Trans. Nonferrous Met. Soc. China 24(2014) 1912-1917
Thermodynamics and kinetics of adsorption for heavy metal ions from aqueous solutions onto surface amino-bacterial cellulose
Min LU, Yue-ming ZHANG, Xiao-hui GUAN, Xiao-hui XU, Ting-ting GAO
School of Chemical Engineering, Northeast Dianli University, Jilin 132012, China
Received 17 June 2013; accepted 6 September 2013
Abstract: Amino-bacterial cellulose (amino-BC) was prepared by chemical modification of bacterial cellulose(BC). The adsorption characteristics and mechanism of amino-BC were studied. The results show that adsorption data can be fitted well by Langmuir equation and the pseudo-second order kinetics, indicating that the adsorption of amino-BC would obey monolayer molecule adsorption and the main action was chemisorption��Meanwhile, the adsorption process was studied by the Elovich equation and the intra-particle diffusion model, indicating that the absorption characteristics of metal ions on amino-BC is controlled by both film diffusion and particle diffusion��The increase of reaction temperature will accelerate the adsorbing rate because of endothermic reaction.
Key words: surface amination; bacterial cellulose; adsorption thermodynamics; adsorption kinetics
1 Introduction
Heavy metals found in low concentration wastewater usually exist in nature. An amount of heavy metals go into the atmosphere, water and soil, leading to seriously environmental contamination due to mining, smelting, processing and commercially manufacture. In the meantime, heavy metal pollution will threaten higher life forms including people, because heavy metals have characteristics of enrichment and refractory in the environment. Therefore, people are more and more concerned with pollution of heavy metals. At present, various technologies, such as chemical precipitation, ion exchange, electrochemical process, and adsorption processes, all have been so far proposed and adopted for the removal of heavy metals from wastewater [1-3], while the technology of adsorption for its rapid absorption, low cost, easy operation, no secondary pollution has been widely used in wastewater treatment and recycling of heavy metal ions [4-7]. Bacterial cellulose (BC) has potential to be used as a new adsorbent for effective separation of heavy metal ions [8-10] due to the unique properties including high water holding capacity, fine fiber network, and high tensile strength, no secondary pollution, high specific surface, porous and many hydroxyl groups in the chains. However, BC is not suitable for heavy metal ions adsorption because of lower adsorption capacity and poorer selectivity. Therefore, the modification by new functional groups that will improve the adsorption activity of BC has become one of hot topics. The focal point of the BC-modified technique study is in two aspects: adding chemicals in the process of fermentation and chemical treatment of gelatinous membrane [11-13].
In this work, a new absorbent, surface amino-BC, was prepared by chemical modification. The product was characterized and analyzed. Adsorption characteristics of thermodynamics and kinetics were studied and adsorption mechanism was further discussed to set up proper mathematics models to provide theory to support the practical application of amino-BC in the water treatment field.
2 Experimental
2.1 Materials and equipments
BC was prepared according to the reported method [10,14]. Materials included Pb(NO3)2, CuSO4 and CdSO4 (analytical pure). Equipments included AL204 accurate electronic balance, AA7000 atomic absorption spectrometer, TDL80-2B centrifuge, Nicolet5700 infrared spectroscope, X��Pert Pro X-ray diffractometer and JSM-6390LV scanning electron microscope.
2.2 Methods
2.2.1 Preparation of surface amino-BC
According to the method in Ref. [15], epoxy group bacterial cellulose ether was prepared using BC as raw material. 10 g of Na2CO3 was placed into reaction vessel and diluted with water to 100 mL. 0.1000 g of sulphamate and 1.0000 g of epoxy group bacterial cellulose ether were added respectively to react for 2 h in thermostatic oscillator at 50 ��C. After amination reaction, the product was washed with clean water to be neutral and was dried under the vacuum condition at 60 ��C. The product was amino-BC.
2.2.2 Adsorption
The adsorption process was as follows. 1.0 g of adsorbent amino-BC was placed in 20.00 mL of the solution containing heavy metal ions at pH 5.5 for 60 min. The amount of heavy metal ions adsorbed on amino-BC at equilibrium was calculated to evaluate adsorbent quantity by measuring the difference of heavy metal ions before and after adsorption. And the concentration of heavy metal ions was measured by an atomic absorption spectrophotometer. The adsorption quantity was calculated by Eq. (1).
Q = (��0����t)V/m (1)
where Q is the adsorption quantity of heavy metal ions on adsorbent amino-BC; ��0 and ��t are initial concentration before adsorption and concentration of heavy metal ions at time t in the adsorption process, respectively ; m is the mass of adsorbent; V is the volume of heavy metal ions solution.
2.3 Adsorption isotherms
Linear fitting equations of Langmuir, Freundlich, Slips and Dubinin-Raduskevich isotherm models can be expressed by Eqs. (2)-(5), respectively.
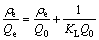 (2)
(2)
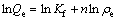 (3)
(3)
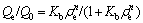 (4)
(4)
lnQe=lnQ0-BA2 (5)
A=RTln(1+1/��e) (6)
where ��e is the equilibrium concentration of heavy metal ions in aqueous solution; Qe is the adsorption capacity of adsorbent at equilibrium; Q0 is the saturated adsorption capacity of heavy meta ions on amino-BC; KL is the Langmuir constant, representing adsorption heat in the adsorption process of adsorbent; Kf and n are the constants of Freundlich isotherm model at a given temperature; Kb is the constant of slips isotherm model; B is the constant of Dubinin-Raduskevich isotherm model, representing average free energy in adsorption process; A is the Polanyi adsorption potential energy, defined as the work done by 1 mol molecular adsorbed from infinity to the distance x between adsorbate and adsorbent surface; R is the mole gas constant; T is the thermodynamic temperature.
2.4 Adsorption thermodynamics
The thermodynamics parameters, ��G, ��S and ��H, can be calculated according to the adsorption equilibrium constant KL as shown in Eqs. (7)-(9).
��G=-RT ln KL (7)
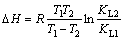 (8)
(8)
 (9)
(9)
where KL1 and KL2 are constants of Langmuir isotherm model at temperature T1 and T2, respectively.
2.5 Adsorption kinetics models
The adsorption mechanism of heavy metal ions on amino-BC are commonly analyzed using Lagergren pseudo-first-order kinetics, pseudo-second-order kinetics, Elovich equation and intra-particle diffusion equation as follows:
ln(Q0-Qt)= lnQ0-k1t (10)
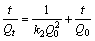 (11)
(11)
Qt=A+kelnt (12)
Qt =kit1/2+C (13)
where Qt is adsorption capacity at time t; k1 and k2are the constants of Lagergren pseudo-first-order and pseudo-second-order kinetics, respectively; ke and A are the constants of Elovich equation; ki is the constant of intra-particle diffusion equation; C is the constant of intra-particle diffusion equation.
3 Results and discussion
3.1 Characterization and analysis of BC and amino- BC
BC and amino-BC were characterized by FT-IR, XRD and SEM, respectively to compare differences before and after modification.
FT-IR spectrum is a useful tool to identify functional groups of molecule. The FT-IR spectra (400- 4000 cm-1) of BC and amino-BC obtained by surface amination are shown in Fig. 1. The major peak located around 3441 cm-1 is attributed to ��OH stretching vibration. The characteristic bands at 1047 cm-1, corresponding to C��O��C stretching vibration, are also observed in BC and amino-BC. Besides, compared with the spectrum of BC, the increase of broad peak around 1375 cm-1 and 3441cm-1 is observed, indicating the existence of ��NH�� and ��NH2 in amino-BC.

Fig. 1 FI-IR spectra of BC and amino-BC
The degree of crystallinity was smaller than that of BC and crystalline structure was weakened (such as 2��=22.5��) after modification [11] as shown in Fig. 2. This fully shows that if we are trying to improve the accessibility and reactivity of cellulose, it is necessary to alkalize BC. The larger the accessibility of cellulose is, the lower the degrees of crystallinity and vice-versa are. It is beneficial to decrease the degree of crystallinity of amino-BC by etherification because structure of hydrogen bonding is destroyed. At the same time, adsorption efficiency is improved by the introduction of amidogen into the structure of BC.
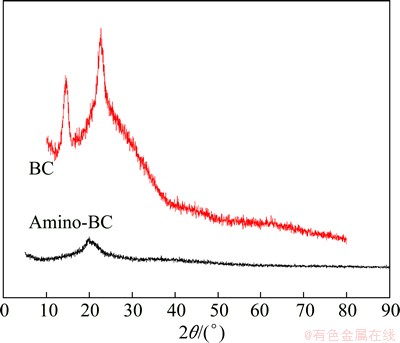
Fig. 2 XRD patterns of BC and amino-BC
The dense network formed with BC is revealed in Fig. 3(a). The width of BC ribbon is 80-100 nm. Compared with BC, amino-BC has a denser network (Fig. 3(b)) and large specific surface area, which would enhance adsorption ability.

Fig. 3 SEM images of BC (a) and amino-BC (b)
3.2 Research on adsorption thermodynamics
3.2.1 Effect of initial concentration of heavy metal ions
Figure 4 presents that the adsorption capacity of heavy metal ions on amino-BC increases with increasing initial concentration. In the start-up phase the adsorption capacity becomes larger with increasing initial concentration, which is attributed to the fact that the adsorbent and heavy metal ions would meet very often in solution and the possibility of adsorption by amino-BC would be improved. After that the slope of curve decreases obviously and the curve flattens out, which might be explained by the fact that metal ions adsorbed on adsorbent and in solution repel each other. The adsorption capacity tends to be equilibrated until the active sites of amino-BC surface are completely covered by adsorption layer. Therefore, amino-BC could be a potentially effective adsorbent for wastewater containing heavy metal ions.
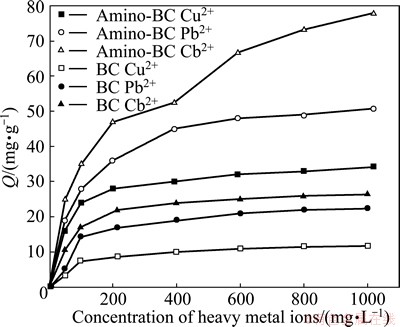
Fig. 4 Effect of initial heavy metal ions concentration on adsorption
3.2.2 Fitting and analysis of adsorption isotherms
It is very important for us to understand adsorption mechanisms by equilibrium adsorption isotherms. Thus, the experimental adsorption data for heavy metal ions on amino-BC were fitted by applying the different equations which are typically used in adsorption of solid-phase adsorbents. And the fitting parameters and correlation coefficients r are shown in Table 1.
Table 1 Fitting parameters of adsorption isotherms models with amino-BC

The Langmuir, Freundlich and Slips adsorption isotherms exhibit an approximately linear relationship for amino-BC. The fitting results of absorption to Cu2+ and Pb2+ by Langmuir equation are much better than other fitting equations, indicating that adsorption process of amino-BC would obey monolayer molecule adsorption. And fitting results of absorption to Cd2+ are much better than those of other fitting equations, indicating that amino-BC has some characteristics of multiphase and asymmetry because crystallinity decreases during BC etherification modification [12]. The adsorption capacity becomes higher with the increase of the KF in Freundlich equation; while adsorption process proceeds more easily with decreasing the value of n due to the fact that the adsorption capacity of Cd2+ is the maximum and easier to be adsorbed.
3.2.3 Adsorption thermodynamics
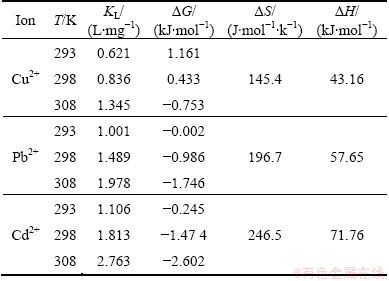
As shown in Table 2, KL increases with the increase in reaction temperature, indicating that adsorption processes are endothermic. ��H values are positive at different temperatures to account for the endothermic adsorption. According to ��G values, adsorption to Pb2+ and Cd2+ is spontaneous in the range of experimental temperature, while adsorption to Cu2+ is spontaneous at 308 K.
Table 2 Parameters of adsorption thermodynamics with amino-BC
3.3 Research on adsorption kinetics
3.3.1 Effect of absorption time
Figure 5 shows the typical results of the time-dependent adsorption performance of BC and amino-BC. The adsorption capacity for heavy metal ions onto BC and amino-BC rapidly increased in the first 30 min and then slowly augmented. The absorption equilibrium has been achieved after more than 60 min, indicating that amino-BC has saturated. Compared with BC, amino-BC has a much higher adsorption capacity, especially for Cd2+ adsorption. The results demonstrate that it is beneficial to increase the adsorption quantity by modification of the surface aminaiton on BC. At the same time, the adsorption capacity is greater with the enhancement of the ability to form complex between heavy metal ions and ��NH (or ��NH2). It is consistent with the previous research [8].
3.3.2 Fitting of adsorption kinetics
Adsorption kinetics was studied to understand adsorption mechanism of amino-BC. Adsorption rate can actually reflect adsorption capacity of adsorbent in unit time, that is, the slope of the tangent line at the point represents instantaneous adsorption rate (dQt/dt) [13].
Table 3 Fitting parameters of adsorption kinemics with amino-BC
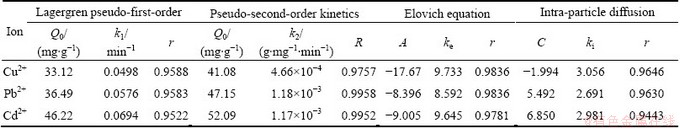
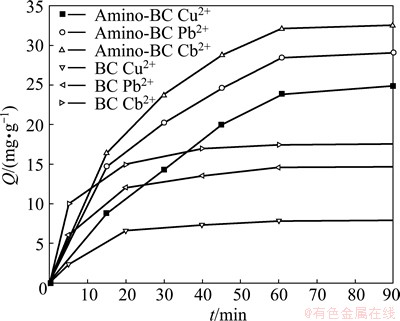
Fig. 5 Effect of absorption time on adsorption
Fitting the relationship between adsorption capacities on amino-BC and absorption time by different adsorption kinetic equations is achieved, and the fitting parameters and correlation coefficients r are shown in Table 3.
As illustrated in Table 3, the adsorption capacity of heavy metal ions with different time could be described according to the pseudo-second-order equation, which indicated that both physical and chemical adsorption existed in adsorption process, but chemical adsorption was dominated. The adsorption capacity of Cd2+was the maximum, while that of Cu2+was the minimum. The k2 values also revealed that adsorption rate was Pb2+>Cd2+> Cu2+. At the same time, fitting the experimental data also showed that adsorption process kinetics was in good relevance to the particle diffusion equation. The ki order was Cu2+>Cd2+>Pb2+, which is the opposite of k2. This demonstrated that the faster the adsorption rate on surface of adsorbent is, the slower the diffusion rate of heavy metal ions into inner adsorbent within a long time. At the same time, because C values are not zero, the adsorption process is controlled not only by internal diffusion of particles but by both film diffusion and particle diffusion.
4 Conclusions
1) Adsorption data can be fitted well by Langmuir equation and the pseudo-second-order kinetics, indicating that the adsorption of amino-BC would obey monolayer molecule adsorption and the main action was a chemisorption.
2) The absorption process of metal ions on amino-BC is controlled by both film diffusion and particle diffusion��And the adsorption process is an endothermic reaction, indicating that the adsorbing rate could be accelerated with the increase of reaction temperature.
References
[1] UYSAL M, AR I. Removal of Cr(VI) from industrial waste waters by adsorption part I: Determination of optimum conditions [J]. Journal of Hazardous Materials, 2007, 149: 482-491.
[2] LI Rong-hua, ZHANG Zeng-qiang, MENG Zhao-fu, LI Hong-yan. Biosorption of Cr(VI) by corn stalk biomass: Thermodynamics and mechanism [J]. Acta Scientiae Circumstantiae, 2009, 29: 1434-1441.
[3] MO Jian-jun, XIONG Chun-hua. Sorption behavior and mechanism of iminodiacetic acid resin for cadmium [J]. The Chinese Journal of Nonferrous Materials, 2006, 16(5): 924-928. (in Chinese)
[4] NASIRUDDIN K M, FAROOQ W M. Characterization of chemically modified corncobs and its application in the removal of metal ions from aqueous solution [J]. Hazardous Materials, 2007, 141: 237-244.
[5] WANG Jiu-si, LAI Feng-xi, HAN Di, ZHANG Peng-hui. Research on ultrasound-ferrite methods for treatment of the wastewater containing Cr6+[J]. Guangzhou Chemical Industry, 2009, 37(8): 193-195. (in Chinese)
[6] FENG Qi-ming, FENG Bo, LU Yi-ping. Influence of copper ions and calcium ions on adsorption of CMC on chlorite [J]. Transactions of Nonferrous Metals Society of China, 2013, 23: 237-242.
[7] FENG Qi-ming, FENG Bo, LU Yi-ping. Influence of copper ions and calcium ions on adsorption of CMC on chlorite [J]. Transactions of Nonferrous Materials Society of China, 2013, 23: 237-242.
[8] ABBAS R, GHOLAMREZA A, MOHAMMAD M. Removal of arsenic using acetobacter xylinum cellulose [J]. Journal of Biological Sciences, 2008, 8: 209-212.
[9] SHEN Wei, CHEN Shi-yan, SHI Shuai-ke, LI Xin, ZHANG Xiang, HU Wei-li, WANG Hua-ping. Adsorption of Cu(II) and Pb(II) onto diethylenetriamine bacterial cellulose [J]. Carbohydrate Polymers, 2009, 75: 110-114.
[10] LU Min, LI Yan-ying, GUAN Xiao-hui, WEI De-zhou. Preparation of bacterial cellulose and its adsorption of Cd2+[J]. Journal of Northeastern University: Natural Science, 2010, 31(8): 1196-1199. (in Chinese)
[11] CHEN Shi-yan, ZOU Yu, YAN Shi-yong, SHEN Wei, SHI Shuai-ke, ZHANG Xiang, WANG Hua-ping. Carboxymethylated-bacterial cellulose for copper and lead ion removal [J]. Journal of Hazardous Materials, 2009, 161: 1355-1359.
[12] LU Min, GUAN Xiao-hui, WEI De-zhou. Removing Cd2+by composite adsorbent of nano-Fe3O4�Mbacterial cellulose [J]. Chemical Research in Chinese Universities, 2011, 27: 1031-1034.
[13] WAN Y Z, HUANG Y, YUAN C D, RAMAN S, ZHU Y, JIANG H J, HE F, GAO C. Biomimetic synthesis of hydroxyapatite/ bacterial cellulose nanocomposites for biomedical applications [J]. Materials Science and Engineering C, 2007, 27: 855-864.
[14] Guan Xiao-hui, YIN Zong-jie, LU Min, LI Yan-ying, WU Bi. Optimization of fermentation conditions for bacterial cellulose production and analysis on its structure [J]. China Brewing, 2010(10): 68-71.
[15] GUAN Xiao-hui, LI Yan-ying, LU Min, XU Ying, WU Bi, GUO Xiao-yu. Use of response surface methodology for optimizing process parameters for the synthesis condition of epoxy group bacterial cellulose ether [J]. Materials Review, 2011, 25: 135-138.
���氱����ϸ����ά�ض�ˮ���ؽ�����������������ѧ�붯��ѧ
³ �����������������ԣ���С�ۣ�������
����������ѧ ��ѧ����ѧԺ������ 132012
ժ Ҫ����ϸ����ά��(BC)Ϊԭ�ϣ�ͨ����ѧ���Է��Ʊ����͡���Ч�Ҽ������������ϡ������氱����ϸ����ά�أ���������б�����������ͬʱ��Cu2+��Pb2+��Cd2+Ϊ�о���������������ѧ�Ͷ���ѧ���Խ����о�����̽������������������ѧģ�͡����������������ϸ����ά�ض�Cu2+��Pb2+��Cd2+���������̸��õط���Langmuir���·��̺�������Ӧ����ѧģ�ͣ�˵����������Ϊ�����Ӳ��������Ի�ѧ��������Ϊ����Elovich���̡���������ɢģ��Ҳ�ܺܺõط�ӳ����ģʽ��˵����������ҺĤ��ɢ����������ɢ��ͬ���õĽ�����������������ȷ�Ӧ���ʵ����߷�Ӧ�¶Ƚ�����������������
�ؼ��ʣ����氱������ϸ����ά�أ���������ѧ����������ѧ
(Edited by Hua YANG)
Foundation item: Project (20130206059G X) supported by Science and Technology Key Project of Jilin Province, China; Project (20101553) supported by the Natural Science Foundation of Jilin Province, China; Project (BSJXM-201226) supported by Doctor Science Research Starting Projects of Northeast Dianli University, China; Project (2013) supported by the 12th Five-Year Enhancing Innovation Projects of Northeast Dianli University, China
Corresponding author: Min LU; E-mail: lumin19770919@163.com
DOI: 10.1016/S1003-6326(14)63271-4