-zhong(ӡ����)1, HAN Yue-xin(��Ծ��)1, XIE Feng(л��)2
1. School of Resource and Civil Engineering, Northeastern University, Shenyang 110004, China;
2. Department of Materials Engineering, University of British Columbia, Vancouver V6T IN6, Canada
? Central South University Press and Springer-Verlag Berlin Heidelberg 2010
Abstract: The flotation of pure and natural carbonaceous iron ore samples in the oleate flotation system was investigated. Starch can depress hematite effectively in a wide pH range, but cannot depress siderite efficiently in neutral conditions. The flotation recovery of pure hematite, siderite, and quartz in the oleate-starch-CaCl2 system is significantly different when the slurry pH varies from 4 to 12. A novel two-step flotation process was developed for the separation of iron concentrate from Donganshan carbonaceous iron ore through which the siderite concentrate is first recovered and the high quality hematite concentrates with relative high iron recovery can be obtained in the second step flotation. The siderite concentrate may be utilized directly or undergo further concentration steps to increase iron grade.
Key words: iron ores; siderite; hematite; reverse anionic flotation; recovery
1 Introduction
Flotation is the usual concentration method employed in iron ore beneficiation. As iron ores commonly occur as oxides such as magnetite (Fe3O4), hematite (Fe2O3) and goethite (FeOOH) with gangue minerals or impurities as siliceous, calcareous siliceous- various, and siliceous and/or calcareous clay [1-2], various flotation routes have been developed and practised. Anionic fatty acids (mainly oleic acid), alkyl sulphate, sulfonates, or hydroxamate were commonly used as collectors for flotation of iron oxides from siliceous gangue materials [3-5]. Some amines and their salts were also used in the reverse cationic flotation process. The starch and its derivatives are proved to be effective depressant and are commonly used in the flotation of iron minerals [6-8]. However, the research work on flotation of carbonate minerals such as siderite (FeCO3) and their potential effect on iron ore flotation is lacking due to their uncommon occurrence [9-10]. The effect of radiation pretreatment by accelerated electrons radiation on floatability of magnesite and siderite with sodium dodecyl sulphate was examined and a decreased consumption of the collecting agent was reported when floating carbonate from sulfide minerals [11]. The reverse flotation of calcite and dolomite from sulfide minerals with oleate as the collector and a mixture of thioglocollic acid and citric acid as the depressant were proposed. Calcite can be selectively floated away from the sulfide minerals between pH 9 and 11 and dolomite can be separated from pyrite but cannot be separated from chalcopyrite under the test conditions [12].
A special case emerged in recent development in Donganshan processing plant, one of the largest iron concentrate producers in China. Previously, iron mainly occurred as hematite (Fe2O3) in the ore and the gangue mineral mainly as quartz [13-14]. The reverse flotation of quartz from the iron ore has been successfully practised for nearly 10 years (Fig.1). Regent RA715, an industrial product with similar formula to sodium oleate, was used as the collector. Starch was used as the depressant for iron minerals and CaO as the activator for quartz. After flotation, iron concentrate with iron grade of about 65% and iron recovery of higher than 70% could be usually produced [15]. However, it was found that the content of carbonaceous minerals typically siderite (FeCO3) in the ore significantly increased (up to 10%) with the development of mining depth. Both iron grade and recovery decreased significantly during treating the newly mined ore. Though strong evidences were still lacking, it was highly suspected that this phenomenon was caused by the presence of carbonaceous iron minerals especially siderite. The geological analysis indicated that millions of tons of such type of iron ore reserve were undermined in the mining area. As a result, there is great need to develop a suitable process for treating these carbonaceous iron ores. In this work, the floatability of three pure minerals (hematite, siderite and quartz) in sodium oleate flotation system was investigated and the effect of siderite on the reverse anionic flotation of quartz from the synthetic iron ore was examined. The feasibility of recovering iron concentrate from Donganshan carbonaceous iron ore by the novel two-step flotation process was studied.

Fig.1 Simplified beneficiation flowsheet of Donganshan flotation plant
2 Experimental
Three natural mineral samples, hematite, siderite and quartz (particle size P80=50 ��m) were supplied by Wuhan Steel Co., Anshan Steel Co. and Benxi Steel Group, respectively. The chemical analysis of the samples indicated that their compositions were close to pure minerals (Table 1). X-ray diffraction (XRD) analysis of the three samples further confirmed the main structure of hematite, siderite and quartz. The mixed mineral samples were obtained by mixing the three natural minerals with different mass ratios. The iron ore samples taken from the feed to the reverse anionic flotation process (a compound iron concentrate from magnetic separation process) in Donganshan processing plant were used for comparison tests. The chemical analysis and XRD analysis of the actual ore sample indicated the occurrence of hematite, siderite and quartz in the ore (Table 1 and Fig.2). The flotation experiment was carried out with the XFG self-aeration mechanical flotation machine. For each flotation test, both the concentrate and the tailings were collected for further analysis. Slurry pH was controlled by direct addition of NaOH and H2SO4 solutions. All the tests were conducted at indoor temperature.
3 Results and discussion
3.1 Effect of siderite on reverse flotation
The effect of siderite on the reverse anionic flotation of the mixed mineral samples was first examined. The samples were made up of hematite, siderite and quartz with different mass ratios and the content of siderite was calculated based on the mineral composition. Sodium oleate was used as the collector and CaO as the activator for quartz. Starch was used for the depression of iron minerals. The results are shown in Fig.3. It shows that the iron recovery decreases significantly with an increase of the siderite content in the mixture. The iron grade in the concentrate also exhibits the same trend. When there is no siderite in the mixed sample, the iron grade and recovery of the iron concentrate are 61.2% and 96.8%, respectively. When the siderite level is increased to 10%, they decrease to 42.3% and 53.2%, respectively. Though the addition of siderite to the mixture slightly decreases the iron grade in the feed and may potentially influence the flotation results, the effect of siderite on the iron grade and recovery of the concentrate is much more significant than expected. The SEM images of the products are shown in Fig.4. Compared with the SEM images of pure minerals (hematite and quartz), some fine particles are observed on the surface of the mineral particles from both the tailings and the concentrate. EDX analysis of four typical points on the particle surface (points A, B, C, and D in Fig.4) indicates the adsorption of fine siderite particles on the surface of quartz and hematite minerals. It is believed that since siderite is much softer than the other two minerals (Mohs hardness of 3.5-4.0 for siderite, 5.5-6.5 for hematite and about 7 for quartz [16]), it turns into slime during ore dressing and may exhibit good adsorption on other minerals, resulting in the floatability of quartz and hematite minerals similar to siderite during flotation. The deteriorated separation of iron concentrate in the anionic reverse flotation process took place.
Table 1 Mineral analysis results of pure minerals and actual iron ore
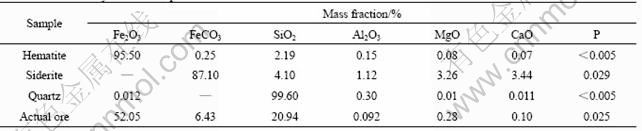

Fig.2 XRD pattern of actual ore sample
3.2 Flotation of pure minerals
The flotation of three pure minerals, namely hematite, siderite and quartz, in oleate-starch system under different pH was examined. The results are shown in Fig.5. It shows that starch can effectively depress the hematite flotation in the pH range of 2 to 12. However, it cannot depress the siderite flotation in the same pH range and shows a peak recovery at pH 7. The results for the flotation of the three minerals in oleate-starch-CaCl2 system are shown in Fig.6. In neutral conditions (pH 5-7), siderite exhibits the best floatability among the three minerals. The recovery of siderite significantly decreases when pH is further increased. When pH is higher than 9, the floatability of quartz significantly increases due to the activation function of calcium ion [2-3]. However, hematite is depressed in the whole range of pH used in the tests (pH 4-12) due to the depression function of starch [5, 8]. The important finding suggests that a possible solution to separation of iron concentrate from Donganshan carbonaceous iron ore by which siderite (or partial) can be first recovered by reverse anionic flotation (sodium oleate and starch) at near neutral pH and a high quality of iron concentrate (hematite) can be obtained through a second stage of flotation in alkaline conditions.
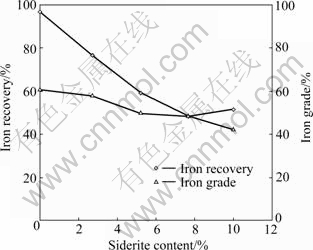
Fig.3 Effect of siderite on iron grade and recovery of iron concentrate (Sodium oleate, 1 500 g/t; CaO 300 g/t; starch, 500 g/t)

Fig.4 SEM images of flotation products: (a) Pure quartz; (b) Tailings from flotation of minerals mixture; (c) Pure hematite; (d) Iron concentrate from flotation of minerals mixture
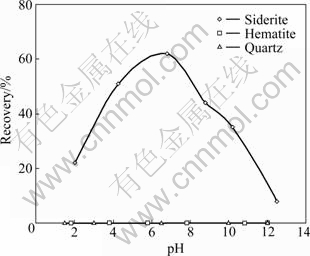
Fig.5 Flotation recoveries of siderite, hematite and quartz as function of pH in presence of 1 500 g/t sodium oleate and 500 g/t starch
Fig.6 Flotation recoveries of siderite, hematite and quartz as function of pH in presence of 1 500 g/t sodium oleate, 500 g/t starch and 500 g/t CaCl2
3.3 Flotation of natural iron ore
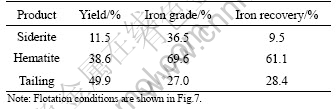
For comparison, the recovery of iron concentrate from Donganshan iron ore sample by direct reverse anionic flotation process was examined. The flotation conditions were same as those shown in Fig.1. The flotation results are shown in Table 2. The iron grade of the concentrate (55.4%) is significantly lower than that of the previous product obtained in the Donganshan plant (which is usually higher than 65%). The iron recovery (69.1%) also shows a slight decrease compared to that (about 72%) in Ref.[15]. A two-step flotation process was thus developed. The process flowsheet is shown in Fig.7. In the first step flotation, a siderite concentrate (or middling) was separated by controlling the slurry pH at 6.0-6.5 with sodium oleate as siderite the collector and starch as the depressant for hematite. In the subsequent flotation of the tailings from the first step flotation, the reverse anionic flotation process was used to float quartz and an iron concentrate (mainly as hematite) was produced. The flotation results are shown in Table 3. It shows that a low grade siderite concentrate (iron grade of 36.5%) is obtained with iron recovery of about 10%. At the second stage of reverse flotation, an iron concentrate is produced with an iron grade of 69.6% and an iron recovery of about 61.1%. Compared with the flotation results of the direct reverse anionic flotation, the total iron recovery of the two processes are similar, however, a high quality of iron concentrate is produced through the two-step flotation process. The yield of the hematite concentrate in the two-step flotation process decreases slightly due to the separation of siderite in the first step. The siderite concentrate may undergo further concentration steps to increase iron grade. The theoretical iron grade of pure siderite is about 48%, and the high quality siderite concentrate may be mixed with high quality hematite concentrates in a suitable ratio to be fed to the iron smelter.
Table 2 Results of reverse anionic flotation of actual iron ore (Sodium oleate, 1 500 g/t; starch, 25 g/t; CaO, 300 g/t; pH, 11.5)


Fig.7 Flowsheet of two-step flotation process
Table 3 Results of two-step flotation of actual iron ore
4 Conclusions
(1) The reverse anionic flotation of pure minerals and the natural carbonaceous iron ore samples indicates that the presence of siderite significantly decreases iron grade and recovery of the iron concentrate. Starch can depress hematite effectively in a wide pH range, but cannot depress siderite efficiently in neutral conditions. Siderite, hematite and quartz exhibit different floatability in the presence of calcium cation when the slurry pH varies from 4 to 12.
(2) A novel two-step flotation process is developed for the separation of iron concentrate from Donganshan carbonaceous iron ore through which the siderite concentrate is recovered in the first stage of flotation and the hematite concentrate can be obtained in the second step flotation. A high quality iron concentrate is produced with a relatively high overall iron recovery. The siderite concentrate obtained through the process may be utilized directly or may undergo further concentration steps to increase iron grade in order to be fed to the iron smelter.
References
[1] RAO K H, Forssberg K S E. Chemistry of iron ore flotation [C]// Froth Flotation: A Century of Innovation. Littleton: SME, 2007: 498-513.
[2] Fuersteanau M C, Han K N. Principles of mineral processing [M]. Littleton: SME, 2003: 245-299.
[3] Fuersteanau M C, Palmer B R. Anionic flotation of oxides and silicates [C]// Flotation. New York: American Institute of Mining and Metallurgical and Petroleum Engineers. 1976: 148-196.
[4] Araujo A C, Viana P R M, Peres A E C. Reagents in iron ore flotation [J]. Minerals Engineering, 2005, 18: 219-224.
[5] Quast K. Flotation of hematite using C6�CC18 saturated fatty acids [J]. Minerals Engineering, 2006, 19: 582-597.
[6] Smith W R., Akhtar S. Cationic flotation of oxides and silicates [C]// Flotation. New York: American Institute of Mining and Metallurgical and Petroleum Engineers. 1976: 87-106.
[7] Peres A E C, Correa M I. Depression of iron oxides with iron starches [J]. Minerals Engineering, 1996, 9: 1227-1234.
[8] Pearse M J. An overview of the use of chemical reagents in mineral processing [J]. Minerals Engineering, 2005, 18: 139-149.
[9] Zhang P, Snow R, Peres A E C, EI-Shall H, EI-Midany A. Depressants in nonsulfide mineral flotation [C]// Froth Flotation: A Century of Innovation. Littleton: SME, 2007: 555-574.
[10] EI-Midany A, EI-Shall H, SOMASUNDARAM P, STANA R, MOUDGIL B. Novel flotation technique for carbonate flotation [C]// Proceedings of Century Flotation Symposium. Brisbane: AusIMM, 2007: 743-747.
[11] Florek I. The effect of radiation pre-treatment on the floatability of magnesite and siderite [J]. Minerals Engineering, 1995, 8: 329-331.
[12] Liu Y, Liu Q. Floatation separation of carbonate from sulfide minerals: I. Floatation of single minerals and mineral mixtures [J]. Minerals Engineering, 2004, 17(7/8): 855-863.
[13] Zhang M, Zhou J. Research on high capacity separation technology for Donganshan refractory iron ore [J]. Metal Mine, 2005(s): 120-127. (in Chinese)
[14] Jiang Y, Yang Y. Study on technological mineralogy of Donganshan refractory ores [J]. Metal Mine, 2006(7): 40-43. (in Chinese)
[15] TIAN J. The concentration mechanism of Anshan Type hematite ore beneficiation process [J]. Metal Mine, 2006(s): 42-46. (in Chinese)
[16] LIDE D R. CRC Handbook of chemistry and physics [M]. 90th ed. Boca Raton: CRC Press, 2010: 212.
Foundation item: Project(2006DFB72570) supported by the Grand Project of International Cooperation of Ministry of Science and Technology of China
Received date: 2009-10-09; Accepted date: 2010-02-12
Corresponding author: XIE Feng, PhD; Tel: 1-604-822-1357; E-mail: xiefeng@interchange.ubc.ca
(Edited by YANG You-ping)