以抗坏血酸作协同试剂时红土镍矿在硫酸溶液中的溶解
来源期刊:中国有色金属学报(英文版)2018年第8期
论文作者:Sait KURSUNOGLU Zela Tanlega ICHLAS Muammer KAYA
文章页码:1652 - 1659
关键词:红土矿;恰尔达红土镍矿;协同试剂;抗坏血酸;镍;钴
Key words:laterite ore; Caldag lateritic nickel ore; synergistic reagent; ascorbic acid; nickel; cobalt
摘 要:研究硫酸与抗坏血酸联用对恰尔达红土镍矿中镍和钴的溶解作用。作为比较,也研究柠檬酸、马来酸和硬脂酸等有机酸作为协同试剂的应用情况。结果表明,抗坏血酸和柠檬酸的使用显著改善了钴的溶解,而其他两种有机酸对浸出率的协同作用很小。对于镍的溶解,抗坏血酸是最有效的协同试剂,其次是柠檬酸、马来酸和硬脂酸。在本研究获得的最优条件下,即1 mol/L硫酸中加入4 g/L抗坏血酸、80 °C、固液比1/10,浸出4 h后钴和镍的浸出率分别达到99%和98%以上。此外,浸出行为对抗坏血酸浓度的变化(2~4 g/L)较不敏感,这从操作角度来看是非常理想的。
Abstract: The dissolution of nickel and cobalt from Caldag lateritic nickel ore using the combination of sulphuric and ascorbic acids was investigated. The use of other organic acids, namely citric, maleic and stearic acids, as synergistic reagents was studied for comparison. The results revealed that the use of ascorbic and citric acids markedly improved the dissolution of cobalt compared to the other two organic acids that only showed slight synergistic effect on the leaching rate. In terms of nickel dissolution, ascorbic acid is the most effective synergist, followed by citric, maleic and stearic acids in descending order. Under the most optimized conditions found in this study, i.e., using 1 mol/L of sulphuric acid with the presence of 4 g/L of ascorbic acid at 80 °C and solid-to-liquid ratio of 1/10, more than 99% and 98% leaching rates of cobalt and nickel, respectively, can be achieved within 4 h of leaching. In addition, the leaching performance is relatively insensitive to the change of ascorbic acid concentration from 2 to 4 g/L which is highly desirable from operational perspective.
Trans. Nonferrous Met. Soc. China 28(2018) 1652-1659
Sait KURSUNOGLU1, Zela Tanlega ICHLAS2, Muammer KAYA3
1. Department of Materials Science & Nanotechnology Engineering, Abdullah Gul University, Kayseri 38100, Turkey;
2. Department of Metallurgical Engineering, Faculty of Mining and Petroleum Engineering, Institut Teknologi Bandung, Bandung 40132, Indonesia;
3. Division of Mineral Processing, Department of Mining Engineering, Eskisehir Osmangazi University, Eskisehir 26480, Turkey
Received 3 August 2017; accepted 4 January 2018
Abstract: The dissolution of nickel and cobalt from Caldag lateritic nickel ore using the combination of sulphuric and ascorbic acids was investigated. The use of other organic acids, namely citric, maleic and stearic acids, as synergistic reagents was studied for comparison. The results revealed that the use of ascorbic and citric acids markedly improved the dissolution of cobalt compared to the other two organic acids that only showed slight synergistic effect on the leaching rate. In terms of nickel dissolution, ascorbic acid is the most effective synergist, followed by citric, maleic and stearic acids in descending order. Under the most optimized conditions found in this study, i.e., using 1 mol/L of sulphuric acid with the presence of 4 g/L of ascorbic acid at 80 °C and solid-to-liquid ratio of 1/10, more than 99% and 98% leaching rates of cobalt and nickel, respectively, can be achieved within 4 h of leaching. In addition, the leaching performance is relatively insensitive to the change of ascorbic acid concentration from 2 to 4 g/L which is highly desirable from operational perspective.
Key words: laterite ore; Caldag lateritic nickel ore; synergistic reagent; ascorbic acid; nickel; cobalt
1 Introduction
Laterite nickel ore is an increasingly important mineral resource for nickel and its valuable by-product, cobalt, due to the depletion of the traditional sulphide ores. The two metals are essential in numerous modern applications such as rechargeable batteries for electric vehicles and modern devices, and superalloys for aerospace and other high-temperature applications. In addition, due to its substantial role in such applications, cobalt has been recognized as a critical metal [1,2] and therefore its recovery from the ore is highly desirable.
Pyrometallurgical and hydrometallurgical routes have been used to produce nickel from laterite nickel ore, but only the latter is able to recover nickel in its metallic form, and thus can be used for the above-mentioned modern applications, and with cobalt as a separate product. High pressure sulphuric acid leaching (HPAL), including its variant such as enhanced pressure acid leaching (EPAL) process, is currently the only operating commercial hydrometallurgical technology to produce nickel and cobalt from laterite nickel ores because the last Caron process operation in Yabulu, Australia, has remained inactive since the early 2016. This technology, however, is at best marginal in terms of process economics due to its high unit of capital cost that makes even a successful operation, such as the Coral Bay HPAL operation, struggle to generate high rate of return on investment [3,4]. Therefore, the development of new processing technologies for laterite nickel ores remains a major research interest.
Many new technologies are being developed to overcome the technical and economic challenges of HPAL such as atmospheric hydrochloric acid leaching (Neomet) process [5], atmospheric nitric acid leaching (Direct Nickel) process [6], high pressure nitric acid leaching process [7] and starved acid leaching technology (SALT) process [4,8], which had been demonstrated at a pilot scale. Reportedly, a small stand-alone heap leaching plant was operated in Yuanjiang, China, in the period of 2014 to 2007 to annually produce 1×104 t of nickel but no public information is available on this operation [9]. In addition, the use of microorganism to enhance the dissolution of nickel and cobalt from laterite nickel ore, i.e., biohydrometallurgical route, has been investigated. In particular, the use of chemilithotrophic bacteria such as Acidithiobacillus thiooxidans and Acidithiobacillus ferroxidans has shown that high nickel and cobalt extractions can be achieved via this route [10-12]. The use of a range of fungal strains such as Aspergillus niger and Penicillium funiculosum has also been investigated [13-15]. The dissolution of the metals is enabled by the fungi ability to excrete organic acid such as citric acid, but its ability to also excrete oxalic acid simultaneously may reduce the leaching efficiency due to low solubility of cobalt and nickel oxalates [8].
The direct chemical leaching of laterite nickel ores with organic acid, instead of using metabolites to generate the acids including citric, oxalic, pyruvic, tartaric, malonic, benzoic, lactic, fumaric, acetic, protocatechuic, gluconic, malic, propionic, adipic, succinic, glumatic, salicylic and formic acids has been reported by a number of investigators [14,16-21]. It is generally agreed that among the mentioned acids, citric acid is the most effective leaching agent for nickel but its leaching efficiency is still far from sufficient. It is, however, reported that as high as 89.6% and 82.9% of nickel and cobalt recoveries, respectively, can be achieved via a sequential organic leaching method using citric acid in the first step and oxalic acid in the second step under atmospheric conditions in relatively short leaching duration [20].
The present authors noted that the use of ascorbic acid, which is readily available at low cost, to leach nickel and cobalt from laterite nickel ore has not been explored and hence, the present study is aimed to do so. The organic acid will be combined with sulphuric acid solutions to leach the ore to investigate the synergistic effect between the two acids as recommended by KURSUNOGLU [22]. The use of citric, maleic and stearic acids was also investigated for comparison.
2 Experimental
2.1 Sample preparation
The lateritic nickel ore was provided by Caldag Nickel in Turkey. The supplied ore was crushed with a jaw crusher and reduced to less than 212 μm using a laboratory ball mill. The ground samples were dry sieved using standard sieve plates and separated into different size fractions via vibration. The obtained fractions were weighed and stored for further studies. All leaching experiments were carried out using only -53 μm particle size fraction.
2.2 Material characterization
Firstly, the particle size distribution of the -53 μm sample was measured using Master Sizer 2000 (Malvern). The chemical composition of the sample was analyzed using X-ray fluorescence (XRF) spectrometer (Philips PW-2404), while its mineralogical composition was examined using X-ray diffractometry (XRD) (Bruker DX Advance). The XRD pattern was confirmed using Fourier transform infrared spectroscopy (FT-IR, Thermo Scientific Nicolet 6700 FT-IR spectrometer). Comprehensive mineralogical description of the used ore was given in the previous studies [20,23-27].
2.3 Experimental procedure
The experimental set-up is shown in Fig. 1. The leaching tests were performed in a 500 mL Erlenmeyer flask covered with aluminium foil to prevent evaporation. The flask was immersed in a temperature-controlled water-bath equipped with ultrasonic vibrator to enhance the leaching kinetics. For each experiment, 25 g of the sample was transferred into the flask before adding 250 mL of sulphuric acid at a pre-determined concentration. The required amount of organic acid was added into the flask and then heated to a desired temperature. During sampling at the selected time intervals, the apparatus was switched off, 5 mL of the slurry was then quickly withdrawn and centrifuged at 7500 r/min. The supernatant was diluted and analyzed using atomic adsorption spectroscopy (AAS, Thermo 3300).

Fig. 1 Experimental set-up
The organic acids used in the experiments namely ascorbic, citric, maleic and stearic acids were prepared from analytical grade reagents (≥100% purity) provided by Sigma Aldrich. The used sulphuric acid solutions were also prepared from an analytical grade reagent (95%-97% purity). In all leaching experiments, the solid-to-liquid ratio was maintained at 1/10. Most of the leaching tests were duplicated to confirm the reproducibility of the test results.
3 Results and discussion
3.1 Materials characterization
The particle size distribution of the sample is shown in Fig. 2. The results showed that 90% of the sample is less than 46 μm. The chemical composition of the sample is given in Table 1.
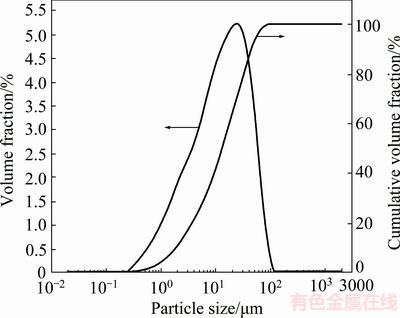
Fig. 2 Particle size distribution of -53 μm sample
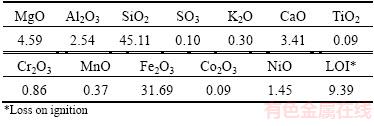
Table 1 Chemical composition of used laterite nickel ore (mass fraction, %)
The XRD analysis (Fig. 3) revealed the presence of quartz, kaolinite and calcite as the major phases. Minor mineral peaks were attributed to goethite, hematite, albite, lizardite, serpentine and asbolane, which are the nickel- and cobalt-rich phases.
FT-IR spectrum of the sample is shown Fig. 4. The absorption at 617 cm-1 is the characteristic of goethite resulting from FeO6 hexagon; those at 793 and 1018 cm-1 are related to quartz because of Si and O bonds; and those at 1642, 2990 and 3424 cm-1 are due to the bonds between H and O. These results are in good agreement with the pattern of laterite nickel ore from Adatepe, Turkey, given by GIRGIN et al [28]. It is therefore verified that quartz and goethite are the two major phases as observed from the absorption peaks in the obtained IR pattern.
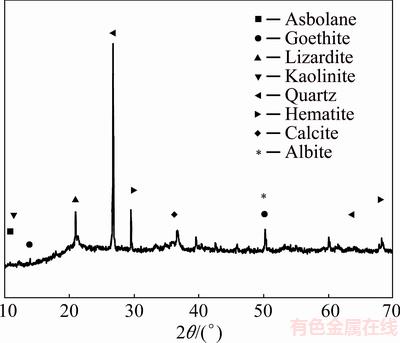
Fig. 3 XRD pattern of -53 μm sample

Fig. 4 FT-IR spectrum of -53 μm sample
3.2 Effect of sulphuric acid concentration and temperature
A series of leaching tests were carried out at various sulphuric acid concentrations (0.5, 1.0 and 1.5 mol/L) and temperatures (60 and 80 °C) as a function of time using the -53 μm sample and a 1/10 solid-to-liquid ratio. Figures 5 and 6 show the effect of sulphuric acid concentration and temperature on the nickel, cobalt and iron dissolutions from the ore sample at 60 and 80 °C, respectively. The leaching rate of the metals increased with increasing concentration of sulphuric acid and leaching time at both temperatures. The dissolution rates of nickel, cobalt and iron were increased from 39.8% to 89.7%, 48.9% to 87.8% and 16.5% to 58.3%, respectively, when the acid concentration increased from 0.5, 1.0 to 1.5 mol/L at 60 °C. Similarly, the dissolution rates of nickel, cobalt and iron were increased from 58.4% to 98.2%, 73% to 99.2% and 29.7% to 73%, respectively, when the acid concentration increased from 0.5, 1.0 to 1.5 mol/L at 80 °C.
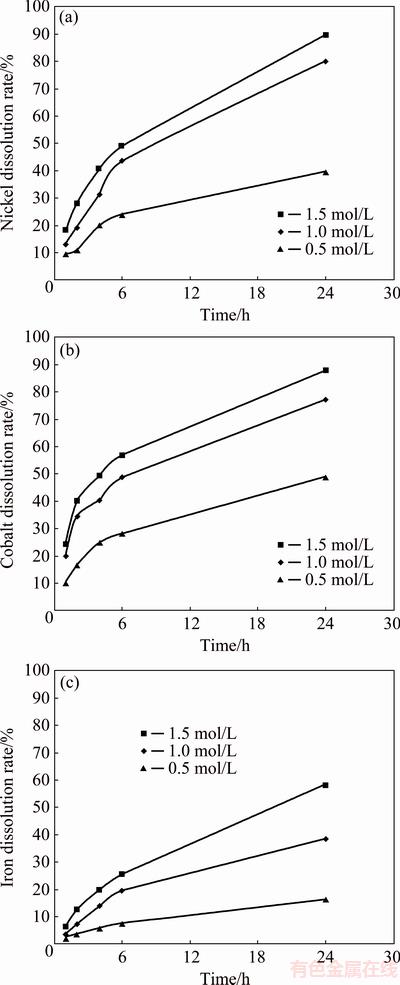
Fig. 5 Dissolution rates of metals only with sulphuric acid at different concentrations and 60 °C
It is interesting to note that the dissolution of metals at 60 °C was relatively significant throughout the leaching duration, but at 80 °C, it was only significant within the first 6 h of leaching and then increased only slightly after leaching for 6 h. This may be due to the diminished amount of acid at the time and hence the leaching rate was reduced. This is supported by the fact that the amount of metals dissolved within the 24 h of leaching at 60 °C was generally lower than that dissolved within the first 6 h at 80 °C and therefore, there was a still sufficient driving force for significant dissolution to occur for the whole duration of leaching at 60 °C. The most probable leaching reactions with sulphuric acid are given in Table 2.

Fig. 6 Dissolution rates of metals with only sulphuric acid at different concentrations and 80 °C
Table 2 Most probable reactions for atmospheric leaching of metal oxides with sulphuric acid

3.3 Synergistic effect of various organic acids on metals dissolution
The synergistic effect of organic acid on the dissolution of nickel, cobalt and iron is shown in Fig. 7. The following conditions were used in this study: 0.5 mol/L sulphuric acid, 4 g/L organic acid and 80 °C. It is clearly seen that the addition of organic acid provides positive impact on the dissolution of the metals. Similar synergistic effect on the combination of organic acids namely acetic, oxalic and lactic acids with sulphuric acid was reported by TZEFERIS [17]. The results showed that the addition of ascorbic acid as synergist gave the highest dissolution rate for the three metals, followed by citric, maleic, and stearic acids. Using ascorbic acid as synergist, cobalt leaching rate reached 99% within the first 6 h, which may be attributed to the reductive role of the acid in the leaching process instead of chelation [29]. The low dissolution of iron, which was only 44.7% in the first 6 h, is desirable but this may also be the reason for the low recovery of nickel, which is 61.2%, as a significant amount of nickel may be trapped within iron-bearing minerals and cannot be leached.
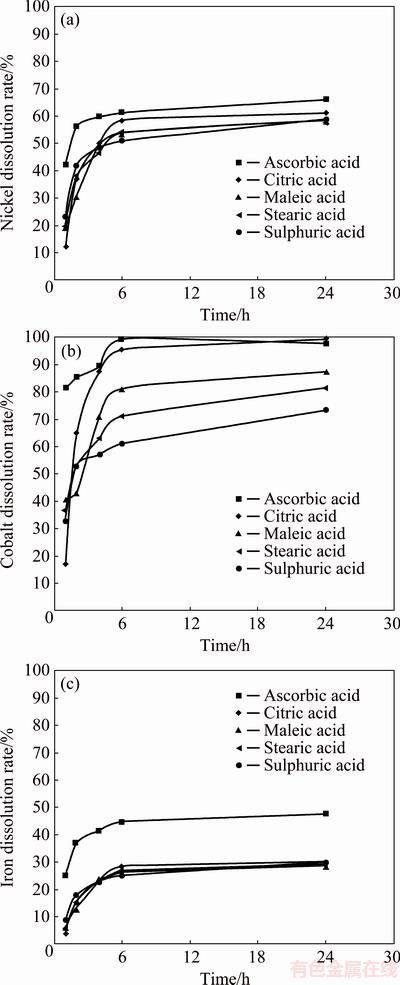
Fig. 7 Dissolution rates of metals with combination of 4 g/L of various organic acids and sulphuric acid concentration of 0.5 mol/L at 80 °C
3.4 Synergistic effect of ascorbic acid on metals dissolution
In this study, the concentrations of sulphuric acid and ascorbic acid were varied to determine the optimal conditions to achieve the highest dissolution rate for nickel and cobalt. Figure 8 shows that the increase of sulphuric acid concentration from 0.5 to 0.75 mol/L and the decrease of ascorbic acid concentration from 4 to either 3 or 2 g/L have a slightly negative impact on the metals dissolution rate. Interestingly, as shown in Fig. 9, when the sulphuric acid concentration is increased to 1 mol/L, the nickel dissolution rate is increased significantly, and as high as 98.1% dissolution rate can be achieved within the first 4 h using 4 g/L ascorbic acid. In addition, complete cobalt extraction under such conditions can be achieved in only 2 h of leaching, while the iron dissolution rate reached a plateau at about 70%.

Fig. 8 Dissolution rates of nickel, cobalt and iron at decreased concentrations of ascorbic acid (3 and 2 g/L), sulphuric acid concentration of 0.75 mol/L and temperature of 80 °C

Fig. 9 Dissolution rates of nickel, cobalt and iron at increased sulphuric acid concentration (1 mol/L) with various ascorbic acid concentrations at 80 °C
The results show a clear sign of synergistic effect of the two acids. The increase in nickel dissolution may be due to the reductive role of ascorbic acid to enhance the dissolution of goethite [30], which is more difficult to be leached than hematite or ferrihydrate [31], with sulphuric acid to liberate the nickel. This is apparent by the fact that the increase in ascorbic acid concentration does not provide significant effect on nickel dissolution. Nevertheless, further characterization study is still required to confirm this.
4 Conclusions
1) The present study showed that the addition of ascorbic, citric, maleic and stearic acids to sulphuric acid leaching of laterite nickel ores provides synergistic effect on the dissolution rates of the nickel and cobalt from laterite nickel ore.
2) Ascorbic acid is the most effective synergist for dissolving nickel and cobalt from the ore followed by citric, maleic and stearic acids in descending order. The superior synergistic effect of ascorbic acid compared to the other three acids is probably due to its ability to the dissolution of goethite.
3) It is shown that for an effective dissolution, the presence of sulphuric acid is necessary as the present study showed that only at sulphuric acid concentration of 1 mol/L, high nickel extraction (98.1%) can be achieved with 4 g/L of ascorbic acid.
4) The leaching performance is relatively insensitive to the change of ascorbic acid concentration from 2 to 4 g/L which is highly desirable from operational perspective.
5) Using ascorbic acid as synergist for sulphuric acid leaching of laterite nickel ore is promising because unlike using oxalic acid, which has been known to enhance the dissolution of goethite, the dissolved nickel will not be precipitated afterwards and remain stable in the aqueous phase.
Acknowledgments
The authors would like to thank S. Dadi from Abdullah Gul University for FT-IR analysis and H. S. Gokcen from Eskisehir Osmangazi University for particle size analysis. Sait is grateful to Professor R. Gurler from Eskisehir Osmangazi University for XRD analysis.
References
[1] European Comission Directorate-General for Internal Market, Industry, Entrepreneurship and SMEs. Study on the review of the list of critical raw materials [R]. European Union, 2017: 38-61.
[2] MUDD G M, JOWITT S M, WERNER T T. The world’s by-product and critical metal resources part I: Uncertainties, current reporting practices, implications and grounds for optimism [J]. Ore Geology Reviews, 2017, 86: 924-938.
[3] TAYLOR A. Technical & cost comparison of laterite treatment processes [C]//Proceedings of ALTA Nickel-Cobalt-Copper Conference 2014. Perth, WA: ALTA Metallurgical Services, 2014: 35-46.
[4] DREISINGER D. Atmospheric leaching of nickel and cobalt from nickel saprolite ores using the starved acid leaching technology [C]//Proceedings of AIP Conference. Bandung, West Java, 2017.
[5] HARRIS B, WHITE C. Recent development in the chloride processing of nickel laterite [C]//Proceedings of Alta Nickel- Cobalt-Copper Conference 2011. Perth, WA: ALTA Metallurgical Services, 2011: 12-24.
[6] BROCK G, MCCARTHY F. DNi test plant program 2013 in review [C]//Proceedings of Alta Nickel-Cobalt-Copper Conference 2014. Perth, WA: ALTA Metallurgical Services, 2014: 80-99.
[7] MA Bao-zhong, WANG Cheng-yan, YANG Wei-jiao, YANG Bo, ZHANG Yong-lu. Selective pressure leaching of Fe(II)-rich limonitic laterite ores from Indonesia using nitric acid [J]. Minerals Engineering, 2013, 45: 151-158.
[8] DREISINGER D, CLUCAS J, REYMENANTS G. The starved acid leaching technology (SALT) for Ni/Co recovery from lean ores and residues [C]//Proceedings of Alta Nickel-Cobalt-Copper Conference 2014. Perth, WA: ALTA Metallurgical Services, 2014: 113-137.
[9] OXLEY A, SMITH M E, CACERES O. Why heap leach nickel laterites? [J]. Minerals Engineering, 2016, 88: 53-60.
[10] COTO O, GALIZIA F,  L, MARRERO J, DONATI E. Cobalt and nickel recoveries from laterite tailings by organic and inorganic bio-acids [J]. Hydrometallurgy, 2008, 94: 18-22.
L, MARRERO J, DONATI E. Cobalt and nickel recoveries from laterite tailings by organic and inorganic bio-acids [J]. Hydrometallurgy, 2008, 94: 18-22.
[11] du PLESSIS C A, SLABBERT W, HALLBERG K B, JOHNSON D B. Ferredox: A biohydrometallurgical processing concept for limonitic nickel laterites [J]. Hydrometallurgy, 2011, 109: 221-229.
[12] HALLBERG K B, GRAIL B M, du PLESSIS C A, JOHNSON D B. Reductive dissolution of ferric iron minerals: A new approach for bio-processing nickel laterites [J]. Minerals Engineering, 2011, 24: 620-624.
[13] BOSECKER K. Leaching of lateritic nickel ores with heterotrophic microorganisms [J]. Acta Biotechnologica, 1987, 7: 389-399.
[14] COTO O, BRUGUERA N,  Y. Bioleaching of Cuban nickeliferous serpentinite [C]// Biohydrometallurgy: Fundamentals, Technology and Sustainable Development (Part A). Amsterdam: Elsevier, 2001: 175-182.
Y. Bioleaching of Cuban nickeliferous serpentinite [C]// Biohydrometallurgy: Fundamentals, Technology and Sustainable Development (Part A). Amsterdam: Elsevier, 2001: 175-182.
[15] VALIX M, USAI F, MALIK R. Fungal bio-leaching of low grade laterite ores [J]. Minerals Engineering, 2001, 14: 197-203.
[16] MCKENZIE D I, DENYS L, BUCHANAN A. The solubilization of nickel, cobalt and iron from laterites by means of organic chelating acids at low pH [J]. International Journal of Mineral Processing, 1987, 21: 275-292.
[17] TZEFERIS P G. Leaching of a low grade hematitic laterite ore using fungi and biologically produced acid metabolites [J]. International Journal of Mineral Processing, 1994, 42: 267-283.
[18] TZEFERIS P G, AGATZINI-LEONARDOU S. Leaching of nickel and iron from Greek non-sulphide nickeliferous ores by organic acids [J]. Hydrometallurgy, 1994, 36: 345-360.
[19] LI Guang-hui, RAO Ming-jun, LI Qian, PENG Zhi-wei, JIANG Tao. Extraction of cobalt from laterite ores by citric acid in presence of ammonium bifluoride [J]. Transactions of Nonferrous Metals Scoiety of China, 2010, 20: 1517-1520.
[20] KURSUNOGLU S, KAYA M. Dissolution behavior of Caldag lateritic nickel ore subjected to a sequential organic acid leaching method [J]. International Journal of Minerals, Metallurgy and Materials, 2015, 22: 1131-1140.
[21] ASTUTI W, HIRAJIMA T, SASAKI K, OKIBE N. Comparison of effectiveness of citric acid and other acids in leaching of low-grade Indonesian saprolitic ores [J]. Minerals Engineering, 2016, 85: 1-16.
[22] KURSUNOGLU S. Recovery of nickel and cobalt from Caldag lateritic nickel ore by leaching and solvent extraction [D]. Eskisehir: Eskisehir Osmangazi University, 2016. (in Turkish)
[23] ARSLAN F, PEREK K T, ONAL G. Acidic leaching of Turkish lateritic nickel ore [C]//Sohn International Symposium on Advanced Processing of Metals and Materials. San Diego: TMS, 2006: 339.
[24] OXLEY A, SIRVANCI N, PURKISS S. Caldag nickel laterite atmospheric heap leach project [J]. Association of Metallurgical Engineers of Serbia, 2007, 13: 5-10.
[25] HELVACI C, GUNDOGAN I, OYMAN T, SOZBILIR H, PARLAK O. Geology, minerology, geomechanical properties of the Caldag Ni-Co laterite deposits [J]. Bulletin of the Earth Sciences, 2013, 32: 101-132.
[26] ONAL M A R, TOPKAYA Y A. Pressure acid leaching of Caldag lateritic nickel ore: An alternative to heap leaching [J]. Hydrometallurgy, 2014, 142: 98-107.
[27] KURSUNOGLU S, KAYA M. Atmospheric pressure acid leaching of Caldag lateritic nickel ore [J]. International Journal of Mineral Processing, 2016, 150: 1-8.
[28] GIRGIN I, OBUT A,  A. Dissolution behaviour of a Turkish lateritic nickel ore [J]. Minerals Engineering, 2011, 24: 603-609.
A. Dissolution behaviour of a Turkish lateritic nickel ore [J]. Minerals Engineering, 2011, 24: 603-609.
[29] SENANAYAKE G, CHILDS J, AKERSTROM B D, PUGAEV D. Reductive acid leaching of laterite and metal oxides-A review with new data for Fe(Ni,Co)OOH and a limonitic ore [J]. Hydrometallurgy, 2011, 110: 13-32.
[30] PARIDA K M, DAS J, DATTA P. Application of statistical design of experiments in the study of dissolution of goethite (α-FeOOH) in hydrochloric acid in the presence of ascorbic acid [J]. Hydrometallurgy, 1997, 46: 271-275.
[31] ZINDER B, FURRER G, STUMM W. The coordination chemistry of weathering: II. Dissolution of Fe(III) oxides [J]. Geochimica et Cosmochimica Acta, 1986, 50: 1861-1869.
Sait KURSUNOGLU1, Zela Tanlega ICHLAS2, Muammer KAYA3
1. Department of Materials Science & Nanotechnology Engineering, Abdullah Gul University, Kayseri 38100, Turkey;
2. Department of Metallurgical Engineering, Faculty of Mining and Petroleum Engineering, Institut Teknologi Bandung, Bandung 40132, Indonesia;
3. Division of Mineral Processing, Department of Mining Engineering, Eskisehir Osmangazi University, Eskisehir 26480, Turkey
摘 要:研究硫酸与抗坏血酸联用对恰尔达红土镍矿中镍和钴的溶解作用。作为比较,也研究柠檬酸、马来酸和硬脂酸等有机酸作为协同试剂的应用情况。结果表明,抗坏血酸和柠檬酸的使用显著改善了钴的溶解,而其他两种有机酸对浸出率的协同作用很小。对于镍的溶解,抗坏血酸是最有效的协同试剂,其次是柠檬酸、马来酸和硬脂酸。在本研究获得的最优条件下,即1 mol/L硫酸中加入4 g/L抗坏血酸、80 °C、固液比1/10,浸出4 h后钴和镍的浸出率分别达到99%和98%以上。此外,浸出行为对抗坏血酸浓度的变化(2~4 g/L)较不敏感,这从操作角度来看是非常理想的。
关键词:红土矿;恰尔达红土镍矿;协同试剂;抗坏血酸;镍;钴
(Edited by Wei-ping CHEN)
Corresponding author: Sait KURSUNOGLU; E-mail: sait.kursunoglu@agu.edu.tr
DOI: 10.1016/S1003-6326(18)64808-3

