
Preparation of micro-sized and uniform spherical Ag powders by
novel wet-chemical method
AN Bing(安 兵)1, 2, CAI Xiong-hui(蔡雄辉) 1, 2, WU Feng-shun(吴丰顺) 1, 2, WU Yi-ping(吴懿平) 1, 2
1. Division of Optoelectronic Materials & Micro/Nano Manufacturing,
Wuhan National Laboratory for Optoelectronics, Wuhan 430074, China;
2. Department of Materials Science and Engineering,
Huazhong University of Science and Technology, Wuhan 430074, China
Received 17 April 2009; accepted 30 June 2009
Abstract:A novel wet-chemical method was presented for the preparation of the micro-sized and uniform spherical Ag powders on a mass-production scale. The well-defined particles were synthesized by mixing the iron(II) sulfate heptahydrate solution with silver nitrate solution directly by high-speed stirring at room temperature. It is found that a large number of micro-sized and uniform spherical particles with rough surfaces are obtained. The mass ratio of iron(II) sulfate heptahydrate to silver nitrate greatly affects the shape of particles, and when it is relatively low, spherical particles cannot be obtained. The reaction temperature has a great impact on the particle size. As the reaction temperature increases from 8 to 15 °C, the mean diameter of particles decreases from 3.5 to 1.6 mm. The additive n-methyl-2-pyrrolidone improves the surface smoothness and compactness of the particles while the particle size is kept unchanged. Scanning electron microscopy, X-ray diffractometry and energy dispersive X-ray analysis were used to characterize the particle products.
Key words: silver powder; wet-chemical method; iron(II) sulfate heptahydrate; micro-sized powder
1 Introduction
The ultra?ne metallic powders are commonly used in electrically conductive paste, solid oxide fuel cells and chemical catalysts, etc. Considerable interests in synthesizing silver powder have been ever-increased during the past few years because of its high electrical conductivity, high melting point, high inoxidizability, excellent solderability and reasonable price. Among the preparation methods of silver powders, wet-chemical reduction route is preferred due to the advantage of controllable size and shape of the particles. A variety of wet-chemical methods were developed to prepare silver powders, however, most of them focused on the synthesis of nano Ag particles. Only a few researches reported the preparation of the micro-sized spherical Ag powders. YAKUTIK and SHEVCHENKO[1] produced the monodispersed spherical silver particles (radius up to 1.5 mm) by using K-Na-tartrate as the reductant for Ag+ in the presence of gelatin. SUN et al[2] prepared largeuniform spherical Ag particles (radius up to 0.95 mm) through directly mixing the AgNO3 aqueous solution with orthophenylenediamine n-methyl-2-pyrrolidone solution at room temperature. Other researchers synthesized Ag microspheres through reducing Ag+ with different reductants, such as ascorbic acid[3-5], hydrazine hydrate[6], water-soluble formates[7] and hydroquinone[8] in the aqueous solution in the presence of the polymeric stabilizers. FISCHER at al[9] employed ethylene glycol to reduce Ag2CO3 at 100 °C and obtained the spherical polymodal Ag powders with diameter of 0.3-4.0 mm. However, these processes often involved multiple steps, or a uniform size of the Ag particles cannot be achieved. A simple method for the preparation of the micro-sized and uniform spherical Ag powders is highly anticipated. Ferrous iron was considered as the reductant for the Ag+ in a few reports; unfortunately, the resultant ultra-fine silver particles were in random geometries[10-11]. In this work, a novel one-step wet-chemical method based on the ferrous iron reductant is presented to produce micro-sized and uniform spherical Ag particles.
2 Experimental
2.1 Material
Silver nitrate (AgNO3, analytical reagent, Shanghai Chemical Reagent Co. Ltd.), iron(II) sulfate heptahydrate (FeSO4×7H2O, analytical reagent, Sinopharm Chemical Regent Co. Ltd.), n-methyl-2-pyrrolidone (C5H9NO, analytical reagent, Tianjin Guangfu Fine Chemical Research Institute) and de-ion water were used as raw materials without further purification.
2.2 Preparation of silver powders
The solutions of silver nitrate and iron(II) sulfate heptahydrate were prepared by dissolving them in de-ion water, respectively. Both of the solutions were cooled to room temperature, and then iron(II) sulfate heptahydrate solution was added in silver nitrate solution with high-speed stirring. After the reaction, the precipitates were separated from the solution, washed and dried in a vacuum dry charber.
The powders were then characterized by SEM, EDX (Sirion 200, FEI, Holand) and XRD (X’Pert PRO, PANalytical B.V, Holand).
3 Results and discussion
3.1 Typical reaction product
Firstly, a typical reaction process was exemplified. The 20 g/L silver nitrate and 88 g/L iron(II) sulfate heptahydrate solutions were cooled to 8 °C and then were mixed and stirred. The mass ratio of Fe2+ to Ag+ was 1.395. The aqueous solution turned into black instantly, and then yellow. The reaction stopped when the color did not change. The products were characterized as shown in Fig.1. It is clear that the powders consist of a large quantity of well-separated, uniform and micro-sized spherical particles (Fig.1(a)). The surface is a little rough (Fig.1(b)). The EDX result indicates that the particles mainly consist of Ag crystal (Fig.1(c)). The particle size distribution histogram shown in Fig.1(d) reveals that the diameter is almost 2-4.5 μm and the mean size is 3.5 mm. Apparently, the powders have a narrow particle size distribution.
The XRD pattern of the particles in Fig.2 shows the significant diffraction peaks located at 38.3?, 44.4?, 64.6?, 77.6? and 81.7?, which correspond to the (111), (200), (220), (311) and (222) crystal planes of Ag, respectively, indicating the formation of silver crystal[12]. The strongest diffraction peak (111) implies that the crystal growth direction of silver seed is favorably oriented parallel to the [111] direction[13].
The formation of colloidal dispersions with narrow size distribution is inferred to accomplish in two stages: first, nano-size primary particles are nucleated in a supersaturated solution, and then aggregate to form larger uniform secondary particles. In the initial stage, the aqueous solution becomes black due to the formation
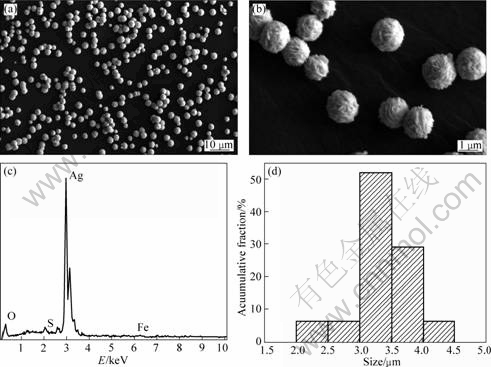
Fig.1 SEM images (a)-(b), EDX spectrum (c) and particle size distribution (d) of silver powders at mass ratio of Fe2+ to Ag+ of 1.395
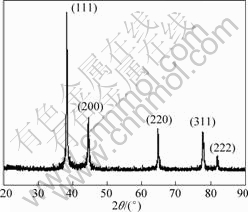
Fig.2 XRD pattern of silver powders
of nano Ag particles. Then SO42- acts as a suitable stabilizer of nano Ag particles, providing a “suitable” condition for the aggregation to form large grains in the aqueous solution. Thus, the color of solution turns yellow slowly with the continuous consumption of Ag+ and the growth of Ag particles. This is in good accordance with the kinetic model proposed by PRIVMAN et al[14] and PARK et al[15].
3.2 Effect of mass ratio of Fe2+/Ag+
Mass ratio of Fe2+ to Ag+ has a great influence on the shape of silver particles. As the concentrations of the iron(II) sulfate heptahydrate solution change to 44 and 176 g/L, respectively, the mass ratios of Fe2+ to Ag+ are 0.698 and 2.79, respectively. The reaction products are shown in Fig.3. When the mass ratio is 0.698, most particles are irregularly shaped (Fig.3(a)), because the primary particles agglomerate incompactly and disorderly. While the mass ratio of Fe2+ to Ag+ is 2.79, the silver particles are spherical (Fig.3(b)), but the size is smaller than that with the mass ratio of 1.395 (see Figs.1(a) and (b)), and the mean diameter is only 2 mm.
The synthesis kinetics of the above reactions is illuminated. Once the two solutions are mixed, the nano Ag nuclei form, and the SO42- and the cations attach on the nano Ag nucleuses successively from the inner to the outer. So, the electrical double layers around the particles are formed in aqueous solutions. The thickness of the layers decreases with the increase of the ionic strength, which leads to the decrease of the range of the repulsion forces between the nano particles. When the mass ratio of Fe2+ to Ag+ is 1.395, the ionic strength is at a rather high level, and the range of the repulsion forces is small. This benefits the aggregation of nano silver particles to form larger spherical secondary particles. When the mass ratio of Fe2+ to Ag+ decreases to 0.698, the ionic strength decreases, and thus the electrical double layer becomes thicker and the range of the repulsion forces enhances.

Fig.3 SEM images of Ag particles obtained at mass ratio of Fe2+ to Ag+ of 0.698 (a) and 2.79 (b)
The silver seeds hardly attract each other to form larger uniform secondary particles. At the same time, the reaction rate decreases because the mass ratio of Fe2+ to Ag+ decreases, which induces the preferred crystal orientation growth of silver seeds. As mentioned above, (111) plane is the dominant crystal growth plane for Ag crystal, and it would grow more rapidly than other planes. Obviously, the Ag crystal preference growth is unfavorable to the formation of compact spherical secondary particles. At last, the nano Ag particles agglomerate incompactly and inhomogeneously, and the spherical micro-sized silver powders are not obtained (Fig.3(a)). Reversely, when the mass ratio of Fe2+ to Ag+ increases to 2.79, the thickness of the electrical double layer decreases, which makes the agglomeration of the nano seeds become relatively easy. At the same time, the reduction rate increases and results in the formation of smaller metal particles due to a larger number of nuclei generation. Finally, the smaller secondary particles are formed (Fig.3(b)), which indicates that the ratio of reactants has a great effect on the morphologies and the size of the Ag particles.
3.3 Effect of reaction temperature
The effect of reaction temperature on the particle size was also examined. As the mass ratio of Ag+ to Fe2+ is 1.395, compared with that at a reaction temperature of 8 °C, the expected Ag particles with uniform size and shape are obtained at 15 °C, as shown in Fig.4. It is clear that the particles are also well-separated uniform micro-spheres (Fig.4(a)). The mean diameter of the particles is 1.6 mm (Fig.4(b)), indicating that higher reaction temperature produces smaller particles. The reason is that higher reaction temperature accelerates the reaction and makes the electrical double layer thinner. Furthermore, higher reaction rate induces more silver seeds to grow in the initial reaction stage, and the thinner electrical double layer leads to the agglomeration of nano seeds more easily. As a result, more particles are synthesized but their sizes turn smaller, which implies the size of the Ag micro-spheres can be adjusted by changing the reaction temperature.

Fig.4 SEM images of Ag powders obtained at 15 °C
3.4 Effect of additive
In order to improve the smoothness of the surface of the particles, the additive n-methyl-2-pyrrolidone was added to the silver nitrate solution with the concentration of 1.0 g/L. Other conditions were identical. The microspheres of the silver with/without the additive are shown in Fig.5.
It is clear that the whole appearance and the size of both particles are almost the same. Only the surface of particles with n-methyl-2-pyrrolidone is more compact and smoother (Fig.5(b)) than that obtained without additive. The additive, polymeric stabilizer, can enwrap on the surfaces of the nano-sized primary particles,

Fig.5 SEM images of spherical silver microcrystals with no additive (a) and 1 g/L n-methyl-2-pyrrolidone (b)
which causes the production of smaller primary nanoparticles. It also affects the aggregation of primary nanoparticles; however, this reaction is far from remarkable under such condition, and a large number of smaller primary nanoparticles are produced in the initial reaction stage. When they aggregate to form larger uniform secondary particles, the final microcrystals become more compact and smoother. It is concluded that the appropriate polymeric stabilizers make the spherical silver microcrystals more compact and smoother and do not change the particle size.
4 Conclusions
1) The uniform micro-sized spherical Ag powders with rough surface can be prepared by a one-step method in which the iron(II) sulfate heptahydrate solution and silver nitrate solution are directly mixed at room temperature.
2) It is found that the mass ratio of reactants plays an important role in the particle shape. If the mass ratio of Fe2+ to Ag+ is relatively low, the spherical micro-sized silver powders cannot obtain. Besides, the reaction temperature determines the particle size at the same mass ratio.
3) Appropriate n-methyl-2-pyrrolidone in the solution as a stabilizer can improve the surface smoothness and compactness of the particles.
4) It is concluded that iron(II) sulfate heptahydrate and SO42- are a suitable reductant and stabilizer for the preparation of the micro-sized spherical Ag particles, respectively.
References
[1] YAKUTIK I M, SHEVCHENKO G P. Self-organization of silver nanoparticles forming on chemical reduction to give monodisperse spheres [J]. Surface Science, 2004, 566/567/568: 414-418.
[2] SUN Xu-ping, DONG Shao-jun, WANG Er-kang. Rapid preparation and characterization of uniform, large, spherical Ag particles through a simple wet-chemical route [J]. Journal of Colloid and Interface Science, 2005, 290(1): 130-133.
[3] VELIKOV K P, ZEGERS G E, van BLAADEREN A, van BLAADEREN A. Synthesis and characterization of large silver particles [J]. Langmuir, 2003, 19(4): 1384-1389.
[4] YANG Jin-hu, QI Li-min, ZHANG Dong-bai, MA Ji-ming, CHENG Hu-min. Dextran-controlled crystallization of silvermicrocrystals with novel morphologies [J]. Crystal Growth and Design, 2004, 4(6): 1371-1375.
[5] HALACIUGA I, GOIA D V. Preparation of silver spheres by aggregation of nanosize subunits [J]. Journal of Materials Research, 2008, 23(6): 1776-1784.
[6] UEYAMA R, HARADA M, UEYAMA T, HARADA A, YAMAMOTO T, SHIOSAKI T, KURIBAYASHI K. Synthesis of monodispersed spherical single crystalline silver particles by wet chemical process[J]. Journal of Ceramic Society of Japan, 1999, 107(1): 60-65.
[7] GURAY T S, HOWARD D G. Process for making finely divided particles of silver metal[P]: US Patent, 4979985. 1990-12-25.
[8] AHN J G, KIM D J, LEE J R, JUNG H S, KIM B G. Synthesis of mono-dispersed spherical silver powders by chemical-reduction method [J]. Materials science forum, 2007, 539/543: 2782-2786.
[9] FISCHER C, HELLER A, DUBE G. Preparation of silver powder with specific properties by reduction in organic medium: Reduction of silver carbonate by ethylene glycol [J]. Materials Research Bulletin, 1989, 24(10): 1271-1277.
[10] BERADZE D L, SHAPLYGIN I S. Production of very fine silver powders by reduction from solution [J]. Zhurnal Neorganicheskoi Khimii. 1984, 29(11): 2988-2989.
[11] SHAPLYGIN I S, KURNYAVKO V K. Manufacture of fine metallic silver powders by a reduction technique [J]. Powder Metallurgy and Metal Ceramics, 1973, 12(7): 517-520.
[12] LI Ling-yin, CAO Xue-bo, YU Fei, YAO Zhen-yu, XIE Yi. G1 dendrimers-mediated evolution of silver nanostructures from nanoparticles to solid spheres [J]. Journal of Colloid and Interface Science, 2003, 261(2): 366-371.
[13] SUN Xu-ping, DONG Shao-jun, WANG Er kang. Large-scale synthesis of micrometer-scale single-crystalline Au plates of nanometer thickness by a wet-chemical route [J]. Angewandte Chemie International Edition, 2004, 43(46): 6360-6363.
[14] PRIVMAN V, GOIA D V, PARK J, MATIJEVI? E. Mechanism of Formation of monodispersed colloids by aggregation of nanosize precursors [J]. Journal of Colloid and Interface Science, 1999, 213(1): 36-54.
[15] PARK J, PRIVMAN V, MATIJEVI? E. Model of Formation of Monodispersed Colloids [J]. Journal of Physical Chemistry B, 2001, 105(47): 11630-1163.
(Edited by FANG Jing-hua)
Foundation item: Project(2006AA04A110) supported by the National High-tech Research and Development Program of China; Project(60976076) supported by the National Natural Science Foundation of China
Corresponding author: AN Bing; Tel: +86-27-87544454, E-mail: anbing@mail.hust.edu.cn
DOI: 10.1016/S1003-6326(09)60337-X