J. Cent. South Univ. Technol. (2008) 15: 204-208
DOI: 10.1007/s11771-008-0039-2
Industrial experiment of copper electrolyte purification by copper arsenite
ZHENG Ya-jie(֣�Ž�)1, XIAO Fa-xin(Ф����)1, WANG Yong(�� ��)1, 2,
LI Chun-hua(���)1, 2, XU Wei(�� ��)1, 2, JIAN Hong-sheng(�����)1, 2, MA Yu-tian(������)1
(1.School of Metallurgical Science and Engineering, Central South University,
Changsha 410083, China;
2. Daye Nonferrous Metal Limited Company, Huangshi 435005, China)
Abstract: Copper electrolyte was purified by copper arsenite that was prepared with As2O3. And electrolysis experiments of purified electrolyte were carried out at 235 and 305 A/m2, respectively. The results show that the yield of copper arsenite is up to 98.64% when the molar ratio of Cu to As is 1.5 in the preparation of copper arsenite. The removal rates of Sb and Bi reach 74.11% and 65.60% respectively after copper arsenite is added in electrolyte. The concentrations of As, Sb and Bi in electrolyte nearly remain constant during electrolysis of 13 d. The appearances of cathode copper obtained at 235 and 305 A/m2 are slippery and even, and the qualification rate is 100% according to the Chinese standard of high-pure cathode copper(GB/T467-97).
Key words: copper arsenite; copper electrolyte; purification; electrolysis: industrial experiment
1 Introduction
Recently, with the rapid development of copper smelting industry, the competition of raw material gets fiercer in China, which induces the increase of impurities in raw material. The impurities of Sb and Bi cannot be removed in pyrometallurgy process, which results in high contents of Sb and Bi in anode copper, therefore Sb and Bi can only be removed by purification methods in electrorefining process[1-4]. At present, most copper smeltings still adopt electrowinning to remove impurities in copper electrolyte[5-7]. In this method, current efficiency is low and energy consumption is high[8-9]. To the worse, the toxic arsine gas is produced at the end of electrowinning[10-11]. Arsenic, antimony and bismuth in the electrolyte mostly exist in the forms of 
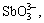
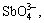 SbO+ and BiO+. These ions easily interact to form arsenate, antimonate and other insoluble compounds[12-13].
SbO+ and BiO+. These ions easily interact to form arsenate, antimonate and other insoluble compounds[12-13].
ZHENG et al[14-15] prepared copper arsenite with As2O3 and waste water containing arsenic. The results of laboratorial experiments show that the concentrations of Sb and Bi decrease greatly after copper arsenite is added. In this work, the industrial experiment of electrolyte purification by copper arsenite was performed based on the laboratorial experiments.
2 Experimental
2.1 Materials and main equipment
The following materials were used in the industrial experiment: industrial grade arsenic trioxide, sodium hydroxide, copper sulfate, concentrated sulfuric acid, copper electrolyte, copper anode (0.75 m��0.78 m) and starting sheet (0.77 m��0.81 m). The main equipment in the industrial experiments consisted of stainless-steel reactor (d 2.0 m��2.5 m), agitator, heater, pump, elec- trolytic cell (3.60 m��0.89 m��1.40 m), belt filter (1.1 m2) and box filter (40 m2).
2.2 Preparation of copper arsenite
Copper arsenite was prepared by dissolving copper sulphate in arsenious solution that was made from As2O3 and NaOH solution at 1.5 of the molar ratio of Cu to As. Concentrated H2SO4 and NaOH solution were used to adjust pH value of solution. The product was sent for washing and filtering and the contents of Cu and As were quantitatively analyzed by chemical analysis.
2.3 Purification of copper electrolyte by copper arsenite
The purification experiment was carried out by dissolving copper arsenite in electrolyte in a stainless-
steel reactor under stirring (200-400 r/min) for several hours at 60-70 ��. The purified electrolyte was then leached to remove the precipitate. The flow sheet of purification of copper electrolyte by copper arsenite is shown in Fig.1.
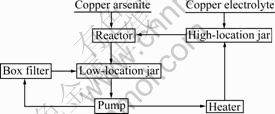
Fig.1 Flow sheet of electrolyte purification by copper arsenite
2.4 Electrolysis
The additive was added into purified electrolyte and the electrolysis experiment was carried out at 235 A/m2 for 7 d and at 305 A/m2 for 6 d. The quality of cathode copper was inspected after experiments. Six electrolytic cells were used in this experiment and the bulk of each cell was 3.5 m3. The quantity of anode copper and starting sheet in each cell was 28 and 27, respectively. The electrode distance was 95 mm, electrolysis temperature was 62-64 �� and the circulation speed of electrolyte was about 7.14 L/(min��m3).
3 Results and discussion
3.1 Preparation of copper arsenite
The amounts of material and yield of copper arsenite during the experiment are listed in Table 1 and the chemical compositions of product are listed in Table 2.
Table 1 Amounts of material and yield of product in prepara- tion of copper arsenite

Table 2 Compositions of copper arsenite (mass fraction, %)

It can be seen that the yield of copper arsenite is 98.64%, the mass fractions of arsenic, copper and water are 5.25%, 6.84% and 68.61%, respectively, in the product of copper arsenite. The following reactions occur after As2O3 is added into NaOH solution:
As2O3+2OH-=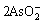 +H2O (1)
+H2O (1)
As2O3+6OH-=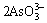 +3H2O (2)
+3H2O (2)
The components of copper arsenite are various, e.g. CuHAsO3, Cu3(AsO3)2 and Cu(AsO2)2[14], therefore, the following reactions occur when copper sulfate is added.
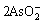 +Cu2+=Cu(AsO2)2�� (3)
+Cu2+=Cu(AsO2)2�� (3)
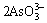 +3Cu2+=Cu3(AsO3)2�� (4)
+3Cu2+=Cu3(AsO3)2�� (4)
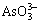 +H++Cu2+=CuHAsO3�� (5)
+H++Cu2+=CuHAsO3�� (5)
The molar ratio of copper to arsenic in the product is 1.54, which accords with that of Cu3(AsO3)2 approximately. The contents of arsenic and copper in filtrate are 0.31 and 0.28 g/L, respectively. This product was sent to wastewater workshop for further disposal.
3.2 Purification of electrolyte by copper arsenite
The compositions of copper electrolyte before and after purification are listed in Table 3 and the influences of arsenic on concentration of antimony and bismuth in electrolyte are presented in Fig.2 and Fig.3, respectively.
Table 3 Composition of copper electrolyte before and after purification (��/(g?L-1))
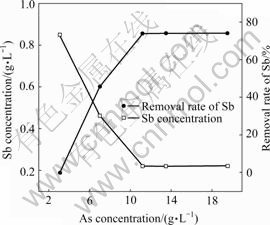
Fig.2 Influence of As concentration on removal rate of Sb
It can be seen that the concentration of Sb decreases from 0.85 to 0.22 g/L and Bi from 0.220 to 0.086 g/L when concentration of As increases from 3.10 to 11.16 g/L and the removal rates of Sb and Bi reach 74.11% and
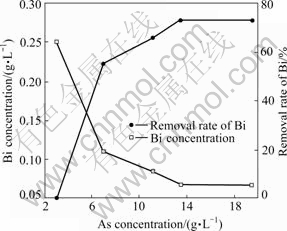
Fig.3 Influence of As concentration on removal rate of Bi
65.60%, respectively. It can be seen from Fig.2 and Fig.3 that the removal rates of Sb and Bi increase with the increase of concentration of As up to 11.16 g/L and remain constant with further increase. Therefore, the suitable As concentration is 11.16 g/L in copper electrolyte purification by copper arsenite.
The compositions of the precipitate were determined chemically[16]. The result shows that the molar ratio of nAs(III)?nSb(III)?nSb(V) of the precipitate is 3?2?4. The XRD (D/max�CrA, Rigaku Corporation of Japan) result of the precipitate is shown in Fig.4. The FTIR spectrum (NExus670, Nicolet Corporation) of the precipitate is shown in Fig.5.
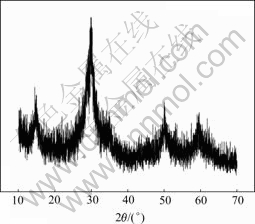
Fig.4 XRD pattern of precipitate
From Fig.4, it can be seen that the crystal performance of the precipitate of industrial experiment is not so good. It can be seen from Fig.5 that the FTIR bands at 3 418.29 cm-1 and 1 640.86 cm-1 are the symmetrical and anti-symmetrical stretching vibration absorption spectra of O��H[17], the band at 1 261.87 cm-1 is the bending vibration absorption spectrum of As��OH
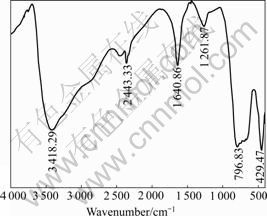
Fig.5 FTIR spectrum of precipitate
and Sb��OH[18-19], the bands at 796.83 and 429.47 cm-1 are the anti-symmetrical stretching vibration absorption spectra of As-O-Sb and Sb��O��Y (Y=As, Sb) respect- tively[18, 20]. Therefore, the valence bands of As-OH, Sb��OH, As��O��Sb, Sb��O��Sb, O��H are included in the structure of the precipitate.
According to FTIR results, the precipitate is antimony arsantimonate and its chemical structure can be described as Sb(OH)2��O��[Sb(OH)3��(O��As(OH)�� O��Sb(OH)3)3]��O��Sb(OH)2��8H2O since the nAs(III)?nSb(III)? nSb(V) of the precipitate is 3?2?4. The reaction of forming the precipitate is shown as follows:
3H2O+2HSO4-+3HAsO2+2SbO++4HSb(OH)6��Sb(OH)2��O��[(Sb(OH)3��(O��As(OH)��O��Sb(OH)3)3)��O��Sb(OH)2��8H2O+2H2SO4
Therefore, Sb and Bi can be removed after copper arsenite is added.
3.3 Concentrations of As, Sb and Bi in electrolyte during electrolysis
The influences of time on the concentrations of total As (AsT), As(��), Sb and Bi in electrolyte are listed in Table 4.
It can be seen that the As(��) concentration decreases from 7.62 to 1.20 g/L, total As, Sb and Bi are in the range of 10.81-11.55 g/L, 0.19-0.28 g/L and 0.066-0.110 g/L, respectively during electrolysis of 13 d without additional purification.
In the process of electrolysis, anode copper dissolves continuously and the trivalent As, Sb and Bi enter into electrolyte. However the concentrations of As, Sb and Bi remain constant. This indicates that the electrolyte itself has fine self-purification function and Sb and Bi are removed greatly in the form of anode slime.
Table 4 Influence of electrolysis time on concentrations of AsT, As(��), Sb and Bi (��/(g��L-1))

3.4 Quality of cathode copper
The electrolysis experiments were carried out at 235 and 305 A/m2 respectively to respect the influences of arsenic on electrolysis and the pictures (SONY Digital Camera, DSC-T200) are shown in Figs.6-8.

Fig.6 Picture of cathode copper gained from electrolyte without copper arsenite at 235 A/m2

Fig.7 Picture of cathode copper gained from electrolyte with copper arsenite at 235 A/m2
In Figs.6-8, there are some particles on the surface of cathode copper from electrolyte without copper arsenite, the surface of cathode copper obtained at 235 and 305 A/m2 from electrolyte with copper arsenite is even, and the former is more slippery than the latter. The qualification rate of cathode copper from electrolyte with copper arsenite is 100% according to the Chinese standard of high-pure cathode copper (GB/T467-97).

Fig.8 Picture of cathode copper gained from electrolyte with copper arsenite at 305 A/m2
The electrolysis results indicate that fine floating slime of SbAsO4 and BiSbO4 forms easily in the electrolyte when concentrations of Sb and Bi excede 0.50 g/L. Therefore, the cathode copper deteriorates with antimony and bismuth contamination in copper electrorefining. The results of electrolysis show that concentrations of Sb and Bi are less than 0.28 and 0.11 g/L respectively, which benefit the quality of cathode copper. Obviously, the technology of electrolyte purification by copper arsenite has the advantages of convenient operation, fine purification performance and low cost, so it can be expected to apply to industry broadly.
4 Conclusions
1) The yield of copper arsenite is 98.64% when the molar ratio of Cu to As is 1.5 in the preparation of copper arsenite with As2O3. The molar ratio of copper to arsenic of copper arsenite is 1.54, which accords with that of Cu3(AsO3)2 approximately.
2) The Sb concentration decreases from 0.85 to 0.22 g/L and Bi from 0.220 to 0.086 g/L when As concentra- tion increases from 3.10 to 11.16 g/L and the removal rates of Sb and Bi reach 74.11% and 65.60% respectively after copper arsenite is added in electrolyte.
3) The concentration of As(��) decreases gradually, the concentrations of total As, Sb and Bi nearly remain constant during electrolysis of 13 d. The appearance of cathode copper from electrolyte with copper arsenite at 235 and 305 A/m2 is slippery and even. The qualification rate of cathode copper is up to 100% according to the Chinese standard of high-pure cathode copper (GB/ T467-97).
References
[1] WANG Xue-wen, CHEN Qi-yuan, YIN Zhou-lan, ZHANG Ping-min, LONG Zi-ping, SU Zhong-fu. Removal of impurities from copper electrolyte with adsorbent containing antimony [J]. Hydrometallurgy, 2003, 69(1/3): 39-44.
[2] WANG Xue-wen, CHEN Qi-yuan, YIN Zhou-lan, XIAO Lian-sheng. Identification of arsenato antimonates in copper anode slimes [J]. Hydrometallurgy, 2006, 84(3/4): 211-217.
[3] PETKOVA E N. Mechanisms of floating slime formation and its removal with the help of sulphur dioxide during the electrorefining of anode copper [J]. Hydrometallurgy, 1997, 46(3): 277-286.
[4] NAVARRO P, SIMPSON J, ALGUACIL F J. Removal of antimony (��) from copper in sulphuric acid solutions by solvent extraction with LIX 1104SM [J]. Hydrometallurgy, 1999, 53(2): 121-131.
[5] RIVEROS G, UTIGARD T A. Disposal of arsenic in copper discharge slags [J]. Journal of Hazardous Materials, 2000, 77(1): 241-252.
[6] PANDA B, DAS S C. Electrowinning of copper from sulfate electrolyte in presence of sulfurous acid[J]. Hydrometallurgy, 2001, 59(1): 55-67.
[7] QIU Yong-hai, TANG Ren-heng, CHEN Bai-zhen. Evolution potential of arsine[J]. The Chinese Journal of Nonferrous Metals, 2002, 10(1): 101-104. (in Chinese)
[8] NAVARRO P, ALGUACIL F J. Adsorption of antimony and arsenic from a copper electrorefining solution onto activated carbon [J]. Hydrometallurgy, 2002, 66(1): 101-105.
[9] RAGHAVAN R, BHATT C V. Comparative study of certain ion-exchange resins for application in copper-bearing process solutions[J]. Hydrometallurgy, 1998, 50(2): 169-183.
[10] LIN H K. Extraction of antimony by a copper chloride extractant[J]. Hydrometallurgy, 2004, 73(3): 283-291.
[11] WANG Xue-wen, CHEN Qi-yuan, LONG Zi-ping, SU Zhong-fu, YIN Zhou-lan, ZHANG Ping-min. Application of antimony in purification of copper electrolyte[J]. The Chinese Journal of Nonferrous Metals, 2002, 12(6): 1277-1280. (in Chinese)
[12] WANG Xue-wen. Study on the mechanism of the formation and action of arsenate antimonic acid in copper electrorefining[D]. Changsha: School of Chemistry and Chemical Engineering, Central South University, 2003: 1-2. (in Chinese)
[13] XIAO Bing-rui, SU Zhong-fu, LI Yi-huang, LONG Zi-ping, HUANG Ming-jin. A new purification method of copper electrolyte: CN, 02129694.4[P]. 2003-03-05. (in Chinese)
[14] ZHENG Ya-jie, XIAO Fa-xin, WANG Yong, BAO Xia-jie. Preparation and application of copper arsenite: CN, 200610031980.7[P]. 2006-07-19. (in Chinese)
[15] ZHENG Ya-jie, WANG Yong, ZHAO Pan-feng. A method of preparing copper arsenate and copper arsenite from waste water containing arsenic: CN, 200610032456.1[P]. 2006-10-25. (in Chinese)
[16] XIAO Fa-xin, ZHENG Ya-jie, XU Wei, LI Chun-hua, JIAN Hong-sheng. Novel technology of purification of copper electrolyte[J]. Trans Nonferrous Met Soc China, 2007, 17(5): 1069-1074.
[17] NAILI H, MHIRI T. X-ray structural, vibration and calorimetric studies of a new rubidium pentahydrogen arsenate RbH5(AsO4)2 [J]. J Alloys Comp, 2001, 315: 143-149.
[18] QURESHI M, KUMAR V. Synthesis and IR, X-ray and ion-exchange studies of some amorphous and semi-crystalline phases of titanium antimonate: Separation of VO2+ from various metal ions [J]. Journal of Chromatography A, 1971, 62(3): 431-438.
[19] MYNENI S C, TRANINA S J, WAYCHUNAS G. Experimental and theoretical vibration spectroscopic evaluation of arsenate coordination in aqueous solutions, solids and at mineral-water interfaces [J]. Geochemical et Cosmochimica Acta, 1998, 62: 3285-3300.
[20] COLOMBAN P H, DOREMIEUX M, PIFFARD Y. Equilibrium between photonic species and conductivity mechanism in antimonic acid H2 Sb4O11��nH2O [J]. J Mol Struct, 1989, 213: 83-96.
(Edited by YANG Hua)
Foundation item: Project(200501045) supported by Innovation Fund of Hubei Daye Nonferrous Metal Limited Company of China
Received date: 2007-11-15; Accepted date: 2007-12-29
Corresponding author: ZHENG Ya-jie, Professor; Tel: +86-731-8836285; E-mail: zzyyjj01@yahoo.com.cn