J. Cent. South Univ. Technol. (2010) 17: 1190-1195
DOI: 10.1007/s11771-010-0617-y
Mechanisms of composite agglomeration of fluoric iron concentrate
JIANG Tao(姜涛), HU You-ming(胡友明), LI Qian(李骞), LI Guang-hui(李光辉),
YANG Yong-bin(杨永斌), ZHANG Yuan-bo(张元波), GUO Yu-feng(郭宇峰)
School of Resources Processing and Bioengineering, Central South University, Changsha 410083, China
? Central South University Press and Springer-Verlag Berlin Heidelberg 2010
Abstract: The effect of composite agglomeration process (CAP) on fluoric iron concentrates sintering was investigated. The yield and quality of the sinter are greatly improved when using CAP assisted with heat airflow and enhancing magnesium oxide (MgO) contents. For conventional sintering of fluoric iron concentrate, due to lower viscosity of binding phase and higher fluidity of liquid phase, the sinter is formed with large thin-walled holes and the strength of the sinter is deteriorated consequently. The novel process forms composite agglomerate in which acid pellets are embedded in basic sinter. The pellets are solid with interconnecting crystals of hematite (Fe2O3) and magnetic (Fe3O4). For basic sintering, after adding MgO, the viscosity of the melting phase increases and the fluidity decreases; and calcium and aluminum silico-ferrites and magnesium ferrite are formed with perfect crystals and good sintering microstructure.
Key words: fluoric iron concentrate; sintering; composite agglomeration process; mechanism
1 Introduction
Baiyun’ Ebo iron concentrates, accounting for the majority of the iron-bearing materials used in sinter plant of Baotou Iron and Steel Corp of China, contain a certain amount of fluorine that is able to reduce the melting point of binding phase and lead to low viscosity and high fluidity of melting phase. So, the sinter is inclined to be formed with large thin-walled holes, and strength of sinter is deteriorated consequently [1-3]. Besides, Baiyun’ Ebo fluoric iron concentrates contain low SiO2 content after effective beneficiation, which results in poor quality of sinter due to shortness of binding phase [4-5].
Pelletizing processes were developed to utilize the concentrates in Baotou Iron and Steel Corp of China. The concentrates contain high grade of fluorine and other elements such as sodium and kalium, which lead to the narrow firing temperature range. There comes into being a great number of melt at higher temperature and the compression strength of the pellets is very low, which is not suitable for rotary kiln. The productivity of tunnel kiln is very small, the equipment of traveling grate is very costly, and the pellet production is also limited [6].
An innovative process for preparing iron making burden, named composite agglomeration process (CAP), was developed by JIANG et al [7-8]. In CAP, part or all of the fine-grained iron-bearing materials were separately made into green pellets with diameters of 8- 16 mm in a balling disc or drum, the rest of fine-grained and all of the coarse-grained materials were granulated after being primarily mixed with return fines, fuels and fluxes. Then, the two granular materials were subjected to a secondary mixing, and the secondary mixture was charged onto the sintering strand. After being ignited, sintered and cooled, the mixture was made into composite agglomerate in which acid pellets were embedded in basic sinter. The CAP was applied to Baotou Iron and Steel Corp of China. The results show that, the process overcomes the drawbacks of fluoric iron concentrates for sintering and pellet, achieves low- basicity and high iron grade sintering, improves the bed permeability of material and productivity of sintering machine. Furthermore, for the rational distribution of fuels, the CAP obviously reduces energy consumption. Additionally, because of its high quality, the BF abolished adding silica and its efficiency and effectiveness are greatly increased.
In this work, the CAP of fluoric iron concentrate was studied, and its mechanisms were discussed.
2 Experimental
2.1 Materials
Chemical compositions of the experimental materials are listed in Table 1. There are two kinds of iron-bearing materials: fluoric iron concentrates and imported iron fine ore from Australia (see Table 1). Fluoric iron concentrate is of magnetite, containing 0.34% fluorine, whose total iron grade is as high as 65.50% and its SiO2 content is only 3.22%. Australian iron ore is of hematite, whose total iron grade is 61.30% and its SiO2 content is 4.16%. The size distribution of ores is listed in Table 2. The mass fraction of fluoric iron concentrate with size less than 0.074 mm accounts for 96.20%.
2.2 Methods
Tests were conducted in a laboratory sinter pot with a diameter of 180 mm, a hearth layer with a depth of 20 mm was used and sintering bed height was fixed at 700 mm, and the mass ratio of fluoric iron concentrate to imported ore was fixed at 85:15. For traditional sintering process (TSP) tests, all the materials, including fine- grained concentrates, coarse-grained ore fines, return fines, fluxes and fuels were firstly proportioned, mixed, and the moist mixture went on being granulated by a 600 mm ×1 200 mm drum into granular material, and then charged into the sintering pot. After ignited, sintered, and cooled, sinter cake was discharged from the pot; the finished sinter was obtained after crushed and screened. Main performances were measured according to the relative criteria finally.
In CAP tests, 45% of fluoric iron concentrate was made into 8-16 mm green pellets with addition of bentonite; and the rest fluoric iron concentrate, i.e., coarser fine ores, fluxes, fuels and return fines, was mixed and granulated. Then, the two granular materials were mixed with each other and the mixture was fed into sintering pot.
The microstructure of the sinter was analysed by Labor Lux 12 PDL reflection microscope.
3 Results and discussion
3.1 Sintering test
Table 3 lists the results under different sintering conditions. Test A is the result of TSP. The sintering parameters are: 3% burnt lime ratio, 30% return fine, 8.6% moisture of mixture, 5.5% coke dosage, igniting at (1 100±50) ℃ for 1 min under suction pressure 5 kPa, heat preservation at (1 000±50) ℃ for 1 min under sinter suction pressure 10 kPa and cooling time 3 min. It can be seen that the strength of the sinter is as low as 57.71% even if the basicity is 2.2.
Table 1 Chemical compositions of sintering materials (Mass fraction, %)

Table 2 Size distribution of materials (Mass fraction, %)
Table 3 Results of TSP and CAP tests
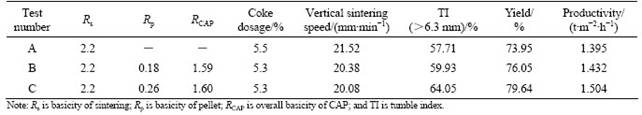
The effect of CAP, test B, was carried out at overall basicity of 1.59 (the basicity of pellet is 0.18), and other parameters were kept the same as those in TSP (test A). It can be seen that the yield and quality of CAP are improved at low basicity of 1.59 and coke dosage of 5.3%. TI increases from 57.71% to 59.93% and yield increases from 73.95% to 76.05%. Furthermore, the iron grade is improved from 58.32% to 59.84% (see Table 4).
Table 4 Chemical compositions of tested samples (Mass fraction, %)

However, it can be observed from Fig.1 that over-melting band still exists even if the yield and quality of CAP increase during test B. And the mixture, especial the pellets in the top layer, is undersintering. Over-melting is brought about by CaF2, which is a kind of low melting point mineral, and always forms lowstrength cuspidite during sintering [9]. According to the CaO-MgO-SiO2 ternary phase diagram, melting point will gradually raise with the increase of MgO content at a fixed basicity, the increase of viscosity of liquid phase and the decrease of fluidity [10-12]. The problem of undersintering is brought about by short of heat [13]. Sintering with aid of hot air is a useful method to supply heat for the upper layer and to improve sinter quality [14-15].

Fig.1 Schematic diagram of undersintering and over-melting bands during CAP
The effect of MgO (adjusting sinter and pellet MgO content to 2.0%) and hot air (300 ℃ for 15 min ) on CAP, test C, is also listed in Table 3. The TI of CAP reaches 64.05%, yield reaches 79.64%, and the productivity goes up to 1.504 t/(m2・h).
3.2 Mechanism of CAP
Fig.2 shows the microstructures of sintering (sample A in Table 4). Because sinter basicity is high, more calcium ferrite with perfect crystal structure is formed (Figs.2(a) and (b)). But large thin-walled holes (Fig.2(c)) and big cracks (Fig.2(d)) are formed in sinter due to lower viscosity and higher fluidity of binding phase. In addition, the strength of binding phase is poor and cracks are formed during cooling. So, the quality of sinter, especial the TI, is worse.

Fig.2 Microstructures of sintering (sample A in Table 4) obtained by TSP: (a) Calcium ferrite; (b) SFCA; (c) Large thin-wall holes; (d) Big cracks
Figs.3-4 respectively show the macrostructure and microstructure of sample B in Table 4. Although majority of fluoric iron concentrates are made into pellets to overcome the harmful effect on sintering in CAP, the rest of fluoric iron concentrates added in the sintering mixture also affect the quality of sinter. It can be seen from Figs.3-4 that there are flow-off marks of thick liquid and large thin-walled holes. In addition, the consolidation strength of binding phase is weaker and sinter and pellet are almost separated (Fig.3 and Figs.4(b), (c) and (d)). Therefore, the quality of CAP products is still poor.
The microstructures of the finished composite agglomerate (FCA) are greatly changed (Fig.5) after adding dolomite and aiding with hot air. It can be seen from Fig.5(a) that Fe2O3 recrystallization increases in pellets and almost all of Fe2O3 and Fe3O4 crystal grains connect each other. Silicate and magnesium ferrite are filled in the interval of Fe3O4 crystals. Therefore, the microstructure of pellet is perfect and the strength is improved. The microstructure of high basicity sinter is also perfect (Figs.5(b)-(c)). Magnesium ferrite increases and is well connected with other iron minerals, such as Fe2O3. Furthermore, calcium and aluminum silico- ferrites are formed with the increase of the MgO content, whose crystals are as column and melting- shaped, which is helpful for improving strength of the sinter. In addition, the viscosity of melting phase is increased and its fluidity is decreased, and sinter and pellet are closely interconnected (Fig.6).

Fig.3 Macrostructure of CAP (sample B)

Fig.4 Microstructures of CAP (Sample B in Table 4): (a) Large thin-walled holes; (b) Sinter parts; (c) Fe3O4; (d) Pellet parts

Fig.5 Microstructures of CAP (Sample C in Table 4): (a) Microstructure of pellet; (b) SFCA, magnesium ferrite; (c) Fe2O3; (d) Interconnected sinter and pellet

Fig.6 Macrostructure of CAP (Sample C in Table 4)
4 Conclusions
(1) Iron concentrate containing fluorine is a typical inferior sintering material. TI and productivity of its sinter are very low. CAP greatly improves sintering performance of fluoric iron concentrate. The product with 64.05% TI, 79.64% yield and 1.504 t/(m2・h) productivity is attained by CAP with supplementary heat of hot air and the increase of MgO content.
(2) In TSP, the sinter is formed with large thin-walled holes and big cracks, due to low viscosity and high fluidity of melting phase bearing fluorine. In CPA, the product is the composite agglomerate, in which acid pellets are embedded in basic sinter, and the pellets are consolidated by solid phase bonding. After adding MgO, the viscosity of the melting phase is increased and the fluidity is decreased, and a large amount of calcium and aluminum silico-ferrites and magnesium ferrite are formed, which are crystallized perfectly, and the microstructure of CAP product is perfect.
References
[1] LI Guang-sen. Basic characteristics of binding phase in sintering ore containing fluorine [D]. Shenyang: Northeastern University, 2006: 20-35. (in Chinese)
[2] BAI Jing-bo, LUO Guo-ping, HAO Zhi-zhong, WU Hu-lin. Study of the influence of fluorine on basic sintering characteristics of Baiyunebo iron ore [J]. Journal of Inner Mongolia University of Science and Technology, 2008, 27(1): 10-14. (in Chinese)
[3] LUO Guo-ping, SUN Guo-long, ZHAO Yan-xia. Experimental study on basic sintering characteristics of Baogang Iron Ore Powder [J]. The Chinese Journal of Process Engineering, 2008, 8(1): 198-204. (in Chinese)
[4] SUN Guo-long, WU Sheng-li. Integrated technique of intensified operation on BF with particular ores at Baotou Steel [J]. Iron and Steel, 2007, 42(11): 21-26. (in Chinese)
[5] LI Guang-sen, JIN Ming-fang, CHU Man-sheng, LI Xiao-gang, SHEN Feng-man. Self-strength of silicate binding phase in fluorine- bearing sinter [J]. Journal of Iron and Steel Research, 2008, 20(1): 10-13. (in Chinese)
[6] WU Hu-lin. Technical progress on iron making system of Baogang [J]. Science and Technology of Baotou Steel (Group) Corporation, 2004, 30(3): 10-16. (in Chinese)
[7] JIANG Tao, LI Guang-hui, HU You-ming, ZHANG Ke-cheng, WU Jie-peng, HUANG Zhu-cheng, FAN Xiao-hui, ZHU De-qing, ZHANG Yuan-bo, XU Bin, GUO Yu-feng, YANG Yong-bin, DONG Hai-gang, LIU Mu-dan, PAN Jian, ZHU Zhong-ping, WANG Hai-tao. Composite Agglomeration process (CAP) of iron ores: China, ZL200510032095.6 [P]. 2009-11-05.
[8] LI Guang-hui, ZENG Jing-hua, JIANG Tao, LI Qian, YANG Yong-bin, WANG Rui-jun, WU Hu-lin. Study and application of composite agglomeration process of fluoric iron concentrate [J]. Journal of Iron and Steel Research International, 2009, 16: 149-153.
[9] WU Hu-lin, XUE Xiang-xin, DUAN Xiang-guang. Optimization of metallurgical properties of complex bf burdens [J]. Iron and Steel, 2006, 41(6): 5-8. (in Chinese)
[10] JIANG Xin, WU Gang-sheng, JIN Ming-fang. Effect of MgO on softening and melting properties of sinter [J]. Journal of Northeastern University: Natural Science, 2006, 27(12): 1358-1361. (in Chinese)
[11] JIANG Xin, WU Gang-sheng, WEI Guo, LI Xiao-gang, SHEN Feng-man. Effect of MgO on sintering process and metallurgical properties of sinter [J]. Iron and Steel, 2006, 41(3): 8-11. (in Chinese)
[12] L? Zhi-yi, CHEN Ge, SHEN Mao-sen. Experimental research on pellet containing fluorine and MgO [J]. Science and Technology of Baotou Steel (Group) Corporation, 2008, 34(4): 4-6. (in Chinese)
[13] HUANG Zhu-cheng, JIANG Yuan, MAO Xiao-ming, XU Bin, GUO Yu-feng, JIANG Tao. Fuel appropriate distribution in iron ore sintering [J]. Journal of Central South University: Science and Technology, 2006, 37(5): 884-890. (in Chinese)
[14] QIU Jin-hou, HE Xiao-yi, HU Xiao-song. Application of hot-blast air sintering at No.1 work shop of iron-making plant of Baotou Steel Corp [J]. Science and Technology of Baotou Steel (Group) Corporation, 2006, 32(6): 8-10. (in Chinese)
[15] LING Zi-yu, SUN Tao-qi, DOU Bao-fen, SHEN Ai-min. Whole project for using waste heat of large-scale sintering equipment [J]. Energy for Metallurgical Industry, 2007, 26(4): 49-51. (in Chinese)
(Edited by CHEN Wei-ping)
Foundation item: Project(50725416) supported by the National Science Fund for Distinguished Young Scholars
Received date: 2010-03-22; Accepted date: 2010-06-19
Corresponding author: JIANG Tao, Professor; Tel: +86-731-88877656; E-mail: jiangtao@mail.csu.edu.cn