Trans. Nonferrous Met. Soc. China 24(2014) 2986-2990
Leaching behaviors of iron and aluminum elements of ion-absorbed-rare-earth ore with a new impurity depressant
Ting-sheng QIU1, Xi-hui FANG1, Hong-qiang WU2, Qing-hua ZENG1,3, Dong-mei ZHU1
1. Faculty of Resource and Environmental Engineering, Jiangxi University of Science and Technology, Ganzhou 341000, China;
2. Ma’anshan Institute of Mining Research, Sino-Steel Group, Ma’anshan 243000, China;
3. School of Computing, Engineering and Mathematics, University of Western Sydney, Penrith, NSW 2751, Australia
Received 8 July 2013; accepted 3 December 2013
Abstract: Ion-absorbed rare-earth ore is an important mineral resource which is widely extracted by in-situ leaching process. And such process generates a significant amount of impurities such as aluminum and iron ions in leaching solution simultaneously. The surface characteristics and interactions by infrared spectroscopy and X-ray diffraction were studied to optimize the leaching conditions. It is found that the environment-friendly depressant LG-01 can react with the impurity ions through the formation of a new complex on the surface of leaching residues. Thus, it reduces significantly the concentration of impurity ions in leaching solution and improves the leaching rate of rare-earth ore. Moreover, a leaching rate of 95.6% and an impurity removal rate of 92% have been achieved under the optimized conditions.
Key words: ion-absorbed-type rare-earth ore; leaching; depression mechanism; aluminum and iron impurity
1 Introduction
Ion-absorbed-type rare-earth ore is a unique mineral resource which was first discovered in Jiangxi Province, China. With further exploration, such rare-earth ore has also been found in other provinces mainly located in Southeast of China, including Fujian, Guangdong, Hunan, Guangxi and Zhejiang. Most of such rare-earth ore deposits are associated with granite, mixed rocks, and other igneous rocks [1-3]. Ion-absorbed-type rare-earth ore is formed through a series of geological processes. Initially, granite and igneous rocks containing rare-earth minerals are subjected to biological and chemical reactions under humid conditions and changed into clay minerals. Meanwhile, rare-earth minerals (e.g., fluoro- carbonate, gadolinite) associated with the rocks are weathered and form rare-earth hydroxyl aqueous ions. Then, such rare-earth ions are absorbed onto clay minerals during the infiltration process, and form ion-absorbed-type rare-earth ores [4-6].
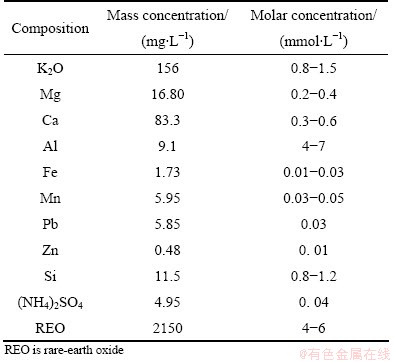
At present, electrolyte leaching is widely used to leach ion-absorbed-type rare-earth ore, which leads to the leaching of impurity ions (e.g., aluminum and iron) into leaching solution [7-9]. Such impurity ions can influence the quality of rare-earth precipitation and increase the cost toward impurity elimination. More importantly, the crystal form of carbonated rare-earth may not be able to form due to the presence of such impurities [10,11]. The main impurities in leaching solution of ion-absorbed-type rare-earth ore are related to the nature of rare-earth ore, leaching process, operating conditions, and kinds of leaching agents. The main chemical composition in leaching solution is shown in Table 1 [12], which indicates that the main impurities are aluminum and iron ions. Thus, such impurities must be removed before the precipitation of leaching solution, which would further increase the production cost. In the past, some studies have been done to inhibit the impurities and leach ion-absorbed-type rare-earth ore [13-15]. Yet, the depressants used have adverse impacts on environment and human being. Moreover, a further understanding and exploration should be done about the mechanisms of the reaction between the impurities (e.g., aluminum and ferric ions) and impurity depressant.
In this study, infrared (IR) spectrometry and X-ray diffraction (XRD) are used to analyze the behaviors of exchange between rare-earth ion and leaching agent as well as the exchange between impurity ions (i.e., aluminum, iron) and leaching agent during the leaching process of ion-absorbed-type rare-earth ore. Then, leaching experiments are conducted using an impurity depressant called LG-01 and the effects of LG-01 on aluminum and iron ions are examined.
Table 1 Main chemical composition in leaching solution of typical ion-absorbed-type rare-earth ore [12]
2 Experimental
2.1 Ore samples and chemicals
Rare-earth ore samples were obtained from a rare-earth mine in Xunwu of Jiangxi Province, China. The samples contained 0.22% ion-phase rare-earth, 71% SiO2, 12% Al2O3, and 2% Fe2O3. In our experiments, tap water was used in leaching experiments and deionized water was used in chemical analysis. The agents in experiment such as (NH4)2SO4, environment-friendly depressant LG-01 (the agents were organic agents containing hydroxyl and carboxyl groups), NH4F, NH4・H2O and Na2S were analytically pure.
2.2 Methods
Leaching experiments were conducted in a leaching system which consists of a d10 cm leaching column, volumetric flasks, beakers, and dosing device. In the process, depressant and leaching agents were dissolved into the water together and then the leaching experiments were carried out. Leach liquor was collected by specialized device. Leaching residue should be dried naturally and used in follow-up study after the end of leaching experiment. IR and XRD techniques were used to analyze the surface properties of ion-absorbed-type rare-earth ore and the interaction mechanisms between leaching agents and minerals as well as depressant and impurity ions.
3 Results and discussion
3.1 IR spectrum analysis
IR analysis has been done on the rare-earth ore and leaching residues in the absence and presence of impurity depressant. Prior to IR test, the samples were dried naturally and placed in a mortar to crush them. Figure 1(a) shows the IR spectrum of rare-earth metal ore in which the characteristic peaks include 3000-4000 cm-1 from hydroxyl single bond or stretching vibration in water molecules, 1600-1660 cm-1 from bending vibration of water molecules, 960-1140 cm-1 from stretching vibration of phosphate polyhedron, and 830-1000 cm-1 from stretching vibration of silicon-oxygen single bond. The rare-earth leaching residue in the absence of depressant (Fig. 1(b)) has similar IR spectrum as that of rare-earth ore, which further confirms that there is no new substance formed and no significant change on the mineral surface.
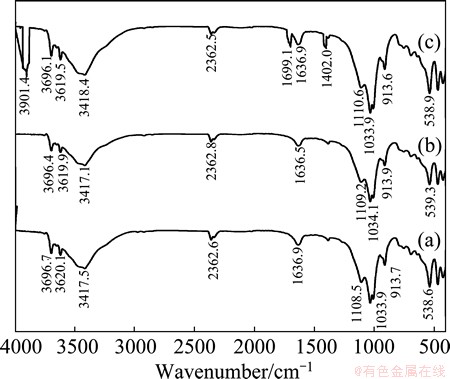
Fig. 1 IR spectra of rare-earth ore (a), rare-earth ore after leaching in the absence of impurity depressant (b), rare-earth ore after leaching in the presence of impurity depressant (c)
However, the IR spectrum of leaching residue obtained in the presence of depressant (Fig. 1(c)) has some new characteristic absorption peaks, including 3901.4 cm-1 from stretching vibration of hydrogen- oxygen single bond, 1699.1 cm-1 form stretching vibration of carbon-oxygen double bonds, and 1402.0 cm-1 from bending vibration of carbon-hydrogen- oxygen single bond. Depressants contain hydrogen- oxygen single bond, the carbon-oxygen double bonds and carbon-hydrogen-oxygen single bond groups. The results indicate that the presence of depressant may lead to certain surface reaction and the formation of new organic complex on the surface of leaching residues [16].
3.2 XRD analysis
XRD has been used to examine the mineral composition of rare-earth ore and leaching residues obtained in the absence and presence of impurity depressant. Figure 2(a) indicates that there are quartz and minerals with aluminum and/or iron in rare-earth ore. In the absence of impurity depressant, the leaching residue (Fig. 2(b)) mainly contains quartz, without obvious evidence of minerals with aluminum and iron. This indicates that the surface aluminum- and iron-containing minerals have been dissolved during leaching process and both aluminum and iron have been released into the leaching solution.
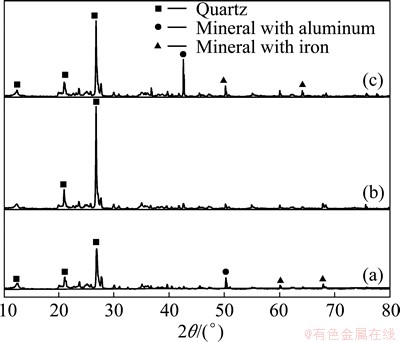
Fig. 2 XRD patterns of rare-earth metal ore (a), rare-earth leaching residue in the absence of depressant (b), and rare-earth leaching residue in the presence of impurity depressant (c)
In the presence of impurity depressant, in addition to the main mineral quartz, leaching residue also contains some aluminum- and iron-bearing compounds (Fig. 2(c)), which may be generated due to the reaction between impurity depressant and dissolved aluminum and iron ions. The formation of such new compounds would reduce the concentration of aluminum and iron ions in leaching solution.
3.3 Leaching of ion-absorbed-type rare-earth ore in the presence of impurity depressant
Based on the above analyses and findings, leaching experiments in the presence of impurity depressant were conducted to optimize the leaching conditions for such ion absorbed-type rare earth ore. In particular, optimization has been made to the depressant dosage, liquid-to-solid ratio, and leaching agent.
3. 4 Effect of impurity depressant dosage
The optimization of depressant dosage has been done under the following conditions: rare-earth ore of 200 g, trickle leaching, (NH4)2SO4 as leaching agent with a content of 5%, a flow rate of 2 mL/min, and a liquid-to-solid ratio of 1.2:1. Figure 3 shows that when the impurity depressant dosage is 0.2% (about 0.013 mol/L), the removal rates of aluminum ion and iron ion are the highest, yet rare-earth extraction rate is reduced slightly. Thus, a content of 0.2% impurity depressant is used for the following experiments. Analysis shows that leaching liquid is mainly composed of RE3+, H+, NH4+, Fe3+, Al3+, Mg2+, Ca2+ and OH-, and leaching solution is made up of these ions under other conditions.
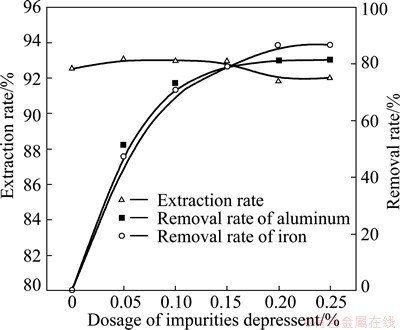
Fig. 3 Effect of impurity depressant dosage on rare-earth extraction rate and removal rate of impurity ions
3.5 Effect of liquid-to-solid ratio
Liquid-to-solid ratio can affect rare-earth extraction rate and the concentration of impurities in leaching solution. Thus, the best rare-earth extraction rate is determined based on the following conditions: raw rare-earth ore of 200 g, trickle leaching, (NH4)2SO4 as leaching agent with a content of 5%, a flow rate of 2 mL/min, and a content of 0.1% impurity depressant.
The result (Fig. 4) indicates that when the liquid-to- solid ratio is 1.2:1, rare-earth extraction rate is higher than that under other conditions, while the removal rate of impurities drops a lot. Thus, the liquid-to-solid ratio of 1:1 is selected.
3.6 Effect of leaching agent concentration
Concentration of leaching agent is an important factor in rare-earth leaching process. A low concentration would not be enough to exchange rare-earth metals completely, while a high concentration may lead to re-adsorption. The experiments on leaching agent concentration were conducted under the following conditions: rare-earth ore of 200 g, trickle leaching, (NH4)2SO4 as leaching agent with a flow rate of 2 mL/min, and a content of 0.1% impurity depressant.
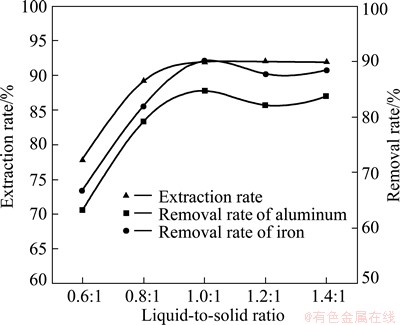
Fig. 4 Effect of liquid-to-solid ratio on rare-earth extraction rate and removal rate of impurity ions
Our results (Fig. 5) indicate that when the content of leaching agent increases, rare-earth extraction rate increases to a certain value. Further increase in leaching agent would lead to the drop of extraction rate and the removal rate of iron impurity. Under a content of 4% leaching agent, rare-earth extraction rate achieves the largest and the removal rates of impurities are high as well. Thus, leaching agent content of 4% is selected.
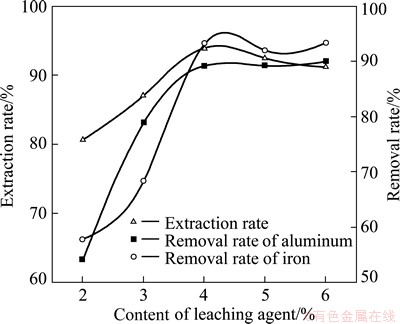
Fig. 5 Effect of leaching agent concentration on rare-earth extraction rate and removal rate of impurity ions
3.7 Effect of flow rate of leaching agent
The flow rate of leaching agent is another important factor that would affect the leaching process, extraction rate, and removal rate of impurity ions. The experiments were done under the following conditions: rare-earth ore of 200 g, trickle leaching, (NH4)2SO4 as leaching agent with a content of 5%, liquid-to-solid ratio of 1.2:1, and a content of 0.1% impurity depressant. Our results (Fig. 6) show that rare-earth extraction rate reaches the highest when the flow rate of leaching agent is 2.4 mL/min. Moreover, the removal rate of impurities is also the highest. Thus, a flow rate of 2.4 mL/min is selected.
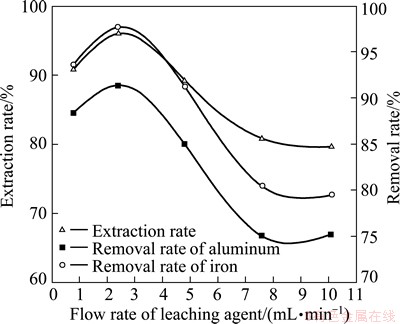
Fig. 6 Effect of agent flow rate on rare-earth extraction rate
4 Conclusions
1) To improve the leaching ratio of ion-absorbed- type rare-earth ore, the surface characteristics of such rare-earth ore and leaching residues in the absence and presence of impurity depressant were studied by IR and XRD. In addition, the exchange process and interactions between agent and ore surface as well as agent and impurity ions were examined. It is found that impurity depressant can react with aluminum and iron ions in leaching solution and form new substance on ore surface, which reduces the impurities in leaching solution but does not affect the leaching process of rare-earth ore. The present study would facilitate the design and selection of effective impurity depressants.
2) A depressant called LG01 was selected as an effective depressant for aluminum and iron impurities. Moreover, the leaching conditions were optimized through a series of experiments, which indicated the depressant content of 0.1%, leaching liquid-to-solid ratio of 1:1, leaching content of 4%, and leaching agent flow rate of 2.4 mL/min. Under such conditions, the extraction rate of ion-absorbed-type rare-earth ore reaches 95.6% and the removal rate of impurities is 92%. The present work would provide a valuable guidance toward the removal of impurities in leaching solution and the optimization of in-situ leaching process of ion-absorbed- type rare-earth ores.
References
[1] CHI Ru-an, TIAN Jun. Chemical metallurgy of weathered crust elution-deposited rare earth ore [M]. Beijing: Science Press, 2006: 172-175. (in Chinese)
[2] HUANG Li-huang. Rare earth extraction technology [M]. Beijing: Metallurgical Industry Press, 2006: 68-98. (in Chinese)
[3] YAO Hui-qin, OUYANG Ke-xian, RAO Guo-hua. A study on leaching rare earth from the weathered elution-deposited rare earth ore with compound leaching reagent [J]. Jiangxi Science, 2005, 23(6): 721-723. (in Chinese)
[4] TIAN Jun, YIN Jun-qun, OUYANG Ke-xian. Rare earth chlorinated solvent extraction from weathering drench multiplicative Gaozhu effect of rare earth mineral leaching solution [J]. Metallurgy of China, 1998(2): 45-49. (in Chinese)
[5] HE Lun-yan, WANG Si-nan. The rare earth mineral ion adsorption type in the south [J]. Rare Earth, l989, 10(1): 39-44. (in Chinese)
[6] CHI Ru-an, WANG Dian-zhuo. Rare earth extraction technology [M]. Beijing: Science Press, 1995: 221-274. (in Chinese)
[7] LI Xiu-fen, CHI Ru-an. Study on removing impurities in the exchanged liquor of rare earth ores [J]. Multipurpose Utilization of Mineral Resources, 1997(2): 10-13. (in Chinese)
[8] TIAN Jun, YIN Jing-qun, OUYANG Ke-xian, CHI Ru-an. Development progress and research connotation of green chemistry of extraction process of rare earth from weathering crust elution- deposited rare earth ores in China [J]. Chinese Rare Earths, 2006. 27(1): 70-73. (in Chinese)
[9] XU Guang-xian. Rare earth [M]. Beijing: Metallurgical Industry Press, 1995: 133-214. (in Chinese)
[10] TIAN Jun, YIN Jing-qun. Resarch on heavy rare earth ore leaching liquid oxalic acid precipitation [J]. Hydrometallurgy of China, 1996(2): 16-19. (in Chinese)
[11] TIAN Jun, LU Sheng-liang, YIN Jing-qun. Kinetic study on leaching a south China rare earth ore [J]. Engineering Chemistry and Metallurgy,1995, 16(3): 354-357. (in Chinese)
[12] CHI Ru-an, WANG Dian-zuo. Beneficiation of rare earth ore and extraction technology [M]. Beijing: Science Press, 2006: 185-187. (in Chinese)
[13] OUYANG Ke-xian, RAO Guo-hua, YAO Hui-qin, MAO Yan-hong. Study of southern RE ore leaching by aluminum inhibition [J]. Rare Metals and Cemented Carbides, 2003, 31(4): 1-3. (in Chinese)
[14] LI Si-jia, YU Qing-hua, CHEN Yi-bo, ZHU Hui-ying, LEI Hao. Impurities inhibited leaching of southern rare earth mineral [J]. Chinese Rare Earths, 1996, 17(2): 29-34. (in Chinese)
[15] MAO Yan-hong, ZHANG Chang-gen, WU Nan-ping, ZENG Ling-jian. Study on the elimination of aluminum in ion adsorption type rare earth [J]. Shanghai Metals: Non-Ferrous Volume, 1993, 14(2): 16-19. (in Chinese)
[16] LIAO Li-bing, WANG Li-juan, YIN Jing-wu. The modern testing technology of mineral material [M]. Beijing: Chemical Industrial Press, 2010: 125-180. (in Chinese).
离子型稀土矿中铝铁杂质的浸出与抑制规律
邱廷省1,方夕辉1,伍红强2,曾清华1,3,朱冬梅1
1. 江西理工大学 资源与环境工程学院,赣州 341000;
2.中钢集团 马鞍山矿山研究院,马鞍山 243000;
3. School of Computing, Engineering and Mathematics, University of Western Sydney, Penrith, NSW 2751, Australia
摘 要:通过红外光光谱测定、XRD检测等测试方法分析了稀土矿浸出过程中各种矿物表面性质的变化,稀土离子及铝、铁杂质离子与浸出剂和抑制剂的浸出交换过程及规律。结果表明,抑制剂的添加会与稀土矿中的铝、铁等杂质离子反应,形成化合物,从而降低浸出母液中铝、铁杂质离子含量,但不会影响离子型稀土的交换浸出过程。在机理分析的基础上,采用对铝铁杂质有高效抑制效果的抑杂剂LG-01进行离子型稀土矿抑制铝铁杂质的浸出实验研究。结果表明,在不影响离子型稀土矿稀土离子浸出率的情况下,LG-01能有效降低离子型稀土矿浸出母液中铝、铁等杂质离子含量,去除率可达92%。
关键词:离子型稀土;浸出;抑制机理;铝铁杂质
(Edited by Hua YANG)
Foundation item: Project (51164010) supported by the National Natural Science Foundation of China; Project (2010GZC0048) supported by the Natural Science Foundation of Jiangxi Province, China
Corresponding author: Ting-sheng QIU; Tel: +86-79-78312008; E-mail: qiutingsheng@163.com
DOI: 10.1016/S1003-6326(14)63435-X