
Preparation of boric acid from low-grade ascharite and recovery of magnesium sulfate
LI Jie(李 杰)1, FAN Zhan-guo(樊占国)1, LIU Yan-li(刘艳丽)1,
LIU Su-lan(刘素兰)1, JIANG Tao(姜 涛)1, XI Zheng-ping(奚正平)2
1. School of Materials and Metallurgy, Northeastern University, Shenyang 110004, China;
2. Northwest Institute for Nonferrous Metal Research, Xi’an 710016, China
Received 6 July 2009; accepted 19 January 2010
Abstract: Boric acid and kieserite were prepared from low-grade ascharite by sulfuric acid method. This method results in the recovery of 71.06% and 45.03% for boric acid and kieserite, respectively. Meanwhile, the boric acid was precipitated from the filtrate at low temperature and the solution was recycled without discharging waste liquid in the whole process. The influence of amount of sulfuric acid, mass fraction of sulfuric acid, reaction temperature and reaction time on the leaching rate of boric acid were studied. The results show that the leaching rate of boric acid reaches 93.80% under the following conditions: the amount of sulfuric acid is 85% of theoretical dosage; the mass fraction of sulfuric acid is 25%; reaction temperature is 95 ℃; and the reaction time is 100 min. Meanwhile, the effects of mass fraction of magnesium sulfate, crystallization temperature and crystallization time on the crystallization of kieserite were investigated and the optimal crystallization conditions are obtained: the mass fraction of magnesium sulfate is 28%; the crystallization temperature is 180 ℃ and the crystallization time is 4 h.
Key words: low-grade ascharite; sulfuric acid method; high temperature crystallization; boric acid; kieserite
1 Introduction
Boric acid is an important industrial resource and it is widely used in many kinds of the manufacturing processes, such as glass[1-2], semiconductor, ceramics, electroplating industry and nuclear industry[3]. Boric acid is also the most commonly used boron compound in the preparation of boron chemicals such as synthetic organic borate salts, borate esters, boron carbide, fluoroborates and boron trihalides[4]. In recent years, boric acid has gained special attention as the catalyst in organic synthesis. Several synthetically useful organic transformations using boric acid as a catalyst have been reported[5-9].
The main method of producing boric acid is sulfuric acid method with high-grade ascharite (w(B2O3)>16%) as the raw material. However, the reserves of high-grade ascharite have been decreased and the low-grade ascharite has not been used to produce boric acid because of the low content of boron. On the other hand, the magnesium content is high in the low-grade ascharite. Thus, it is a waste of magnesium resources if the low-grade ascharite is only used to produce boric acid. Recovery of both boron and magnesium resources is the key problem to utilize low-grade ascharite rationally. Usually, kieserite (MgSO4・H2O) is mined from marine evaporated deposits[10-11]. Kieserite is the important component of cosmetics, detergents, food additives and feed additives[12-13]. It also acts as catalyst in diamond synthesis[14]. It was reported that H3BO3 and MgSO4・H2O were prepared by fractional crystallization process from the liquor of ascharite leached by sulfuric acid[15].
In this study, sulfuric acid was also used to decompose low-grade ascharite. Boric acid was precipitated from the filtrate at low temperature. And high temperature (180 ℃) crystallization method was developed to recover kieserite. The aim of the present study is to investigate the influencing factors (amount of sulfuric acid, concentration of sulfuric acid, reaction temperature and reaction time) on the leaching rate of boric acid (LRBA) from low-grade ascharite and the precipitation conditions of kieserite from the boric acid mother liquor.
2 Experimental
2.1 Materials and reagents
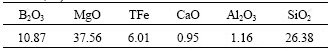
The ascharite sample was taken from the boron ore of Kuandian Garden Valley (Liaoning Province, China) and was ground and dried. All the chemicals were of analytical reagent grade and the deionized water was used throughout. The chemical compositions of ascharite powder are given in Table 1.
Table 1 Main chemical compositions of ascharite (mass fraction, %)
2.2 Apparatus
Boron acid was precipitated in the THD-0515 low temperature water bath and was dried in the DZF-6020 vacuum drying oven. Kieserite was precipitited at high temperature in the FCZ0.1~10 pressure reactor. The X-ray diffraction (XRD) analysis was performed using Cu Ka radiation (l=1.540 8 nm) (X’Pert PRO MPD, PANalytical, Netherlands).
2.3 Procedure
The mineral powder and water were mixed in a certain proportion in a flask. Subsequently, the mixture was heated to 95 ℃, followed by adding concentrated sulfuric acid (95%) to the solution. The acid leaching reaction continued with agitation for 2 h. And then, hydrogen peroxide was added to oxidize Fe2+ to Fe3+. The calcium carbonate was used to adjust the pH value of solution to 6. In the next step, leaching residue was separated from leaching solution and washed twice by hot water. The filtrate was placed for the crystallization of boric acid for 10 h at 10 ℃. Boric acid crystals and the mother liquor containing MgSO4 were separated by solid-liquid separation. Finally, MgSO4・H2O was obtained from the mother liquor by high temperature crystallization. The experiment flow is shown in Fig.1.

Fig.1 Experimental process
2.4 Analysis method
The concentration of boric acid was determined by acid-based titration method. The mannitol was used to strengthen the acidity of boric acid. Phenolphthalein and sodium hydroxide solution were used as the indicator and standard titration solution, respectively. The concentration of Ca2+ was determined by the ethylene- diamine tetraacetic acid (EDTA) titration using calconcarboxylic acid as the indicator in pH 12 [16-17]. The total concentrations of Ca2+ and Mg2+ were determined by (EDTA) titration using eriochrome black T as the indicator at pH 10.
3 Results and discussion
3.1 Factors on leaching rate of boric acid
3.1.1 Effect of amount of sulfuric acid
In the leaching process, if the sulfuric acid is not sufficient, the borate in the ore could not be decomposed completely and the leaching rate of boric acid (LRBA) is small. In addition, the iron and aluminum hydroxides could also precipitate and deteriorate the filtration operation. However, if the sulfuric acid is excess, more impurity will be dissolved into the solution. Moreover, the equipment cannot be tolerant with too much acid. Thus, the proper dosage of sulfuric acid is a key parameter. In Fig.2, the LRBA increases rapidly from 83% to 93.8% when the dosage of sulfuric acid increases from 70% to 85% of theoretical dosage. However, the LRBA increases slowly when the dosage of sulfuric acid increases from 85% to 90% of theoretical dosage. Therefore, 85% is supposed to be a proper dosage. Meanwhile, the leaching solution was easy to filter under this condition.

Fig.2 Effect of amount of sulfuric acid on leaching rate of boric acid
3.1.2 Effect of mass fraction of sulfuric acid
Reaction rate is affected by the mass fraction of sulfuric acid in the solution. The effect of mass fraction of sulfuric acid on the LRBA is shown in Fig.3. The LRBA increases with the increase of the mass fraction of sulfuric acid concentration and it is over 93% when the mass fraction is 25%. The LRBA increases slowly when the mass fraction of sulfuric acid is over 25%. Therefore, 25% is proposed to be a proper mass fraction of sulfuric acid in the leaching process.
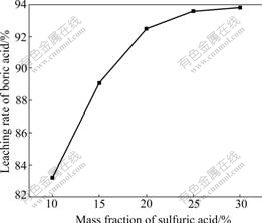
Fig.3 Effect of mass fraction of sulfuric acid on leaching rate of boric acid
3.1.3 Effect of reaction time
The effect of reaction time on LRBA indicates that LRBA increases with the increase of reaction time (Fig.4). Longer reaction time makes higher boric acid yield. When the reaction time is 100 min, the LRBA reaches 93.8%. Further increasing the reaction time gives no remarkable improvement for LRBA. Moreover, longer reaction time could reduce the treatment capacity of equipment and increase energy consumption. Therefore, 100 min is chosen for the subsequent experiments.

Fig.4 Effect of reaction time on leaching rate of boric acid
3.1.4 Effect of leaching temperature
The effect of temperature on LRBA was examined in the temperature range of 65-100 ℃, as shown in Fig.5. Chemical reaction rate accelerates because the diffusion rate of ions in solution speeds up with temperature increasing. A maximum LRBA of 95% was achieved at 100 ℃. Low reaction temperature makes borates decomposed incompletely, which could also make filtration rate slow. So, 95 ℃ was chosen to be optimum for saving energy.
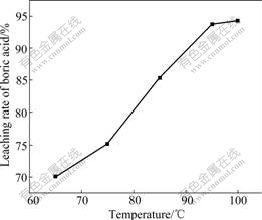
Fig.5 Effect of reaction temperature on leaching rate of boric acid
3.1.5 Optimum reaction conditions for boric acid leaching
When the amount of sulfuric acid is 85% of theoretical amount, concentration of sulfuric acid 20%, leaching time is 100 min and leaching temperature is 95 ℃, the leaching rate of boric acid can reach 93.8%. The filtrate was placed in low-temperature thermostat bath for 10 h at 10 ℃ and the boric acid was crystallized. The recovery rate of boric acid reaches 71.06%.
3.2 Recovery of magnesium sulfate
Boric acid was obtained by lower temperature crystallization from the filtrate. The content of MgSO4 in mother liquor was about 10%-20% (mass fraction). It is a waste of magnesium resources if the mother liquor is discharged without treatment and it will also pollutes the environment. Therefore, MgSO4 needs to be recovered. The solubility of magnesium sulfate decreases with the increase of temperature in the range of 100-200 ℃. MgSO4・H2O will precipitate with the increase of temperature in MgSO4-saturated solution. Since the content of MgSO4 does not reach the saturated value, the recovery rate of MgSO4・H2O (RRM) is low when the mother liquor is directly dealt. In order to get high RRM, mother liquor has to be concentrated. The XRD pattern of product precipitated from the mother liquor (Fig.6) indicates that the product is MgSO4・H2O.

Fig.6 X-ray diffraction pattern of product precipitated from mother liquor
3.2.1 Effect of MgSO4 content
It is known that supersaturation degree is the driving force for crystallization. The crystallization rate of MgSO4・H2O is decided by the supersaturation degree. The limited saturation degree of magnesium sulfate is 8.43% (mass fraction)[18]. This means that MgSO4・H2O could precipitate when the content is over 8.43%. Several solutions of different contents of MgSO4 were obtained by concentrating the mother liquor. The reaction temperature was 180 ℃, and the reaction time was 4 h. The effects of the content of MgSO4 on the yield and RRM are shown in Fig.7. The yield and RRM increase with the increase of MgSO4 content. When the content is 28%, the RRM could reach 45.03%. The saturated content of MgSO4 is 28% at 30 ℃. Thus, the RRM is high while the content of MgSO4 is over 25%.
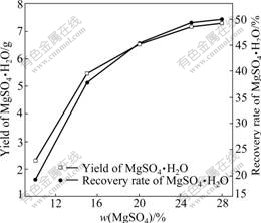
Fig.7 Effect of MgSO4 content on yield and recovery of MgSO4・H2O
3.2.2 Effect of temperature
The solubility of MgSO4 is low at 180 ℃, which benefits the crystallization of MgSO4・H2O. Three concentrated mother liquors with the content of 25% (mass fraction) were heated at different temperatures for 4 h. The effect of temperature on the yield and RRM were examined in the temperature range of 170-190 ℃, as shown in Fig.8. It is found that temperature has a profound effect on RRM. A maximum RRM of 44.76% is achieved at 190 ℃. Since 44.12% MgSO4?H2O is recovered at 180 ℃, this temperature is chosen to be the optimum for saving energy.
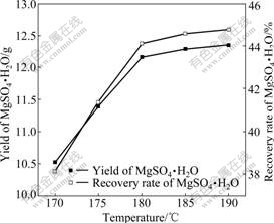
Fig.8 Effect of temperature on yield and recovery of MgSO4・H2O
3.2.3 Effect of crystallization time
Four concentrated mother liquors with content of 25%, were heated to 180 ℃ for different crystallization time. The effect of crystallization time on the yield and RRM is shown in Table 2. From Table 2, it is seen that RRM reaches 42.15% when the crystal time is 4 h. This implies that more crystallization time makes no remarkable variation on RRM. Therefore, 4 h is chosen as the optimal crystallization time.
Table 2 Effect of crystallization time on yield and recovery of MgSO4・H2O

3.2.4 Optimum conditions for recovery of magnesium sulfate
When the content of magnesium sulfate is 28%, crystallization temperature is 180 ℃ and crystallization time is 4 h, magnesium sulfate can be recovered effectively and the RRM reaches 45.03%.
4 Conclusions
1) The optimal reaction conditions of preparation of boric acid from ascharite are as follows: the dosage of sulfuric acid is 85% for theoretical dosage; the reaction time is 100 min; the reaction temperature is 95 ℃; the mass fraction of sulfuric acid is 25%; proper stirring is necessary to make mineral powder suspended. Under these conditions, the LRBA reaches 93.80% and the recovery of boric acid reaches 71.06%.
2) Magnesium sulfate could be recovered effectively from mother liquor under these conditions: the content of magnesium sulfate is over 25% (mass fraction); crystallization temperature is 180 ℃; and crystallization time is 4 h.
3) It is feasible to prepare the boric acid from ascharite by sulfuric acid method and recover magnesium sulfate from the boric acid mother liquor.
References
[1] GARRETT D E. Borates [M]. California: Academic Press, 1998: 401-428.
[2] YE?ILYURT M. Determination of the optimum conditions for the boric acid extraction from colemanite ore in HNO3 solutions [J]. Chemical Engineering and Processing, 2004, 43(10): 1189-1194.
[3] ITAKURA T, SASAI R, ITOH H. Precipitation recovery of boron from wastewater by hydrothermal mineralization [J]. Water Research, 2005, 39: 2543-2548.
[4] TAYLAN, G?RB?Z H, BULUTCU A N. Effects of ultrasound on the reaction step of boric acid production process from colemanite [J]. Ultrasonics Sonochemistry, 2007, 14(5): 633-638.
[5] CHAUDHURI M K, HUSSAIN S, KANTAM M L, NEELIMA B. Boric acid: A novel and safe catalyst for aza-Michael reactions in water [J]. Tetrahedron Letters, 2005, 46(48): 8329-8331.
[6] KONDAIAH G C M, REDDY L A, BABU K S, GURAV V M, HUGE K G, BANDICHHOR R, REDDY P P, BHATTACHARYA A, ANAND V. Boric acid: An efficient and environmentally benign catalyst for transesterification of ethyl acetoacetate [J]. Tetrahedron Letters, 2008, 49(1): 106-109.
[7] SHELKE K F, SAPKAL S B, KAKADE G K, SHINDE P V, SHINGATE B B, SHINGARE M S. Boric acid as an efficient catalyst for the synthesis of 1,1-diacetate under solvent-free condition [J]. Chinese Chemical Letters, 2009, 20: 1453-1456.
[8] KUMAR A, MAURYA R A. An unusual Mannich type reaction of tertiary aromatic amines in aqueous micelles [J]. Tetrahedron Letters, 2008, 49(38): 5471-5474.
[9] SHELKE K E, SAPKAL S B, SONAR S S, MADJE B R, SHINGATE B B, SHINGARE M S. An efficient synthesis of 2,4,5-Triaryl-1H-imidazole derivatives catalyzed by boric acid in aqueous media under ultrasound-irradiation [J]. Bulletin of the Korean Chemical Society, 2009, 30(5): 1057-1060.
[10] GREVEL K D, MAJZLAN J. Internally consistent thermodynamic data for magnesium sulfate hydrates [J]. Geochimica et Cosmochimica Acta, 2009, 73(13): 6805-6815.
[11] KILIC ?, KILIC A M. Recovery of salt co-products during the salt production from brine [J]. Desalination, 2005, 186(1/3): 11-19.
[12] HE Chun-bao, WANG Xiu-ping. Production application and prospect of magnesium sulfate monohydrate [J]. Inorganic Chemicals Industry, 2008, 40(5): 13-15. (in Chinese)
[13] HE Chun-bao. Study on new technology for preparation of purified magnesium sulfate monohydrate from crude magnesium sulfate [J]. Inorganic Chemicals Industry, 2004, 36(5): 31-33. (in Chinese)
[14] WANG Yu-chang, AKAISHI M, YAMAOKA S. Diamond formation from graphite in the presence of anhydrous and hydrous magnesium sulfate at high pressures and high temperatures [J]. Diamond and Related Materials, 1999, 8(1): 73-77.
[15] L? Bing-ling. Fractional crystallization of boric acid and kieserite from the liquor of ascharite leached by sulfuric acid [J]. Inorganic Chemicals Industry, 2006, 38(10): 18-21. (in Chinese)
[16] SPIVACK A J, EDMOND J M. Determination of boron isotope ratios by thermalionization mass spectrometry of the dicesium metaborate cation [J]. Analytical Chemistry, 1986, 58: 31-35.
[17] DAVIDSON G R, BASSETT R L. Application of boron isotopes for identifying contaminants such as fly ash leachate in groundwater [J]. Environmental Science Technology, 1993, 8(27): 172-176.
[18] ZHANG Li-qing, ZHU Jian-xin, LIU Su-lan, PANG Chang-jian. Study of high temperature crystallization kinetics of MgSO4?2H2O system [J]. Journal of Shenyang Institute of Chemical Technology, 1999, 13(2): 147-150. (in Chinese)
Foundation item: Project(2006AA06Z368) supported by the National High-tech Research and Development Program of China
Corresponding author: LI Jie; Tel: +86-24-83687727; E-mail: ljws963@yahoo.com.cn
DOI: 10.1016/S1003-6326(09)60272-7
(Edited by YANG Bing)