
High-rate capability of spinel LiNi0.05Mn1.95O4 cathode for Li-ion batteries prepared via coprecipitated precursor
SUN Bin(孙 斌)1, 2, SHEN Guo-pei(申国培)2, HU Yan-long(胡燕龙)2
1. College of Materials Science and Engineering, South China University of Technology, Guangzhou 510640, China;
2. Guangzhou Hongsen Material Co. Ltd., Guangzhou 510760, China
Received 15 July 2007; accepted 10 September 2007
Abstract: Spinel LiNi0.05Mn1.95O4 cathode material for lithium ion batteries was synthesized by solid-state reaction from coprecipitated Ni-Mn hydroxide precursors and characterized by X-ray diffraction(XRD), scanning electron microscopy(SEM) and galvanostatic charge-discharge tests. It is found that LiNi0.05Mn1.95O4 powder has an ordered cubic spinel phase (space group 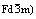 and exhibits superior rate capability. After 450 cycles, the LiNi0.05Mn1.95O4/carbonaceous mesophase spheres(CMS) Li-ion batteries can retain 96.0% and 93.3% capacity at 5C and 10C charge/discharge rate, respectively, compared with 85.3% (5C) and 80.5% (10C) retention for LiMn2O4 batteries. However, the initial discharge capacity of LiNi0.05Mn1.95O4/CMS batteries at 1C charge/discharge rate (96.20 mA?h/g) is slightly lower than that of the LiMn2O4 batteries (100.98 mA?h/g) due to the increased average oxidation state of Mn in LiNi0.05Mn1.95O4.
and exhibits superior rate capability. After 450 cycles, the LiNi0.05Mn1.95O4/carbonaceous mesophase spheres(CMS) Li-ion batteries can retain 96.0% and 93.3% capacity at 5C and 10C charge/discharge rate, respectively, compared with 85.3% (5C) and 80.5% (10C) retention for LiMn2O4 batteries. However, the initial discharge capacity of LiNi0.05Mn1.95O4/CMS batteries at 1C charge/discharge rate (96.20 mA?h/g) is slightly lower than that of the LiMn2O4 batteries (100.98 mA?h/g) due to the increased average oxidation state of Mn in LiNi0.05Mn1.95O4.
Key words: Li-ion battery; rate capability; Ni-doped Mn spinel; coprecipitation
1 Introduction
Lithium-ion batteries have been successfully applied to portable electronic devices such as cellular phones and laptop computers. Besides, they have become possible high power sources for electric tools and HEVs. However, the high cost and toxicity associated with the currently used LiCoO2 cathodes suppress their application to electric tools and HEVs. It is necessary to develop inexpensive and environmentally benign cathode materials to satisfy the requirement for the high power lithium-ion batteries. In this regard, spinel LiMn2O4 has become an attractive cathode due to its low cost, non-toxicity and its high voltage[1-2], but LiMn2O4 also has some disadvantages to high-rate batteries. For example, LiMn2O4 shows severe capacity fade particularly at elevated temperatures that is ascribed to Jahn-Teller distortion[3-4], manganese dissolution into electrolyte[5], and loss of crystallinity during cycling[6]. Additionally, the rate capability of LiMn2O4 is expected to be low because the solid-phase diffusivity of Li+ in LiMn2O4 is poorer than that of LiCoO2[7]. Substitution of other cations for manganese has been found to improve the cyclability at elevated temperatures[8-9] and rate capability of LiMn2O4 spinels[10-11].
As a precursor synthesis method, coprecipitation can mix metal ions homogeneously, resulting in an easier control of the stoichiometric synthesis of the materials at an atomic level[12-14]. In this paper, the Ni-Mn hydroxide with a given ratio was synthesized by coprecipitation, and then the as-obtained precursor was calcined with Li2CO3 at high temperature to form a LiNi0.05Mn1.95O4 spinel. The crystal structure, morphology and electrochemical performance of this cathode were investigated. It is found that the LiNi0.05Mn1.95O4 spinel shows excellent rate capability even at a charge/ discharge rate as high as 10C.
2 Experimental
LiNi0.05Mn1.95O4 spinel was prepared as follows. Firstly, stoichiometric ratios of MnSO4・H2O and NiSO4・6H2O were completely dissolved into distilled water and then slowly dropped into the NaOH solution with a pH value of 12 that was being stirred rapidly at ambient temperature. The precipitate was filtered and washed with distilled water and then dried at 110 ℃ in air overnight. A 5% excessive Li2CO3 (compensation for possible Li loss during high temperature calcination) was mixed with the as-prepared precursor and preheated at 450 ℃ for 5 h, then calcined at 800 ℃ for 24 h and next annealed at 600 ℃ for 5 h in air. For comparison, spinel LiMn2O4 was prepared following the same procedures as LiNi0.05Mn1.95O4. The crystal structure of as-prepared powders was analyzed by XRD using a D/Max-2400 (Rigaku, Japan) diffractometer equipped with Cu Kα radiation. The particle morphology was examined by SEM (LEO 1530VP, Germany).
The charge-discharge experiments were conducted on a lithium-ion cell consisting of a spinel cathode, a CMS (Shanshan Tech Group, China) anode and an electrolyte of 1 mol/L LiPF6 in a 1?1 (in volume) mixture of ethylene carbonate(EC)/dimethyl carbonate(DMC). The cathode was prepared by mixing 90% (mass fraction) spinel, 6% carbon black, and 4% polyvinylidene fluoride(PVDF) binder in a solvent of N- methyl-2-pyrrolidone(NMP) to form slurry. The slurry was coated onto aluminum foil and dried at 120 ℃. After dried, electrodes were cut into appropriate sizes. Then the electrode was pressed to a thickness of 160 ?m. The CMS electrode, composed of 92% CMS with 8% PVDF binder and NMP solvent, was made following the same processing steps as the cathode electrode, except that it was coated onto copper foil. Resultant thickness of the CMS electrode was 120 ?m. After dried, electrodes were cut into appropriate sizes. Next the cathode and anode were wound with separator (Celgard 2300, USA). The roll of electrodes and separator was inserted into a stainless steel case and then dried at 80 ℃ for 12 h in a vacuum chamber before the case was sealed except the injecting hole using laser welder. The sealed cases were moved into a glove box (temperature 25 ℃, relative humidity 0.3%). Electrolyte was injected and then the injecting hole was sealed. The fresh battery had external dimensions of 5.8 mm×30 mm×40 mm.
3 Results and discussion
3.1 Phase structure of cathode
Fig.1 shows the XRD patterns of the LiMn2O4 and LiNi0.05Mn1.95O4 powder. Both samples were confirmed to have an ordered cubic spinel structure with space group 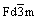 and no secondary phase or impurity phases can be observed, implying that in the LiNi0.05Mn1.95O4 sample, the Mn site is substituted fully by Ni doping. The diffraction lines shifted slightly towards higher angles with Ni substitution, indicating a smaller lattice parameter for the Ni-doped sample. The lattice parameters are 0.825 0 and 0.824 3 nm for LiMn2O4 and LiNi0.05Mn1.95O4, respectively. The decreased lattice
and no secondary phase or impurity phases can be observed, implying that in the LiNi0.05Mn1.95O4 sample, the Mn site is substituted fully by Ni doping. The diffraction lines shifted slightly towards higher angles with Ni substitution, indicating a smaller lattice parameter for the Ni-doped sample. The lattice parameters are 0.825 0 and 0.824 3 nm for LiMn2O4 and LiNi0.05Mn1.95O4, respectively. The decreased lattice
Fig.1 XRD patterns of LiMn2O4 (a) and LiNi0.05Mn1.95O4 (b)
parameter of LiNi0.05Mn1.95O4 is consistent with the increased concentration of Mn4+ in the spinel structure, which has a much smaller ionic radius (r=0.053 nm) than Mn3+ (r=0.064 5 nm)[15]. It is well known that the Jahn-Teller distortion of the [MnO6] octahedron in LiMn2O4 is caused by the Mn3+ cations at the 16d sites and the decreased amount of Mn3+ in LiMn2O4 can suppress the Jahn-Teller distortion[16-17]. Another reason for the decreased lattice parameter of LiNi0.05Mn1.95O4 was proposed to be that Mn(Ni)―O bond (1 029 kJ/mol) in LiNi0.05Mn1.95O4 is stronger than the Mn―O bond (946 kJ/mol) in LiMn2O4, which could enhance the structural stability of the [Mn(Ni)O6] octahedron[18]. Therefore, it was expected that LiNi0.05Mn1.95O4 would exhibit better structural stability and cyclic performance than the non-substituted LiMn2O4.
3.2 Surface morphology analysis
Fig.2 shows the surface morphology LiMn2O4 and LiNi0.05Mn1.95O4. Figs.2(a) and (b) indicate that both samples are composed of small primary faceted crystallites in octahedral shape with clean surfaces. The primary particles sizes are about 1 mm. No change in morphology is observed between the Ni-doped and non-doped Mn-spinels.
3.3 Electrochemical performance
In order to investigate the rate capability of the cathode, we charged both the LiMn2O4/CMS and LiNi0.05Mn1.95O4/CMS lithium-ion batteries using constant-current at 1C rate followed by constant-voltage at 4.2 V, then discharged the batteries at 1C, 5C and 10C rate, respectively. The discharge plots at various rates are shown in Fig.3. While LiMn2O4 exhibits huge polarization losses with increasing rate, the LiNi0.05- Mn1.95O4 exhibits remarkable rate capability, especially

Fig.2 SEM images of LiMn2O4 (a) and LiNi0.05Mn1.95O4 (b)
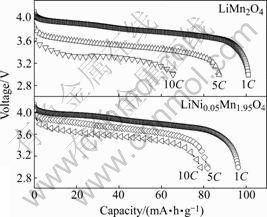
Fig.3 Discharge profiles of LiMn2O4/CMS and LiNi0.05Mn1.95O4/CMS lithium-ion batteries at various discharging rates (1C, 5C and 10C) between 3.0 and 4.3 V (charge rate is fixed at 1C)
at higher rate as 10C. But the initial discharge capacity at 1C rate of LiNi0.05Mn1.95O4 (96.20 mA?h/g) is slightly lower than that of LiMn2O4 (100.98 mA?h/g) because of the less content of Mn3+ in LiNi0.05Mn1.95O4 as a result of Ni substitution for Mn[19].
The cycling performance of two spinels at high charge/discharge rates was also measured as shown in Fig.4. The batteries were charged using constant-current at 5C or 10C rate followed by constant-voltage at 4.2 V, then discharged at the same rate as its charging rate.
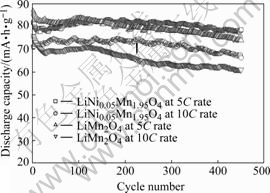
Fig.4 Discharge capacity vs cycle number plots of LiNi0.05Mn1.95O4/CMS lithium-ion batteries between 3.0 and 4.3 V at 5C and 10C charge/discharge rate, respectively
After 450 cycles, LiNi0.05Mn1.95O4/CMS lithium-ion batteries can still retain 96.0% and 93.3% capacity at 5C and 10C, respectively. In contrast, LiMn2O4/CMS batteries just remain 85.3% (5C) and 80.5% (10C). Since both spinel samples show quite similar morphology in term of particle shape and particle size, the difference in the rate capability between LiMn2O4 and LiNi0.05Mn1.95O4 does not result from particle size. According to the structural analysis results (section 3.1), we attributed the excellent cycling stability of LiNi0.05Mn1.95O4 at high rate to the increase in stability of spinel structure resulting from the suppression of the Jahn-Teller distortion and stronger Mn(Ni)―O bond as Mn3+ are substituted by Ni2+.
In addition to the superior rate capability, the spinel LiNi0.05Mn1.95O4 offers the advantages of low cost, low toxicity, and easy synthesis. Therefore, we expect that LiNi0.05Mn1.95O4 cathode would be attractive for high power applications such as electric tools and HEVs despite slightly lower initial capacity.
4 Conclusions
1) The spinel LiNi0.05Mn1.95O4 synthesized from coprecipitated Ni-Mn hydroxide precursors exhibits superior rate capability at 5C and 10C charge/discharge rate with a small sacrifice in the initial discharge capacity.
2) The excellent rate capability of LiNi0.05Mn1.95O4 cathode at high rate is attributed to the increase in structural stability resulting from the depression of the Jahn-Teller distortion caused by decreased content of Mn3+ and stronger Mn(Ni)―O bond as Mn3+ are substituted by Ni2+.
3) The LiNi0.05Mn1.95O4 cathode would be attractive for high power applications such as electric tools and HEVs because of its advantages of low cost, low toxicity, easy synthesis and superior rate capability.
References
[1] TARASCON J M, COOWAR F, AMATUCI G, SHOKOOHI F K, GUYOMARD D G. The Li(1+x)Mn2O4/C system materials and electrochemical aspects [J]. J Power Sources, 1995, 54(1): 103-108.
[2] THACKERAY M M, MANSUETTO M F, BATES J B. Structural stability of LiMn2O4 electrodes for lithium batteries [J]. J Power Sources, 1997, 68(1): 153-158.
[3] MORALES J, SANCHEZ L, TIRADO J. New doped Li-M-Mn-O(M=Al, Fe, Ni) spinels as cathodes for rechargeable 3V lithium batteries [J]. J Solid Electrochemica Acta, 1999, 45: 255-271.
[4] GUMMOW R J, KOCK A D, THACKERAY M M. Improved capacity retention in rechargeable 4V lithium/lithium-manganese oxide (spinel) cells [J]. Solid State Ionics,1994,69(1): 59-67.
[5] WANG Li-fang, OU Chin-ching, STRIEBEL K A. Study of Mn dissolution from LiMn2O4 spinel electrodes using rotating ring-disk collection experiments [J]. J Electrochem Soc, 2003, 150(7): A905-A911.
[6] HUANG Hai-tao, VINCENT C A, BRUCE P G. Correlating capacity loss of stoichiometric and nonstoichiometric lithium manganesse oxide spinel electrodes with their structural integrity [J]. J Electrochem Soc, 1999, 146(10): 3649-3654.
[7] DEISS E. Spurious chemical diffusion coefficients of Li+ in electrode materials evaluated with GITT [J]. Electrochimica Acta, 2005, 50(14): 2927-2932.
[8] PAOLONE A, CANTELLI R, SCROSATI B, REALE P, FERRETTI M, MASQUELIER C. Doping effects on the phase transition of LiMn2O4 by anelastic spectroscopy and differential scanning calorimetry [J]. Mater Sci Eng A, 2006, 442(1): 220-223.
[9] EFTEKHARI A, MOGHADDAM A B, MEHRAN S H. Electrochemical properties of LiMn2O4 cathode material doped with an actinide [J]. J Alloys and Compounds, 2006, 424(1): 225-230.
[10] DENG Bo-hua, NAKAMURA H, YOSHION M. Comparison and improvement of the high rate performance of different types of LiMn2O4 spinels [J]. J Power Sources, 2005, 141(1): 116-121.
[11] BAO Shu-juan, LIANG Yan-yu, ZHOU Wen-jia, HE Ben-lin, LI Hu-lin. Synthesis and electrochemical properties of LiAl0.1Mn1.9O4 by microwave-assisted sol-gel method [J]. J Power Sources, 2006, 154(1): 239-245.
[12] CHO T H, PARK S M, YOSHIO M, HIRAI T, HIDESHIMA Y. Effect of synthesis condition on the structural and electrochemical properties of Li[Ni1/3Mn1/3Co1/3]O2 prepared by carbonate co-precipitation method [J]. J Power Sources, 2005, 142(1): 306-312.
[13] CHO T H, PARK S M, YOSHIO M. Preparation of layered Li[Ni1/3Mn1/3Co1/3]O2 as a cathode for lithium secondary battery by carbonate coprecipitation method [J]. Chem Lett, 2004, 33(6): 704-705.
[14] BELHAROUAK I, LU W Q, VISSERS D, AMINE K. Safety characteristics of Li(Ni0.8Co0.15Al0.05)O2 and Li(Ni1/3Mn1/3Co1/3)O2 [J]. Electrochem Commun, 2006, 8(2): 329-335.
[15] WEI Y J, NAM K W, KIM K B, CHEN G. Spectroscopic studies of the structural properties of Ni substituted spinel LiMn2O4 [J]. Solid State Ionics, 2006, 177(1): 29-35.
[16] LEE J F, TSAI Y W, SANTHANAM R, HWANG B J, YANG M H, LIU D G. Local structure transformation of nano-sized Al-doped LiMn2O4 sintered at different temperatures [J]. Journal of Power Sources, 2003, 119/121(6): 721-726.
[17] HE Xiang-ming, LI Jian-jun, CAI Yan, WANG Yao-wu, YING Jie-rong, JIANG Chang-yin, WAN Chun-rong. Preparation of co-doped spherical spinel LiMn2O4 cathode materials for Li-ion batteries [J]. Journal of Power Sources, 2005, 150(10): 216-222.
[18] ITO Y, IDEMOTO Y, TSUNODA Y, KOURA N. Relation between crystal structures, electronic structures, and electrode performances of LiMn2-xMxO4 (M=Ni, Zn) as a cathode active material for 4V secondary Li batteries [J]. J Power Sources, 2003, 119/121(6): 733-737.
[19] PARK D H, LIM S T, HWANG S J, ChOY J H, CHOI J H, CHOO J. Influence of nickel content on the chemical bonding character of LiMn2-xNixO4 spinel oxides [J]. J Power Sources, 2006, 159(2): 1346-1352.
(Edited by PENG Chao-qun)
Corresponding author: SUN Bin; Tel: +86-20-82210888; E-mail: tiansansnow@tom.com